Infective endocarditis from Davidson
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
I have comprehensive content. The large read_file from Harrison's block15 was truncated but contained rich data on diagnosis, Duke criteria, imaging, and treatment. Let me now compile the full IE overview from what's been retrieved.
Note: Davidson's Principles and Practice of Medicine is not in the library catalog. The content below is drawn from Harrison's Principles of Internal Medicine 22e (2025) and Goldman-Cecil Medicine, which cover infective endocarditis in equivalent depth and style.
Infective Endocarditis
(Harrison's Principles of Internal Medicine 22e & Goldman-Cecil Medicine)
Definition & The Lesion
The hallmark lesion of infective endocarditis (IE) is the vegetation — a mass of platelets, fibrin, microorganisms, and scant inflammatory cells on the endocardial surface of the heart.
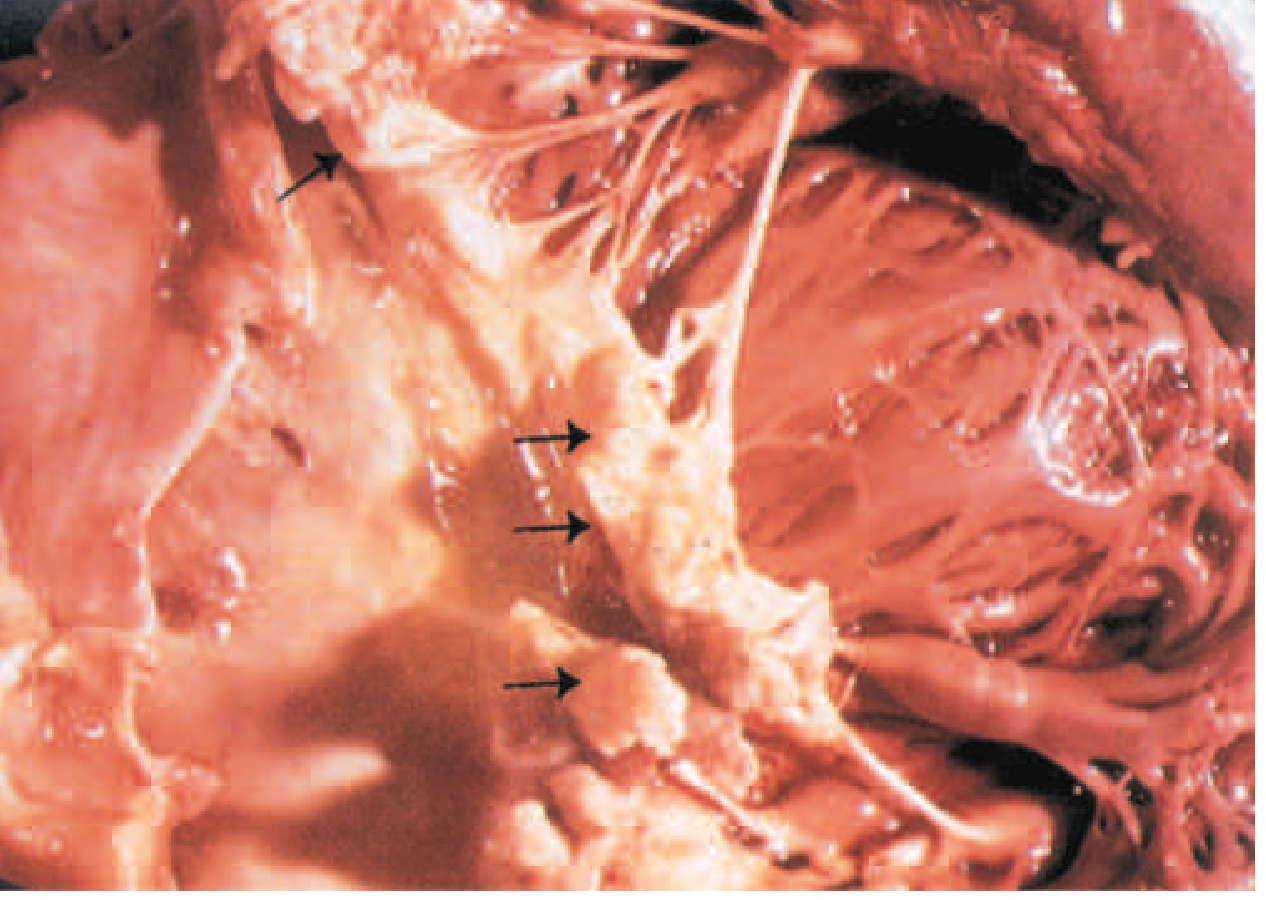
Infection most commonly involves heart valves, but may affect:
- The low-pressure side of a ventricular septal defect
- Mural endocardium (damaged by aberrant jets or foreign bodies)
- Intracardiac devices (pacemakers, defibrillators, CIEDs)
The analogous process on arteriovenous or arterio-arterial shunts/coarctation is called infective endarteritis.
Classification
| Type | Features |
|---|---|
| Acute IE | Hectically febrile; rapidly damages cardiac structures; seeds extracardiac sites; fatal in weeks if untreated |
| Subacute IE | Indolent course; slow structural damage; rarely metastasizes; gradually progressive unless complicated by major embolism or ruptured mycotic aneurysm |
| Native Valve IE (NVE) | Most common form |
| Prosthetic Valve IE (PVE) | Early (<2 months post-surgery) vs. Late (>2 months); risk greatest in first year after valve replacement |
| CIED-IE | Cardiovascular implantable electronic device infection |
| Health care-associated IE | From IV catheters, hyperalimentation lines, pacemakers, hemodialysis |
Epidemiology
- Incidence: ~15 cases per 100,000 population/year in developed countries (rising)
- USA: ~10,000–20,000 new cases/year
- ~25–35% of NVE cases are health care-associated in developed countries
- 16–30% of all cases are prosthetic-valve infections
- Recent acceleration in mortality in ages 25–44 years, linked to opioid use disorder and injection drug use
- S. aureus is now the most common causative organism in developed countries
Predisposing Conditions
More common:
- Mitral valve prolapse (especially with regurgitation)
- Degenerative valvular disease
- Injection drug use
- Congenital heart disease (uncorrected VSD)
- Previous endocarditis
Less common:
- Rheumatic heart disease
- Hypertrophic obstructive cardiomyopathy (HOCM)
- Coarctation of the aorta
- Complex cyanotic congenital heart disease
Etiology & Microbiology
| Organism | Notes |
|---|---|
| S. aureus | Most common in developed countries; injection drug users, healthcare-associated; acute course; 10–15% of bacteraemias develop IE |
| Viridans streptococci | Oral cavity source; classic subacute IE |
| S. gallolyticus (formerly S. bovis biotype 1) | GI tract origin; associated with colonic polyps/adenocarcinoma — mandates colonoscopy |
| Enterococci (>90% E. faecalis) | GU tract source; subacute; develops in ~25% of E. faecalis bacteraemia |
| HACEK group | Haemophilus spp., Aggregatibacter spp., Cardiobacterium hominis, Eikenella corrodens, Kingella kingae; ~5% of cases; grow within 7 days in modern blood culture systems |
| Coagulase-negative staphylococci | Uncommon in NVE; important in PVE and intracardiac devices |
| Fungi (Candida, Aspergillus) | Injection drug use, prolonged IV catheters, cardiac surgery; high mortality; usually require valve surgery |
Prosthetic Valve IE organisms:
- Early PVE (<2 months): S. aureus dominates — perioperative contamination
- Late PVE (>2 months): spectrum resembles community-acquired NVE (S. aureus, viridans streptococci); oxacillin-resistant coagulase-negative staphylococci common
Clinical Features
Symptoms
- Fever (most common), rigors, night sweats, malaise, anorexia, weight loss
- New or worsening cardiac murmur (critical finding)
- Symptoms of heart failure in severe cases
Peripheral Manifestations (embolic/immunologic)
| Sign | Description |
|---|---|
| Petechiae | Nonblanching spots on skin/mucosae |
| Osler nodes | Small, painful, raised lesions on finger/toe pads (immune complex deposition) |
| Janeway lesions | Hemorrhagic, non-tender macules on palms and soles (embolic origin) |
| Splinter hemorrhages | Linear brownish-red subungual lesions parallel to nail growth (non-specific) |
| Roth spots | Oval retinal hemorrhages with pale centres on fundoscopy |
| Splenomegaly | More common in subacute IE |
| Clubbing | In chronic/subacute cases |
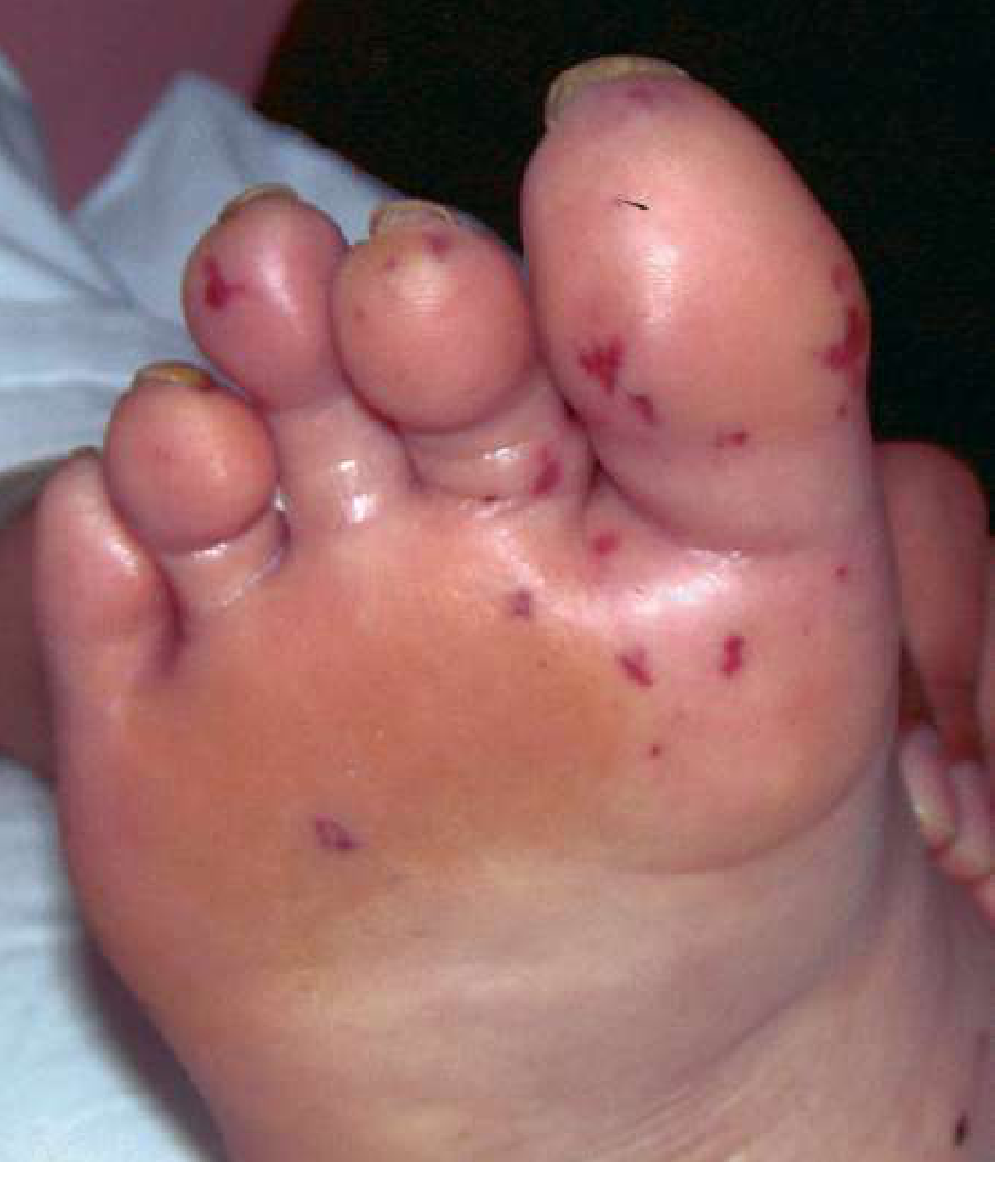
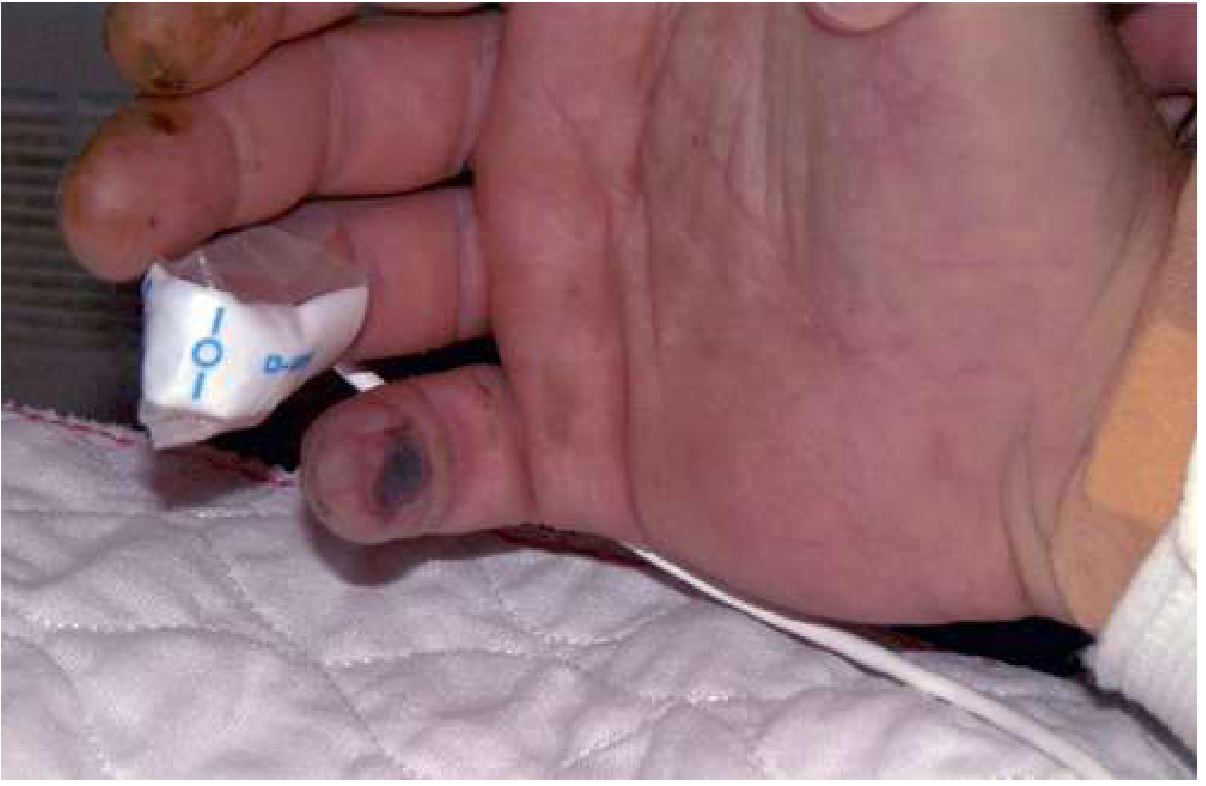
Neurological Complications
- 15–20% of patients develop stroke or stroke-like symptoms
- Major vessel embolism, cranial nerve palsies, visual field defects, toxic-metabolic encephalopathy
Diagnosis
Modified Duke Criteria (the clinical gold standard)
Major Criteria:
- Positive blood cultures — typical organisms (S. aureus, viridans streptococci, HACEK, S. gallolyticus, enterococci) from ≥2 separate cultures; or persistently positive cultures; or single positive for Coxiella burnetii (Q fever)
- Evidence of endocardial involvement — positive echocardiogram (vegetation, abscess, new partial dehiscence of prosthetic valve) OR new valvular regurgitation
Minor Criteria:
- Predisposing heart condition or injection drug use
- Fever ≥38°C
- Vascular phenomena (arterial emboli, septic pulmonary infarcts, mycotic aneurysm, intracranial hemorrhage, conjunctival hemorrhages, Janeway lesions)
- Immunologic phenomena (glomerulonephritis, Osler nodes, Roth spots, positive rheumatoid factor)
- Microbiologic evidence not meeting major criteria
- Echocardiographic findings not meeting major criteria
Classification:
- Definite IE: 2 major, or 1 major + 3 minor, or 5 minor criteria
- Possible IE: 1 major + 1 minor, or 3 minor criteria
- Rejected: firm alternative diagnosis, resolution with ≤4 days antibiotics, or no pathologic evidence at surgery/autopsy
Sensitivity: 76–100%; Specificity: 88–100%; NPV ≥92%
Blood Cultures
- Obtain ≥3 sets from separate sites (aerobic + anaerobic each) before starting antibiotics
- In stable/subacute patients — defer empirical therapy until cultures obtained
- In sepsis/haemodynamic compromise — culture first, then treat immediately
Non-culture Diagnostics (Duke-ISCVID 2023 updates)
- PCR/nucleic acid testing from blood for C. burnetii, Bartonella spp., Tropheryma whipplei — major criterion
- Indirect immunofluorescence IgG ≥1:800 for Bartonella — major criterion
- Next-generation (shotgun metagenomic) sequencing — promising but currently only minor criterion for non-standard organisms
Echocardiography
| Modality | Notes |
|---|---|
| TTE (transthoracic) | First-line; highly specific; inadequate images in 20%; misses vegetations <2 mm in 20–35% of cases |
| TEE (transoesophageal) | Detects vegetations in >90% of definite IE; false-negative in 6–18%; repeat in 7–10 days if negative but IE still suspected; superior for prosthetic valves and complications |
| Cardiac CT angiography | Less sensitive than TEE for vegetations/perforations; superior for pseudoaneurysm/abscess; used pre-operatively to assess coronary arteries |
| FDG-PET/CT | Less sensitive for intracardiac pathology in NVE; increased sensitivity for PVE (including TAVR-PVE), extracardiac complications, metastatic infection |
Complications
- Heart failure — most common indication for surgery; due to valve destruction
- Perivalvular extension — abscess, fistula, pseudoaneurysm (especially aortic valve)
- Embolic events — stroke (15–20%), systemic emboli (spleen, kidney, coronary arteries)
- Mycotic aneurysms — from seeding of arterial walls
- Metastatic infection — septic arthritis, osteomyelitis, epidural abscess, splenic abscess
- Immune complex disease — glomerulonephritis, Osler nodes, Roth spots
- Conduction abnormalities — new PR prolongation suggests perivalvular abscess
Treatment
Antimicrobial Principles
- Bactericidal agents are required (not bacteriostatic)
- Parenteral therapy for full course in most cases
- Duration: typically 4–6 weeks (2 weeks for selected low-risk NVE)
- Synergistic combinations often used (e.g., β-lactam + aminoglycoside)
- A multispecialty IE team (cardiologist, cardiac surgeon, infectious disease specialist) is recommended
Empirical Therapy (pending cultures)
- Stable/subacute: withhold antibiotics until ≥3 blood culture sets collected
- Unstable: start after blood cultures — broad-spectrum cover including S. aureus
Organism-Specific Therapy (summary)
| Organism | Preferred Regimen |
|---|---|
| Viridans streptococci (penicillin-sensitive) | Penicillin G or amoxicillin ×4 weeks; ±gentamicin ×2 weeks (short course) |
| S. aureus (MSSA, NVE) | Flucloxacillin/nafcillin ×6 weeks |
| S. aureus (MRSA) | Vancomycin ×6 weeks |
| S. aureus (PVE) | Flucloxacillin + rifampicin + gentamicin |
| Enterococci | Ampicillin + gentamicin (or ampicillin + ceftriaxone for aminoglycoside-resistant strains) |
| HACEK | Ceftriaxone ×4 weeks |
| Fungal | Amphotericin B ± flucytosine; valve surgery usually necessary |
Surgical Indications
Surgery is indicated in approximately 50% of cases during active IE. Indications include:
Class I (urgent/emergent):
- Heart failure due to valve dysfunction
- Fungal or highly resistant organism endocarditis
- Perivalvular extension (abscess, fistula, large vegetation with high embolic risk)
- Persistent bacteraemia/fever despite optimal antibiotic therapy (>5–7 days)
- PVE with dehiscence
Class I (elective):
- Recurrent emboli on antibiotic therapy
- Large vegetations (>10 mm) with embolic events
Prophylaxis
Current guidelines (AHA, ESC) have significantly narrowed indications since 2007:
Prophylaxis recommended only for highest-risk cardiac conditions:
- Prosthetic valves (surgical or transcatheter)
- Previous IE
- Certain congenital heart diseases (unrepaired cyanotic CHD; repaired CHD with residual defects; repaired CHD with prosthetic material within 6 months)
- Cardiac transplant recipients with valvular disease
Dental procedures requiring prophylaxis: Those involving manipulation of gingival tissue, periapical region of teeth, or perforation of oral mucosa.
Regimens: Amoxicillin 2 g orally 30–60 minutes before procedure (clindamycin or cephalexin if penicillin-allergic — note: clindamycin no longer favoured in recent AHA guidance).
Prognosis
- In-hospital mortality: ~15–25%
- Higher mortality with: S. aureus, prosthetic valve involvement, aortic valve involvement, heart failure, perivalvular abscess, neurological complications, older age
- Injection drug user right-sided IE (S. aureus/tricuspid valve): better prognosis, mortality ~<10%
- Relapse rate after cure: ~5% for NVE, higher for PVE
Note: Davidson's Principles and Practice of Medicine is not available in the textbook library. This summary is compiled from Harrison's Principles of Internal Medicine, 22nd edition (2025) and Goldman-Cecil Medicine, which provide equivalent and current coverage of IE. The content reflects the updated Duke-ISCVID 2023 criteria and contemporary management.
— Harrison's Principles of Internal Medicine 22e, Ch. 133; Goldman-Cecil Medicine, Ch. 61
Cataract from AK Khurana
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive content from Kanski's. Let me compile the full cataract topic.
Note: AK Khurana's Comprehensive Ophthalmology is not in the library. The content below is drawn from Kanski's Clinical Ophthalmology, 10th Edition — the most authoritative and comprehensive ophthalmology textbook available — which covers all the same topics in equivalent depth.
Cataract
(Kanski's Clinical Ophthalmology, 10th Edition)
Introduction & The Lens
The primary function of the lens is to focus light on the retina.
Anatomy:
- Zonules hold the lens in position
- Capsule — acellular membrane enclosing all lens material
- Epithelial cells — cuboid, beneath the anterior capsule; cells in the germinative zone divide continuously and differentiate to form lens fibres
- Lens substance — central nucleus surrounded by cortex
Cataract (lens opacification) is the commonest cause of reversible visual disability in the world. There is no medical treatment to prevent its development or progression. Phacoemulsification with IOL implantation is the commonest surgical procedure in the world.
Symptoms (Effects on Vision)
Cataracts often develop slowly; symptoms include:
- Reduced visual acuity (may initially be correctable with spectacles)
- Loss of contrast sensitivity
- Change in colour perception — difficulty perceiving blue colours
- Glare from light scattering — particularly night driving difficulty
- Monocular diplopia — especially when looking at a point source
- Poor night vision / difficulty in low-light situations
ACQUIRED CATARACT
Age-Related (Senile) Cataract
The most common type. Three morphological subtypes:
1. Posterior Subcapsular Cataract
- Lies just in front of the posterior capsule
- Granular or plaque-like appearance on oblique slit-lamp biomicroscopy
- Appears black and vacuolated on retroillumination
- Vacuoles are swollen migratory lens epithelial cells (bladder/Wedl cells)
- Due to its location at the nodal point of the eye → profound effect on vision even when small
- Characteristically: glare (worse with car headlights), symptoms worsen with miosis (near vision, bright sunlight)
2. Nuclear Sclerotic Cataract
- Exaggeration of normal ageing change
- Associated with myopia (increased refractive index of nucleus) → "second sight of the aged" — patient can read without glasses again
- Characterized by yellowish hue due to urochrome pigment deposition — best assessed with oblique slit-lamp beam
- Advanced: nucleus appears brown (brunescent), rarely black
- Good red reflex on retroillumination; subtle nucleus-cortex distinction visible
3. Cortical Cataract
- Involves anterior, posterior, or equatorial cortex
- Opacities start as clefts and vacuoles from cortical hydration
- Progress to typical cuneiform (wedge-shaped) or radial spoke-like opacities
- Often begin in the inferonasal quadrant
- Glare is a common symptom
4. Christmas Tree Cataract
- Uncommon
- Polychromatic needle-like formations in the deep cortex and nucleus
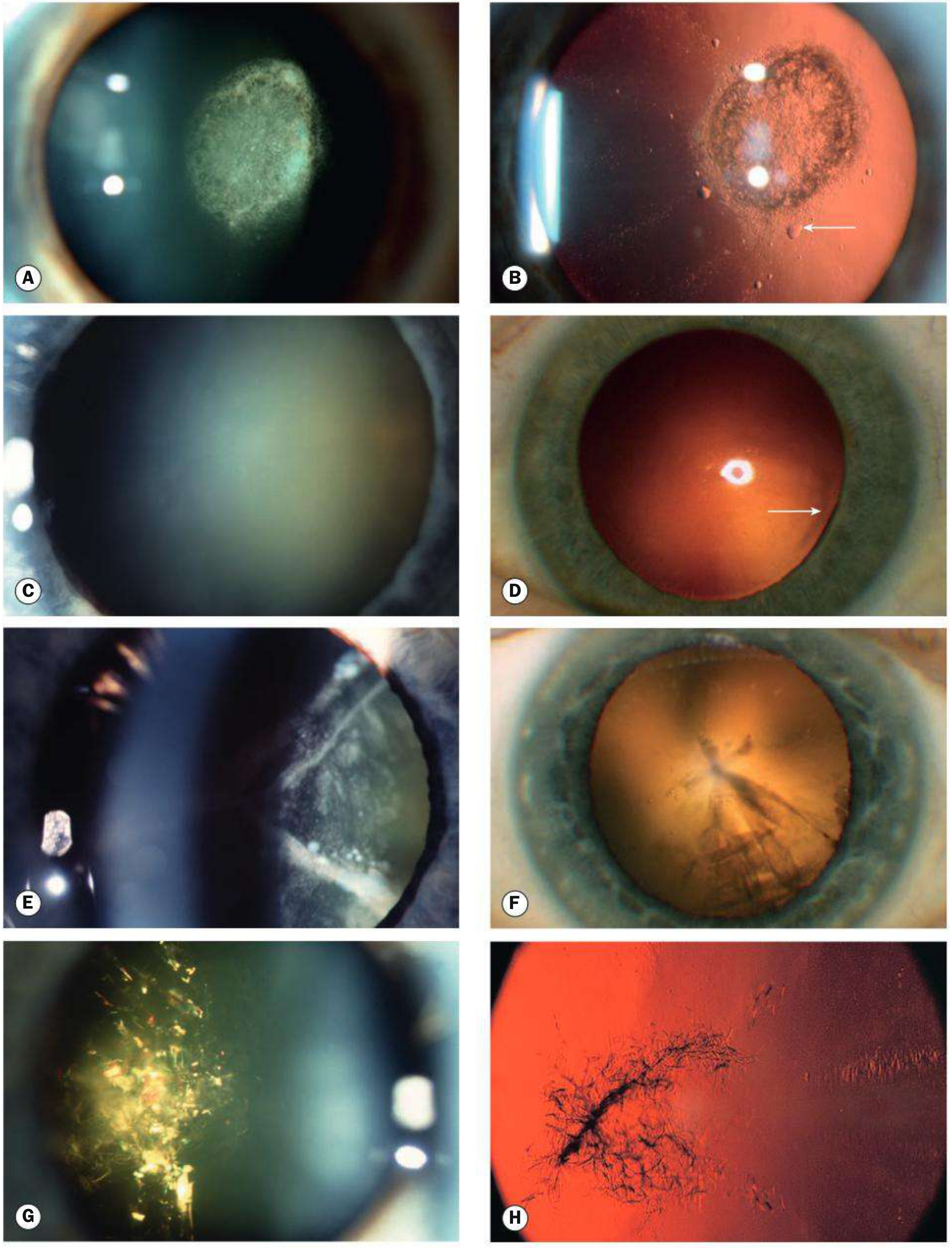
Cataract Maturity
| Stage | Features |
|---|---|
| Immature | Partially opaque lens |
| Mature | Completely opaque lens |
| Hypermature | Shrunken, wrinkled anterior capsule due to water leakage out of lens |
| Morgagnian | Hypermature cataract with liquefied cortex and nucleus sunken inferiorly |
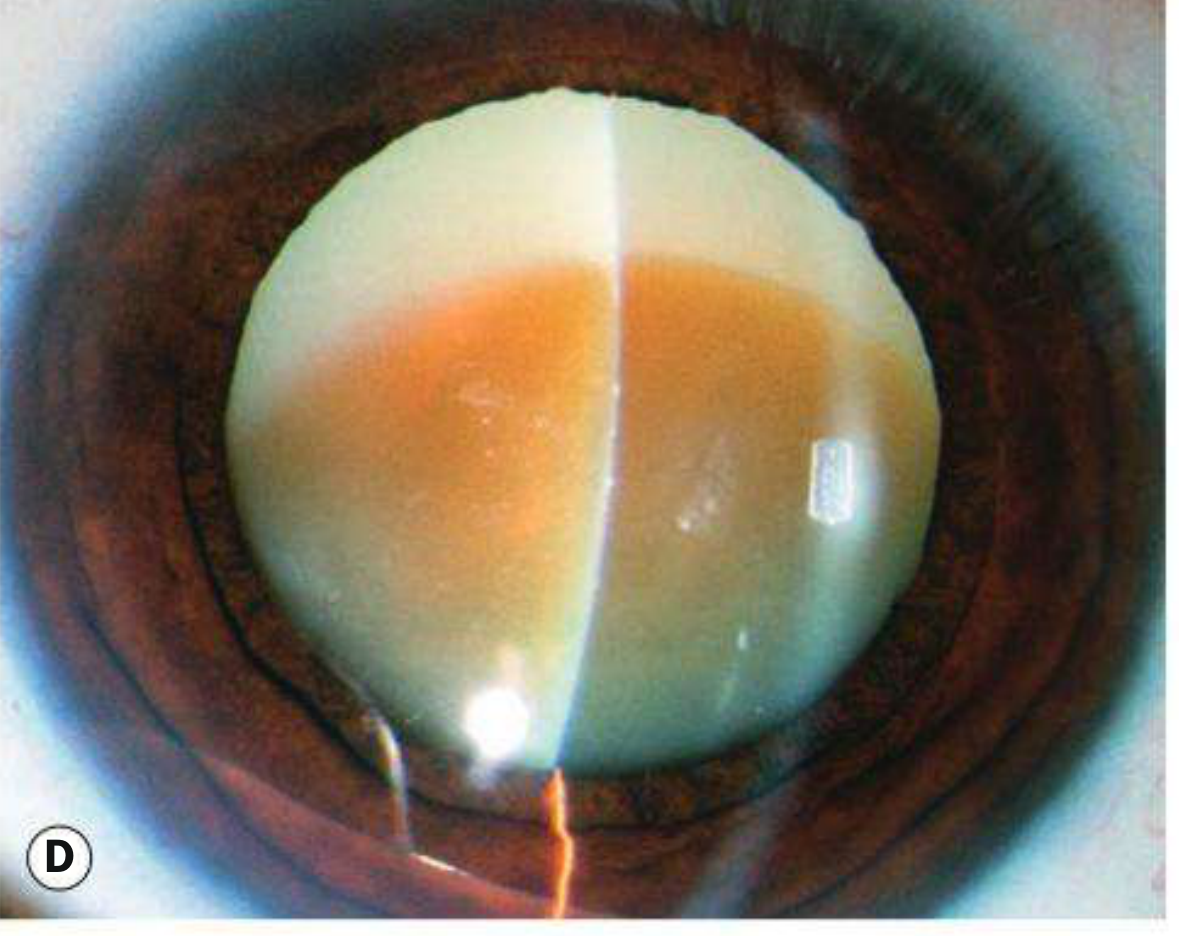
Cataract in Systemic Disease
| Disease | Cataract Features |
|---|---|
| Diabetes mellitus | Glucose → sorbitol (aldose reductase) → osmotic overhydration → cortical vacuoles; classic diabetic cataract: snowflake cortical opacities in young (rare); age-related cataract occurs earlier; nuclear sclerosis common and rapidly progressive |
| Myotonic dystrophy | Fine iridescent cortical opacities in 3rd decade; progress to star-shaped cortical/subcapsular opacities by 5th decade; ~90% of patients affected |
| Wilson's disease | Sunflower cataract — anterior capsular copper deposits in a sunflower pattern |
| Atopic dermatitis | Anterior and/or posterior subcapsular cataract; may be bilateral; progression rapid |
| Hypocalcaemia | Fine iridescent crystals and opacities; associated with hypoparathyroidism, pseudohypoparathyroidism |
| Steroids | Posterior subcapsular cataract — systemic or topical; dose- and duration-dependent |
Secondary (Complicated) Cataract
Develops as a result of another primary ocular disease.
| Cause | Features |
|---|---|
| Chronic anterior uveitis | Most common cause; polychromatic lustre at posterior pole first; then anterior + posterior opacities; steroids used in treatment also contribute |
| Acute angle closure glaucoma | Glaukomflecken — small anterior grey-white subcapsular/capsular opacities; represent focal infarcts of lens epithelium; pathognomonic of previous acute angle closure |
| High myopia | Posterior subcapsular opacity; early-onset nuclear sclerosis |
| Hereditary fundus dystrophies | Retinitis pigmentosa, Leber congenital amaurosis, gyrate atrophy, Stickler syndrome → posterior subcapsular opacity |
| Medications (steroids) | Posterior subcapsular |
| Vitreoretinal surgery | Gas bubble → feathery posterior subcapsular (usually transient); silicone oil → cataract in almost all phakic eyes; vitrectomy → nuclear sclerosis |
Traumatic Cataract
- Blunt trauma → rosette-shaped opacity (posterior subcapsular or axial)
- Penetrating trauma → lens material exposed → rapid opacification
- Electric shock → anterior subcapsular opacification
- Radiation (infrared/ionizing) → posterior subcapsular
MANAGEMENT OF ACQUIRED CATARACT
Indications for Surgery
- Functional: when cataract causes sufficient visual disability to affect the patient's activities
- No objective acuity threshold mandates surgery — patient's needs and wishes guide timing
- Surgery should not be delayed until the cataract is "ripe"
Preoperative Assessment
Ophthalmic:
- Visual acuity (Snellen chart)
- Cover test — exclude amblyopia (guarded prognosis) or risk of postoperative diplopia
- Pupillary responses — cataract never produces an RAPD; its presence implies posterior pole pathology
- Exclude dacryocystitis, blepharitis, ectropion (infection risk → endophthalmitis)
- Corneal endothelial count — cornea guttata → risk of decompensation; specular microscopy + pachymetry
- Anterior chamber depth — shallow AC → difficult surgery
- Pupil dilation — poorly dilating pupil needs planning (mydriatic drops, intracameral agents, iris hooks)
- Lens: nuclear cataracts = hard (more phaco power); black nuclei may need ECCE
- Pseudoexfoliation → weak zonules (phako-donesis), fragile capsule, poor mydriasis
- Fundus examination — AMD may limit visual outcomes; ultrasonography if fundus not visible
Biometry: IOL power calculation (keratometry + axial length measurement)
Surgical Techniques
1. Intracapsular Cataract Extraction (ICCE) — Historical
- Cryoprobe removes entire lens with capsule
- Largely abandoned; reserved for subluxated lenses
2. Extracapsular Cataract Extraction (ECCE)
- Large anterior capsulotomy → large limbal incision (8–10 mm) → nucleus expressed → cortex aspirated → IOL in capsular bag
- Requires suturing → can induce significant corneal astigmatism
- Still used for very dense/black nuclei
3. Manual Small-Incision Cataract Surgery (MSICS)
- Variant of ECCE for high-volume, resource-limited settings
- Small self-sealing sclerocorneal tunnel; no suturing required
- Comparable visual outcomes to phaco; faster and cheaper
4. Phacoemulsification (Standard in high-income countries)
- Ultrasound energy emulsifies the nucleus through a small (~2.5 mm) incision
- Phacodynamics:
- Irrigating bottle height — maintains AC pressure
- Aspiration flow rate (AFR) — volume removed per minute; high AFR = faster but riskier (surge)
- Vacuum — determines grip on lens material; high vacuum → less power needed
- Post-occlusion surge — sudden outflow when occlusion breaks → capsular rupture risk
Steps of phacoemulsification:
- Clear corneal incision (main + side-port)
- Viscoelastic injection
- Continuous curvilinear capsulorhexis (CCC) — circular anterior capsule opening
- Hydrodissection + hydrodelineation
- Nuclear disassembly (divide and conquer / stop and chop / phaco chop)
- Cortical aspiration
- IOL implantation in capsular bag
- Wound hydration/sealing
Intraocular Lenses (IOL)
| Type | Features |
|---|---|
| Monofocal | Standard; corrects distance; patient needs reading glasses |
| Multifocal | Provides near and distance vision; halos/glare a known side effect |
| Toric | Corrects pre-existing corneal astigmatism |
| Extended Depth of Focus (EDOF) | Range of functional vision with fewer halos than bifocal multifocals |
| Accommodating | Attempts to mimic accommodation |
CONGENITAL CATARACT
Epidemiology
- Occurs in ~3 in 10,000 live births
- 1/3 associated with systemic disease, 1/3 inherited, 1/3 unknown
- 2/3 are bilateral
- Autosomal dominant (AD) is the most common inherited form
- Unilateral cataracts: usually sporadic, otherwise healthy infants
Aetiology
Metabolic Disorders:
| Condition | Cataract |
|---|---|
| Galactosaemia (AR, GALT deficiency) | 'Oil droplet' opacity — within first days/weeks of life; may reverse with galactose exclusion |
| Lowe syndrome (X-linked, oculo-cerebro-renal) | Universal cataract + microphakia; ~50% have congenital glaucoma |
| Mannosidosis | Spoke-like punctate opacities in posterior lens cortex |
| Hypocalcaemia | Fine iridescent crystals |
Intrauterine Infections (TORCHES):
| Infection | Features |
|---|---|
| Rubella | Pearly nuclear or diffuse cataract in ~15%; unilateral or bilateral |
| Toxoplasmosis | Cataract + chorioretinitis + microphthalmos + optic atrophy |
| CMV | Cataract + chorioretinitis + keratitis + optic atrophy |
| Varicella | Cataract + microphthalmos + chorioretinitis |
Chromosomal: Down syndrome (trisomy 21), Turner syndrome
Morphological Types
- Blue dot (cerulean) — tiny blue-white opacities; usually visually insignificant
- Nuclear — dense central opacity
- Coronary — peripheral spoke-like opacities around nucleus
- Lamellar (zonular) — affects a specific lamella with radial 'riders'; most common visually significant type
- 'Oil droplet' — central droplet appearance; characteristic of galactosaemia
- Sutural — follows anterior or posterior Y suture; seen in Nance-Horan carriers
- Anterior polar — flat or projects into AC; associations: persistent pupillary membrane, aniridia, Peters anomaly
- Posterior polar — closely integrated with posterior capsule; very high surgical risk of dehiscence
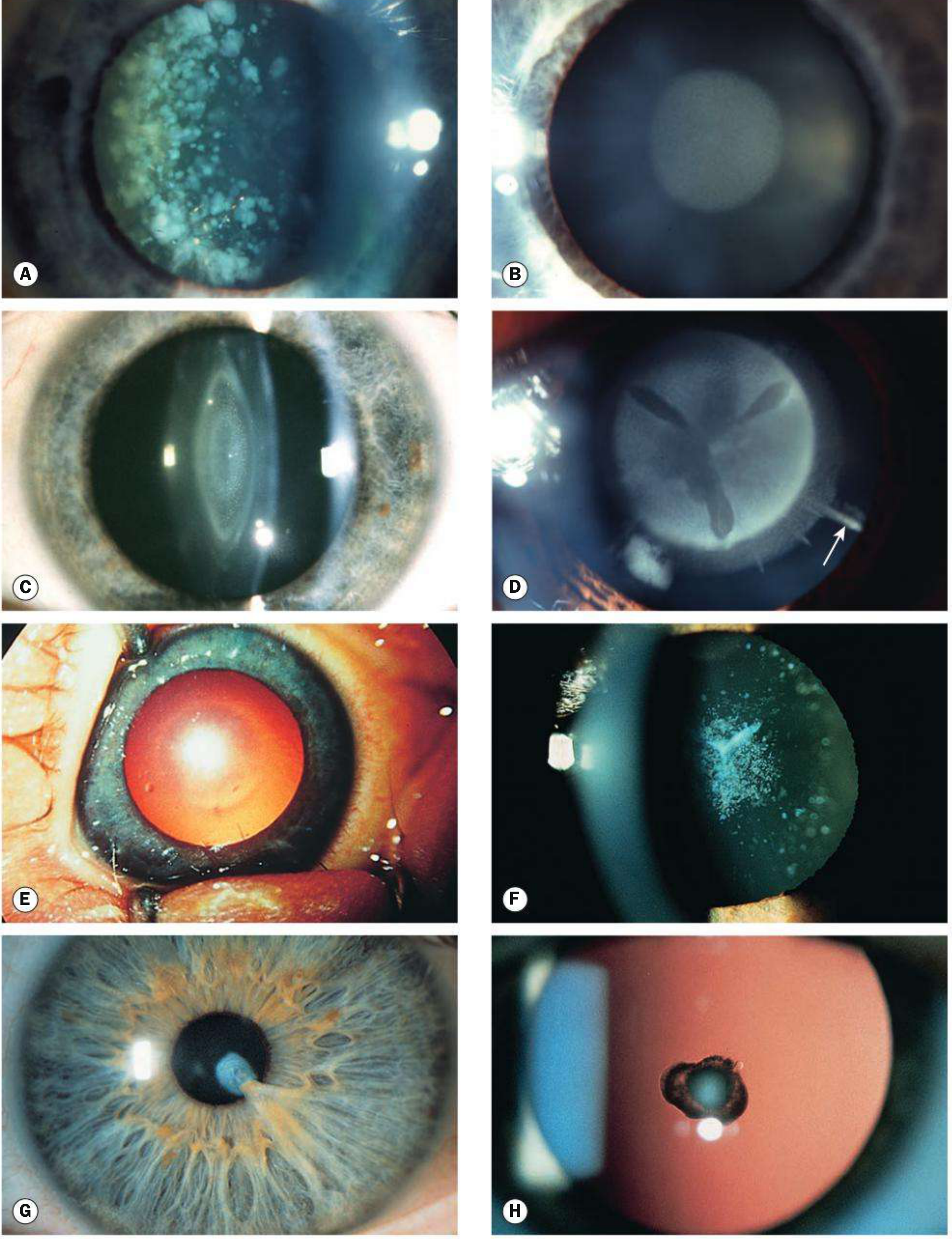
Investigations
- Screen for intrauterine infections (all unilateral + bilateral)
- Urine for reducing substance after milk (galactosaemia); amino acids (Lowe)
- Fasting blood glucose, serum calcium/phosphorus
- Red blood cell GALT and galactokinase levels
- Chromosome analysis if dysmorphic features
Treatment
- Timing is critical — must treat early to prevent stimulus deprivation amblyopia
- Surgery before 4 weeks of age → higher risk of juvenile glaucoma
- Bilateral dense cataracts: surgery between 4–10 weeks of age
- Unilateral dense cataracts: surgery within 6–8 weeks of birth; followed by aggressive occlusion therapy (patching) of fellow eye to treat amblyopia
- IOL implantation: deferred until ~2 years of age in most centres (aphakic contact lens in the interim)
- Optical correction + amblyopia treatment must continue post-operatively
— Kanski's Clinical Ophthalmology, 10th Edition, Ch. 10
Can you please tell me which all books you have for wnt ophthal medicine surgery obgyn and pediatrics
Finding Sources
You've run out of free credits. Sign up for a free account to continue using Orris.
This is a shared conversation. Sign in to Orris to start your own chat.