All time hard disease
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Here is a breakdown of the hardest diseases to treat or cure, drawn from authoritative medical textbooks:
The Hardest Diseases to Treat or Cure
1. 🧠 Prion Diseases (CJD, Fatal Familial Insomnia, Kuru)
Verdict: Uniformly fatal — no effective treatment exists
Prion diseases are caused by misfolded proteins (PrP^Sc) that propagate by converting normal brain proteins. They are unique in being transmissible yet not caused by a virus or bacterium.
"Currently, there is no known cure for human prion diseases; all cases are uniformly fatal." — Bradley and Daroff's Neurology in Clinical Practice
Drugs tested in randomized controlled trials — including quinacrine, flupirtine, and doxycycline — failed to prolong survival. Experimental approaches (e.g., anti-PrP^C antibody PRN100) are being explored but remain unproven. The disease progresses relentlessly from onset to death, typically within months to a year.
2. 🧬 Glioblastoma (Grade IV Brain Cancer)
Verdict: Near-universally fatal; median survival ~15–18 months
Glioblastoma is the most common malignant primary brain tumor, accounting for >12,000 new U.S. cases per year.
"Despite optimal therapy, glioblastomas invariably recur." — Harrison's Principles of Internal Medicine, 22E
The standard regimen (maximal resection + radiation + temozolomide chemotherapy) only extends median survival to 15–18 months, and 5-year survival is ~10%. Tumors with the MGMT enzyme are particularly resistant to chemotherapy. Even bevacizumab (anti-VEGF), while improving progression-free survival, does not improve overall survival in recurrent disease.
3. 🩸 Multiple Myeloma (Plasma Cell Cancer)
Verdict: Incurable; manageable but relapsing
"MM is considered an incurable disease, with most patients..." — Tietz Textbook of Laboratory Medicine, 7th Edition
Despite major advances in treatment (proteasome inhibitors, IMiDs, CAR-T therapy), multiple myeloma remains incurable in the vast majority of patients. Treatment achieves remission, but relapse is nearly universal.
4. 💪 Duchenne Muscular Dystrophy (DMD)
Verdict: Incurable; life-limiting progressive muscle degeneration
DMD is caused by mutations in the dystrophin gene, leading to progressive loss of muscle function and early death (typically in the 20s–30s).
"The life-threatening and debilitating nature of DMD, and the lack of available therapy has opened new hope for what has been considered an incurable and untreatable disorder." — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
Newer gene-targeted therapies (exon skipping) are emerging but remain limited in their scope and benefit.
5. 🦾 Amyotrophic Lateral Sclerosis (ALS)
Verdict: Fatal; median survival 2–5 years from diagnosis
ALS destroys motor neurons progressively, leading to paralysis, respiratory failure, and death. Prion-like spreading of misfolded SOD1 and TDP-43 proteins drives the disease.
"Prion-like spreading in ALS." — Bradley and Daroff's Neurology in Clinical Practice
Only riluzole and edaravone are approved treatments — neither stops the disease, they only modestly slow progression.
6. 🫀 Cardiac Amyloidosis (Historically)
Verdict: Historically incurable; now increasingly treatable
Once considered both rare and untreatable, cardiac amyloidosis has seen dramatic therapeutic advances (tafamidis, patisiran), making it a rare success story among previously incurable diseases.
"A disease that formerly was conceived as uncommon and untreatable is now increasingly recognized..." — Braunwald's Heart Disease
7. 🦠 Rabies
Verdict: Essentially 100% fatal once symptomatic
Once clinical symptoms appear, rabies is almost universally fatal. Pre- and post-exposure prophylaxis (vaccine) is effective, but there is no established treatment after neurological symptoms begin. The "Milwaukee Protocol" has had rare anecdotal successes but is not reproducibly effective.
Why Are These Diseases So Hard to Treat?
| Reason | Examples |
|---|---|
| No druggable target identified | Prion diseases |
| Brain/CNS drug penetration barriers | Glioblastoma, ALS |
| Tumor microenvironment suppresses immunity | Glioblastoma |
| Genetic complexity / multiple mutations | Multiple myeloma |
| Disease too advanced at diagnosis | Many cancers |
| Pathogen hides from immune system | HIV (chronic), Rabies |
| Protein misfolding cascade irreversible | Prions, ALS |
The diseases above represent the frontier of medicine — areas where current science, despite enormous effort, has not yet found a cure.
Stomach ulcer differential diagnosis treatment and investigation
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive clinical overview of Peptic Ulcer Disease (Gastric/Stomach Ulcer) — covering differential diagnosis, investigations, and treatment — drawn from major gastroenterology and clinical medicine textbooks.
Peptic Ulcer Disease (Stomach Ulcer)
Definition
A peptic ulcer occurs when inflammation causes loss of the mucosa and muscularis mucosa, often extending into the submucosa and muscularis propria. Gastric ulcers are located in the stomach; duodenal ulcers in the first part of the duodenum. Together, they constitute 30–50% of all upper GI bleeding admissions.
Aetiology & Risk Factors
| Cause | Detail |
|---|---|
| H. pylori infection | Most common cause — ~90% of duodenal ulcers worldwide; 30–40% in the US |
| NSAIDs / Aspirin | Second most common; inhibit prostaglandins → disrupts mucosal protection |
| Smoking | Delays healing, increases recurrence |
| Zollinger–Ellison syndrome | Gastrin-secreting tumour → massive acid hypersecretion |
| Corticosteroids | Especially combined with NSAIDs |
| Crack cocaine / methamphetamine | Illicit drug use |
| Immunocompromised state | CMV, HSV, fungal infections can cause atypical ulcers |
| Systemic mastocytosis | Rare |
| Blood group O | Genetically linked to higher duodenal ulcer risk |
"Cumulative lifetime prevalence of PUD is 8–14%" — Textbook of Family Medicine 9e
Clinical Presentation
- Epigastric pain — burning or gnawing; often worsens after meals (gastric ulcer) or relieved by food (duodenal ulcer)
- Nausea, bloating, early satiety
- Alarm symptoms (red flags): rectal bleeding/melena, unintentional weight loss, anorexia, persistent vomiting, dysphagia, anaemia, palpable mass
- Complications: upper GI haemorrhage (up to 15%), perforation (~7%), gastric outlet obstruction, penetration into adjacent organs
Differential Diagnosis
Common
| Condition | Distinguishing Features |
|---|---|
| Gastro-oesophageal reflux disease (GERD) | Heartburn, regurgitation; worse lying down/after meals |
| Functional (non-ulcer) dyspepsia | No structural lesion on endoscopy; diagnosis of exclusion |
| Peptic ulcer disease | Epigastric pain; H. pylori or NSAID history |
Less Common
| Condition | Distinguishing Features |
|---|---|
| GI malignancy (gastric cancer) | Alarm symptoms, older age, progressive weight loss |
| Pancreatitis | Epigastric pain radiating to back, elevated lipase/amylase |
| Biliary colic | Right upper quadrant, post-fatty meal colicky pain |
| Gastroparesis | Early satiety, nausea, bloating — often in diabetics |
| Zollinger–Ellison syndrome | Multiple refractory ulcers, severe GERD, diarrhoea |
| Intestinal ischaemia | Postprandial pain, weight loss, vascular risk factors |
| Celiac sprue | Diarrhoea, malabsorption, bloating |
| Lactose intolerance | Bloating, diarrhoea after dairy |
| Viral/bacterial gastroenteritis | Acute onset, fever, diarrhoea |
Investigations
Step 1 — Who Needs Endoscopy?
- Age >45 years with new-onset dyspepsia → upper GI endoscopy (AGA guideline)
- Any age with alarm symptoms → urgent endoscopy
- Younger patients without alarm symptoms → "test and treat" H. pylori strategy first
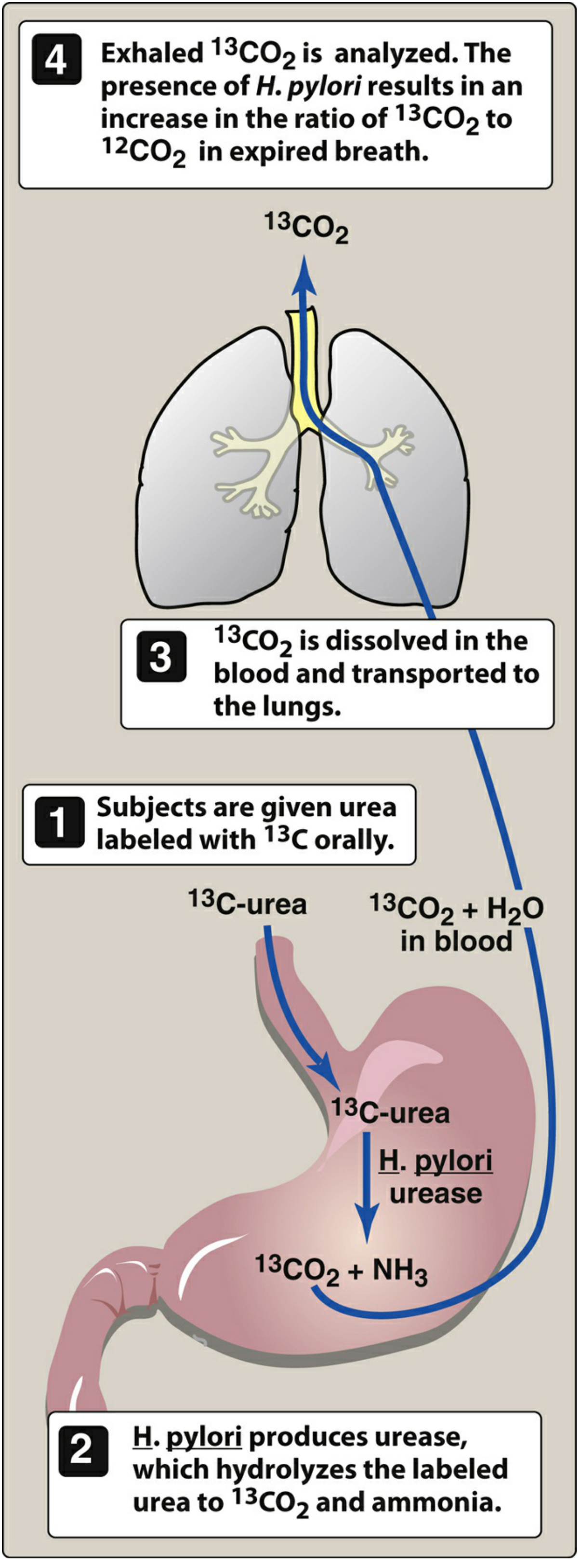
Non-invasive H. pylori Testing
| Test | Details |
|---|---|
| Stool antigen test | Preferred initial non-invasive test |
| Urea breath test (¹³C) | Most accurate non-invasive test; gold standard to confirm eradication |
| Serum IgG serology | Sensitivity 92%, specificity 83%; may remain positive months–years after eradication (false positives) |
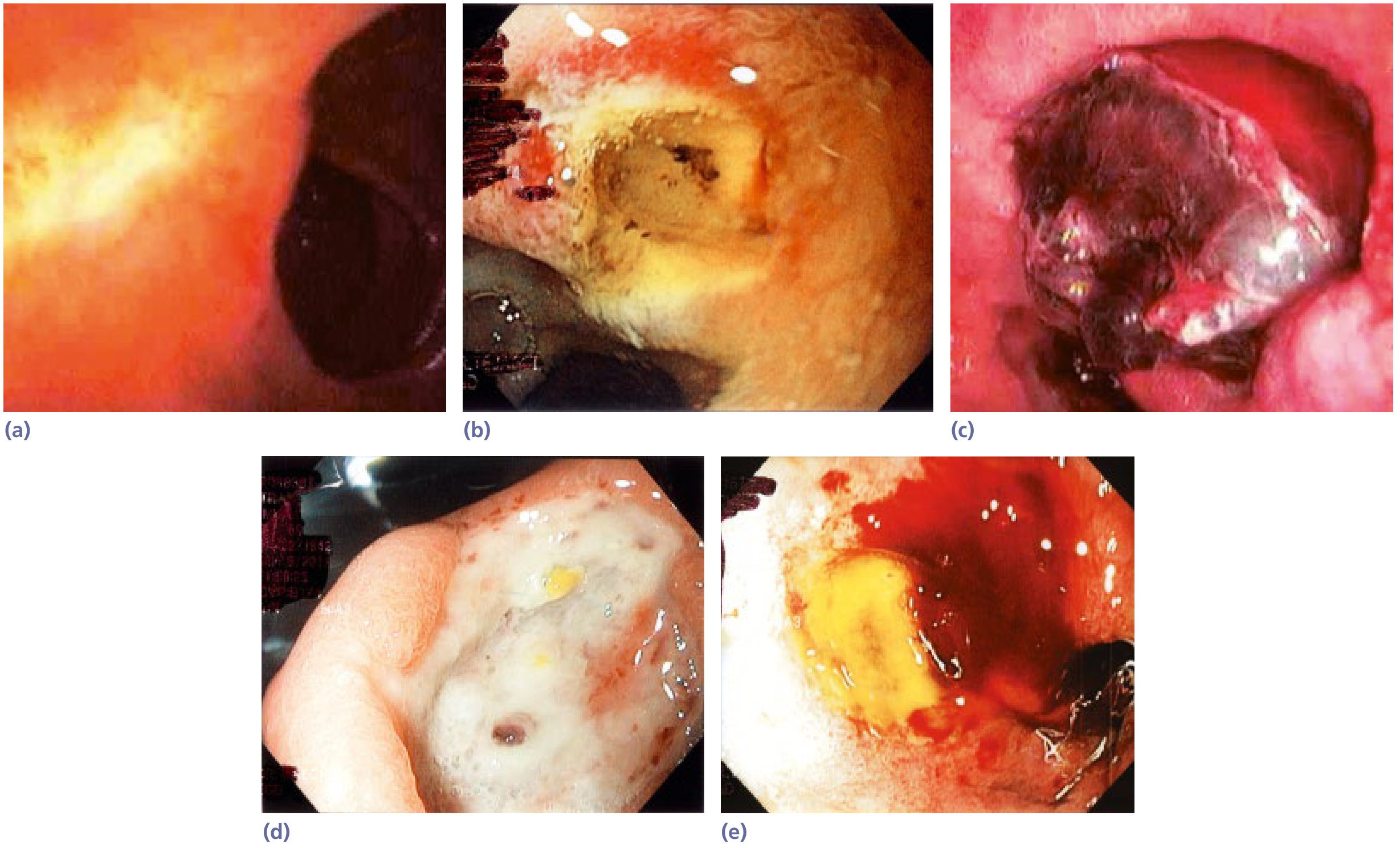
Invasive — Upper GI Endoscopy
- Gold standard for diagnosis; allows direct visualisation, biopsy, and therapeutic intervention
- Document: ulcer location, size, number, edge characteristics, presence of mass or nodules
- Biopsy of gastric ulcers is mandatory (to exclude malignancy) — biopsies also detect H. pylori on gastric mucosa
- Forrest classification of stigmata of recent haemorrhage (used in Europe/Asia):
- High risk: active spurting/oozing, non-bleeding visible vessel
- Intermediate: adherent clot
- Low risk: flat pigmented spot, clean base
Other Investigations
| Test | Purpose |
|---|---|
| FBC | Anaemia from chronic/acute blood loss |
| Urea & Creatinine | Urea often elevated in upper GI bleed (blood digestion) |
| Coagulation screen | If bleeding suspected |
| Serum gastrin | If Zollinger–Ellison suspected (multiple/refractory ulcers) |
| Erect CXR / AXR | Subdiaphragmatic free air if perforation suspected |
| Rockall Score | Risk stratification for rebleeding and mortality in GI haemorrhage |
Treatment
1. H. pylori Eradication (First-line for H. pylori-positive PUD)
14-day Triple Therapy (preferred when clarithromycin resistance is low):
PPI + Clarithromycin + Amoxicillin (or Metronidazole if penicillin-allergic)
- Eradication rate: 70–85%
Bismuth Quadruple Therapy (first-line OR salvage if triple therapy fails):
PPI + Bismuth subsalicylate + Metronidazole + Tetracycline
- Eradication rate: ≥90%
Successful eradication eliminates ulcer recurrence — recurrence drops from 60–100%/year (with acid suppression alone) to <15% after eradication.
Confirm eradication with urea breath test or stool antigen ≥4 weeks after completing therapy.
2. Acid Suppression
| Drug Class | Examples | Mechanism |
|---|---|---|
| Proton Pump Inhibitors (PPIs) | Omeprazole, Pantoprazole, Lansoprazole | Inhibit H⁺/K⁺-ATPase proton pump; most potent acid suppressors |
| H2 Receptor Antagonists | Famotidine, Nizatidine, Cimetidine | Block histamine H2 receptors → reduce acid secretion |
| Antacids | Aluminium hydroxide, Calcium carbonate, Magnesium hydroxide | Neutralise gastric acid; short-term symptom relief |
- After haemostasis in bleeding ulcers: IV PPI for 72 hours, then daily PPI for ≥8 weeks
3. Mucosal Protective Agents
| Agent | Role |
|---|---|
| Sucralfate | Forms protective coat over ulcer base |
| Bismuth subsalicylate | Mucosal protection + antibacterial (part of quadruple therapy) |
4. NSAID-related Ulcers
- Stop NSAID if possible
- Prescribe a PPI for ulcer healing
- If NSAID must continue (e.g. for cardiovascular reasons): use a COX-2 inhibitor + PPI
- High-risk patients (elderly, prior PUD, on anticoagulants/corticosteroids): prophylactic PPI or H2RA
5. Test-and-Treat Strategy (Primary Care)
For patients <45 years with dyspepsia, no alarm symptoms:
- Test for H. pylori (stool antigen preferred)
- Treat if positive; proceed to endoscopy if symptoms persist despite treatment
6. Surgical / Endoscopic Management (Complications)
Bleeding ulcer (endoscopic haemostasis):
- Injection of adrenaline (epinephrine) + thermal coagulation or mechanical clips
- Avoid epinephrine alone — combination therapy reduces rebleeding
- Surgery or interventional radiology if endoscopic haemostasis fails
Perforation:
- Barium/endoscopy contraindicated — confirmed by erect CXR (free air under diaphragm)
- Urgent surgical consultation — laparoscopic or open repair
- Mortality 30–50% in elderly/debilitated patients
Gastric outlet obstruction:
- Endoscopic balloon dilatation or surgical intervention
Summary Algorithm
Dyspepsia
├── Alarm symptoms or age >45?
│ └── YES → Urgent Upper GI Endoscopy + Biopsy
│
└── NO (young, uncomplicated)
└── Test for H. pylori (stool antigen)
├── POSITIVE → 14-day Triple Therapy + PPI
│ → Confirm eradication (UBT at 4 weeks)
└── NEGATIVE → Empirical PPI trial (4–8 weeks)
→ Endoscopy if symptoms persist
Key Sources: Yamada's Textbook of Gastroenterology (7th ed.) | Textbook of Family Medicine 9e | Sleisenger and Fordtran's GI and Liver Disease | Lippincott Illustrated Reviews: Pharmacology
Weight loss
Asking for Preferences
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Here is a comprehensive clinical overview of Unintentional (Involuntary) Weight Loss sourced from Harrison's Principles of Internal Medicine, Goldman-Cecil Medicine, and Yamada's Textbook of Gastroenterology.
Unintentional Weight Loss
Definition
Clinically significant unintentional weight loss (UWL) = loss of >5% of usual body weight over 6–12 months.
| Threshold | Clinical Significance |
|---|---|
| >5% in 6–12 months | Clinically important; warrants investigation |
| >10% of body weight | Associated with protein-energy malnutrition and impaired physiological function |
| >20% of body weight | Nutritional deficiencies and multi-organ dysfunction |
"In a representative US sample, 5% of adults aged 45–54 reported involuntary weight loss of ≥5% of body weight. Prevalence rises to 15–20% in those over 65 years." — Yamada's Textbook of Gastroenterology
UWL is associated with a 22–39% excess mortality rate, primarily reflecting the severity of the underlying pathology.
Pathophysiology
Three fundamental mechanisms drive weight loss:
- Reduced appetite/food intake — anorexia, pain, psychiatric illness, medications
- Impaired nutrient absorption — malabsorption syndromes, pancreatic insufficiency
- Increased catabolism — cytokine-driven (cancer, chronic infections, inflammatory disease)
The Cachexia Pathway (Cancer & Chronic Disease)
Proinflammatory cytokines — IL-1, IL-2, IL-6, interferon-γ, TNF-α — are the key mediators:
- Activate NF-κB → ↓ muscle protein synthesis
- Activate ubiquitin-mediated proteolysis → muscle breakdown
- Stimulate lipolysis → loss of adipose tissue
- Suppress appetite via hypothalamic pathways
"Cytokines also activate the ubiquitin-mediated proteolytic system, which is the principal mechanism for protein catabolism in disease-related catabolic states, including cancer." — Yamada's Textbook of Gastroenterology
Cancer cachexia is distinct from non-cancer weight loss: it involves loss of both adipose tissue AND skeletal muscle, whereas simple starvation primarily depletes fat. It affects up to 80% of GI and pancreatic cancer patients.
Causes of Unintentional Weight Loss
1. Malignancy (~16–40% of cases)
The single most important cause to exclude. ~50% of weight loss-associated malignancies are gastrointestinal.
| Cancer Type | Notes |
|---|---|
| Gastric cancer | Often asymptomatic early; no localising symptoms |
| Pancreatic cancer | Classical cause; may present with weight loss before pain |
| Colon cancer | Change in bowel habit, occult blood |
| Hepatocellular carcinoma | Especially in cirrhotic patients |
| Lung cancer | Particularly in smokers |
| Haematological | Lymphoma, leukaemia — fever, night sweats |
| Breast, ovarian, genitourinary, prostate |
"Malignancy that reveals itself through significant weight loss usually has a very poor prognosis." — Harrison's 22E "Up to 20% of all cancer deaths are caused directly by cachexia."
2. Gastrointestinal Disorders
- Peptic ulcer disease
- Inflammatory bowel disease (especially Crohn's disease)
- Malabsorption (coeliac disease, pancreatic insufficiency, bacterial overgrowth)
- Dysmotility / gastroparesis
- Chronic pancreatitis
- Atrophic gastritis
- Cirrhosis
- Chronic intestinal ischaemia (postprandial pain, "food fear")
- Oral/dental problems (caries, abscess, poor dentition, xerostomia)
3. Metabolic & Endocrine Disorders
| Condition | Mechanism |
|---|---|
| Hyperthyroidism | ↑ metabolic rate; weight loss despite increased appetite |
| Diabetes mellitus (uncontrolled) | Glycosuria, osmotic diuresis, relative insulin deficiency |
| Addison's disease | Cortisol deficiency, anorexia, nausea |
| Phaeochromocytoma | Catecholamine excess, ↑ catabolism |
4. Chronic Infections
| Infection | Notes |
|---|---|
| Tuberculosis | Classic constitutional weight loss, night sweats, cough |
| HIV | ~25% of undiagnosed HIV presents with weight loss; 40% co-infection with TB in some regions |
| Subacute bacterial endocarditis | Fever, embolic phenomena |
| Chronic abscesses / parasitic infections |
5. Psychiatric Disorders
| Condition | Features |
|---|---|
| Depression | Anorexia, loss of interest in food; commonest psychiatric cause |
| Anorexia nervosa | Especially young women; body image distortion |
| Dementia | Forgetting to eat; cognitive decline |
| Anxiety disorders | |
| Alcohol/substance use | Nutritional neglect |
6. Medications (Drug-induced Anorexia/Weight Loss)
| Drug | Mechanism |
|---|---|
| SSRIs | Serotonergic anorexia |
| Metformin | GI intolerance, nausea |
| NSAIDs | GI mucosal damage, nausea |
| Levodopa | Nausea, dysgeusia |
| ACE inhibitors | Dysgeusia |
| Sedatives | Reduced interest in food |
| Chemotherapy | Nausea, mucositis |
| Digoxin, antibiotics | GI side effects |
7. Chronic Organ Disease
- Chronic kidney disease — uraemic anorexia, dysgeusia
- Chronic heart failure — cardiac cachexia; cytokine-mediated
- COPD — increased work of breathing, systemic inflammation
- Rheumatoid arthritis / vasculitis (e.g. polyarteritis nodosa)
8. Physiological / Social Causes (Especially Elderly)
- Anorexia of ageing (↓ smell/taste, slowed gastric emptying, early satiety)
- Social isolation, poverty, immobility
- Visual impairment
- Functional decline, frailty
Investigations
First-line (All Patients)
| Test | Rationale |
|---|---|
| Full blood count (FBC) | Anaemia (chronic disease, GI blood loss, B12/folate deficiency) |
| ESR / CRP | Inflammation, infection, malignancy |
| Urea, electrolytes, creatinine | Renal disease, dehydration |
| Liver function tests | Hepatic malignancy, cirrhosis |
| Thyroid-stimulating hormone (TSH) | Hyperthyroidism |
| Fasting glucose | Diabetes |
| Urinalysis | Glycosuria, proteinuria |
| Chest X-ray | Lung cancer (mandatory in smokers >40 years, or with respiratory symptoms) |
| Faecal occult blood test (FOBT) | GI malignancy screen |
| HIV serology | Low threshold if unexplained; especially with TB risk factors |
"The cause of weight loss is usually evident from the history, physical examination, and routine laboratory studies." — Goldman-Cecil Medicine
Second-line (Directed by First-line Results)
| Finding | Next Step |
|---|---|
| ↑ appetite + weight loss | TSH, fasting glucose, malabsorption screen |
| GI symptoms | Upper GI endoscopy + biopsy, colonoscopy, CT abdomen |
| Malabsorption suspected | Faecal fat (elastase), anti-tissue transglutaminase (coeliac), small bowel biopsy |
| Lymphadenopathy / haematological abnormality | CT chest/abdomen/pelvis, bone marrow biopsy |
| Respiratory symptoms / smoking | CT chest, bronchoscopy |
| Suspicion of infection | TB (Interferon-γ release test / IGRA), HIV, blood cultures |
| Psychiatric features | Formal psychiatric evaluation |
| Tumour markers (when relevant) | CEA, CA 19-9 (pancreatic/GI), AFP (liver), PSA (prostate) |
Advanced Imaging
| Modality | Role |
|---|---|
| CT abdomen/pelvis | First-line cross-sectional imaging; detects most intra-abdominal causes |
| PET/CT (FDG) | Highest sensitivity for occult neoplasia, infection, vasculitis; appropriate when simpler tests are negative |
| MR angiography | Mesenteric ischaemia, vasculitis (e.g. polyarteritis nodosa) |
| Isotope bone scan | Bony metastases (being superseded by PET/CT) |
| Mammography | Women >50 years with unexplained weight loss |
"PET/CT probably has the greatest sensitivity for detection of disorders likely to be relevant to weight loss (neoplasia, infection, vasculitis)." — Yamada's Textbook of Gastroenterology
Management Principles
The cornerstone is treating the underlying cause. In addition:
| Strategy | Detail |
|---|---|
| Treat underlying disease | Cancer, infection, endocrine disorder, depression |
| Stop/switch offending medications | Review all drugs for anorectic side effects |
| Oral nutritional supplements | High-energy drinks between (not with) meals to minimise appetite suppression |
| Nutritional support | Enteral/parenteral nutrition in severe malnutrition |
| Mirtazapine | Antidepressant that significantly increases body weight, body fat mass, and leptin; use in selected patients (side effects: dizziness, fatigue, somnolence ~10%) |
| Exercise programme | Gains muscle protein mass, strength and endurance in wasting conditions |
| Social support | Address poverty, isolation, mobility limitations in elderly |
"Orexigenic, anabolic, and anticytokine agents are not generally recommended [for unexplained UWL]." — Harrison's Principles of Internal Medicine, 22E
Key Red Flag "Alarm" Features Requiring Urgent Investigation
- Age >45 years with new-onset weight loss
- Smoking history (especially men)
- Dysphagia or odynophagia
- Haematemesis or melaena
- Persistent vomiting
- Palpable abdominal mass or lymphadenopathy
- Abnormal blood tests (anaemia, elevated LFTs, elevated inflammatory markers)
- Weight loss >10% body weight
Important note: In up to 25% of patients, no cause of weight loss is found even after thorough investigation. These patients still carry an increased risk of cancer diagnosis within 2 years and warrant follow-up.
Sources: Harrison's Principles of Internal Medicine 22E | Goldman-Cecil Medicine (International Edition) | Yamada's Textbook of Gastroenterology (7th ed.)
This is a shared conversation. Sign in to Orris to start your own chat.