cardiac biomarkers used in IHD along with preanalytical variables
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have sufficient information from multiple authoritative sources to provide a comprehensive answer. Let me synthesize.
Cardiac Biomarkers in Ischemic Heart Disease (IHD)
1. Overview
Cardiac biomarkers are proteins released from necrotic or injured cardiomyocytes into the peripheral circulation. For a diagnosis of acute myocardial infarction (AMI), the criteria require a rise and/or fall in cardiac biomarker values with at least one value above the 99th percentile of the upper reference limit for a normal reference population. — Harrison's Principles of Internal Medicine 22E
2. Cardiac Biomarkers Used in IHD
A. Cardiac Troponins (cTnI and cTnT) — Gold Standard
- cTnT and cTnI have amino-acid sequences distinct from skeletal muscle isoforms, enabling highly specific monoclonal antibody-based immunoassays.
- Detected using high-sensitivity (hs-cTn) assays, which are now the preferred biochemical markers for MI.
- cTnI/cTnT typically remain elevated for 7–10 days after STEMI.
- Rise and fall pattern distinguishes acute myocardial injury from chronic elevation (e.g., renal disease, structural heart disease).
- Serial measurements at presentation and 1–3 h (hs assays) or 3–6 h (conventional assays) are standard.
- Rapid rule-out protocols using hs-cTn at 0 and 1–2 h have negative predictive value >99% when a very low concentration is seen on first sampling (>2–3 h after symptom onset). — Harrison's 22E
| Feature | cTnI | cTnT |
|---|---|---|
| Cardiac specificity | Very high | Very high |
| Skeletal muscle cross-reactivity | None | Minimal |
| Elevation duration post-MI | 7–10 days | 7–14 days |
| Assay type preferred | hs-cTnI | hs-cTnT |
B. Creatine Kinase-MB (CK-MB)
- CK-MB (MB isoenzyme of creatine kinase) was historically the standard biomarker for AMI.
- A relative index (CK-MB mass / total CK activity) ≥2.5 suggests, but is not diagnostic of, a myocardial source.
- More rapid decline after AMI onset compared to troponin — making it useful for detecting early reinfarction during the period troponin remains elevated.
- Not cost-effective to measure both a cardiac-specific troponin and CK-MB simultaneously.
- Poor specificity in trauma patients: elevated in skeletal muscle, liver, diaphragm, or intestinal injury. — Harrison's 22E; Tintinalli's Emergency Medicine
C. Myoglobin
- Early-rising biomarker (appears within 1–2 h of onset).
- Lacks cardiac specificity — also released from skeletal muscle injury.
- Largely replaced by hs-cTn for early rule-out; rarely used alone.
D. B-type Natriuretic Peptide (BNP) / NT-proBNP
- Released primarily from ventricular myocytes in response to increased wall stress/volume overload.
- Not a primary marker for MI diagnosis, but used for risk stratification and heart failure diagnosis in the context of IHD-related LV dysfunction.
- NT-proBNP has a longer half-life; BNP has a half-life of ~13–20 minutes.
- Useful when assessed alongside clinical history and examination for diagnosing HF in IHD patients. — Tietz Textbook of Laboratory Medicine 7E; Harrison's 22E
E. High-Sensitivity Troponin Assay — Key Advances
- Allows detection of myocardial injury in a larger proportion of patients with non-ACS cardiopulmonary conditions.
- Greater negative predictive value than conventional assays.
- Rise/fall delta criterion is crucial to distinguish acute from chronic troponin elevation. — Harrison's 22E
3. Biomarker Release Kinetics in AMI
- Release timing depends on: intracellular location, molecular weight, and local blood/lymphatic flow.
- Biomarkers become detectable once cardiac lymphatic clearance capacity is exceeded and spillover into venous circulation occurs.
- Reperfusion (thrombolysis or PCI) causes earlier peaking due to rapid washout — "washout peak." — Harrison's 22E
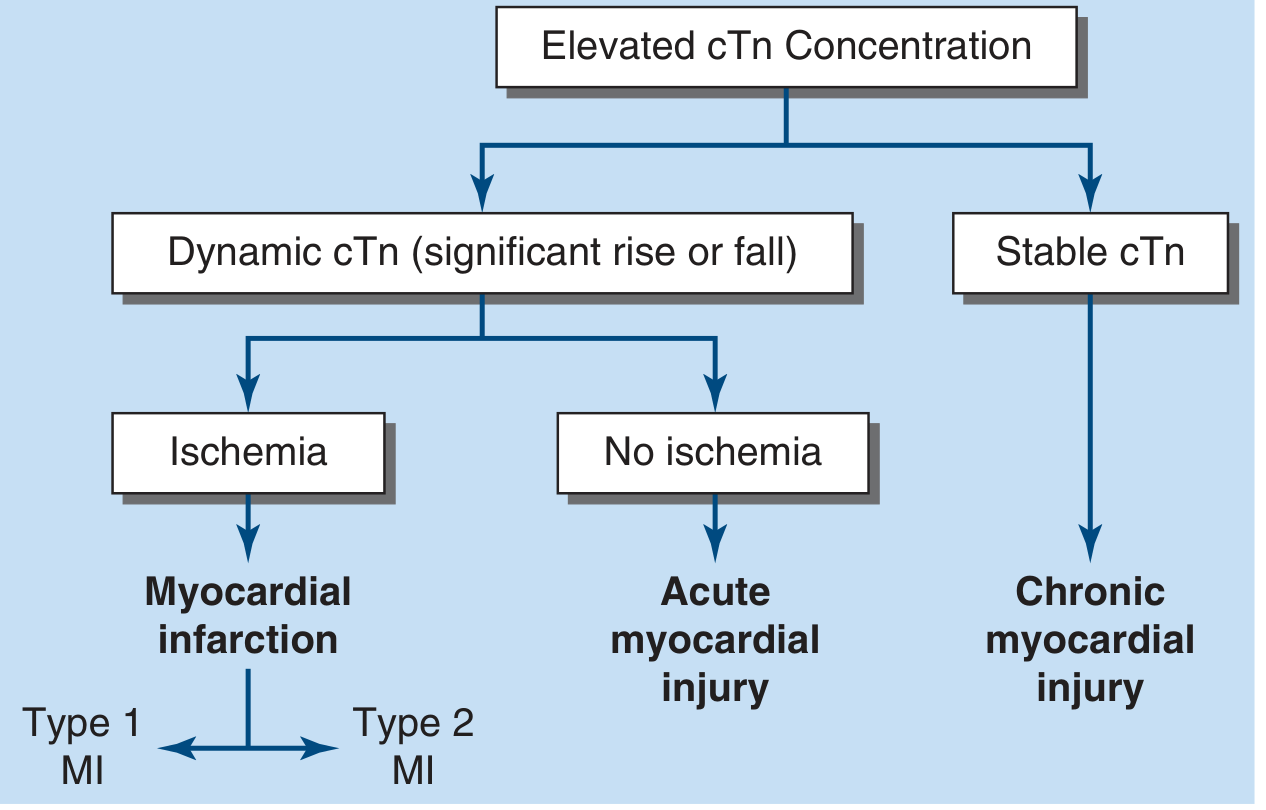
4. Preanalytical Variables Affecting Cardiac Biomarkers
Preanalytical variables are among the most critical sources of error in cardiac biomarker testing. Major preanalytical considerations include:
A. Sample Type and Anticoagulants
- EDTA and heparin are both known to interfere with cTnI and cTnT antibody-binding affinity and produce matrix effect differences between whole blood, plasma, and serum specimens.
- For emergency processing, anticoagulated whole blood or plasma is the optimal specimen — it eliminates extra clotting time and reduces sample handling.
- Different sample types (e.g., serum vs. plasma) should not be mixed during serial, timed sampling for MI rule-in/rule-out. — Tietz Textbook of Laboratory Medicine 7E
B. Specimen Integrity
- Hemolysis: Can falsely elevate or interfere with immunoassay readings; must be documented.
- Lipemia: Can cause light-scattering interference in photometric-based assays.
- Icterus: Bilirubin can interfere with colorimetric methods.
C. Troponin Degradation / Proteolysis
- cTnI undergoes extensive in vitro proteolysis post-sample collection (Western blot analyses confirm progressive fragmentation over hours at 37°C).
- Antibody epitope selection critically determines how much circulating degraded cTn is detected — different antibody pairs on different assays detect varying amounts of fragmented cTn. — Tietz Textbook of Laboratory Medicine 7E
D. cTn Molecular Forms in Circulation
- Troponin circulates in multiple molecular forms: free cTnI, free cTnT, binary complexes (cTnI-cTnC), ternary complexes (cTnI-cTnT-cTnC), and various phosphorylated/oxidized forms.
- Post-MI, the proportion of the ternary ITC complex decreases over time.
- Different assay antibodies may not detect all circulating forms equally — a source of inter-assay discordance.
E. Assay Standardization Issues
- No primary reference material currently exists for cTnI standardization.
- Results are not interchangeable between assays from different (or even the same) manufacturers.
- cTnT has a single manufacturer (Roche), improving comparability; cTnI assays vary widely.
- NIST developed SRM 2921 (cTnI reference material) but it is of limited practical use for harmonization. — Tietz Textbook of Laboratory Medicine 7E
F. Turnaround Time (TAT)
- Guidelines recommend TAT <60 minutes from order to report.
- Studies (CAP Q-Probe, 159 hospitals) showed median TAT of 74.5 min for troponin, with <25% of hospitals meeting the 60-min target.
- Point-of-care (POC) testing can reduce TAT to <30 minutes, particularly valuable in EDs without rapid central laboratory access. — Tietz Textbook of Laboratory Medicine 7E
G. BNP/NT-proBNP Specific Preanalytical Issues (IFCC C-CB Recommendations 2019)
- Analytical and preanalytical quality specifications for natriuretic peptide (NP) assays were formalized by the IFCC Committee on Clinical Applications of Cardiac Bio-Markers (C-CB).
- Glycosylation of proBNP at Thr-71 affects processing: in acute HF, less glycosylation allows more efficient conversion to NT-proBNP and bioactive BNP; in chronic HF, more glycosylation impairs this, resulting in less active BNP.
- A growing diversity of BNP/NT-proBNP assays exist worldwide — results are not interchangeable between platforms. — Tietz Textbook of Laboratory Medicine 7E
5. Summary Table: Biomarkers in IHD
| Biomarker | Rise (h) | Peak (h) | Return to Baseline | Cardiac Specificity | Primary Use |
|---|---|---|---|---|---|
| hs-cTnI/cTnT | 1–3 | 12–24 | 7–14 days | Highest | AMI diagnosis, rule-in/rule-out |
| CK-MB | 3–6 | 12–24 | 2–3 days | Moderate | Reinfarction detection |
| Myoglobin | 1–2 | 6–9 | 24 h | Low | Early marker (largely obsolete) |
| BNP/NT-proBNP | Variable | Variable | Variable | Moderate | HF diagnosis, risk stratification |
Key sources: Harrison's Principles of Internal Medicine 22E, Tietz Textbook of Laboratory Medicine 7th Edition, Tintinalli's Emergency Medicine
This is a shared conversation. Sign in to Orris to start your own chat.