Gastric pathology
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
I now have excellent, comprehensive content from multiple authoritative Robbins textbooks. I have enough information to write a thorough overview of gastric pathology. Let me now compose the response.
Gastric Pathology
A comprehensive overview of the major disease processes affecting the stomach, drawn from Robbins & Kumar Basic Pathology and Robbins, Cotran & Kumar Pathologic Basis of Disease.
1. Gastritis
Acute Gastritis
Acute gastritis is a transient mucosal inflammatory process, most often associated with:
- Heavy NSAID or aspirin use
- Excessive alcohol intake
- Heavy smoking
- Uraemia, systemic infections (e.g., Staphylococcal food poisoning), severe physiological stress (burns → Curling ulcer; CNS injury → Cushing ulcer)
Mucosal injury results from imbalance between aggressive factors (acid, pepsin) and protective factors (mucus, bicarbonate, prostaglandins, blood flow). Histologically, the mucosa shows oedema, vascular congestion, and neutrophilic infiltration. Erosions may form; haemorrhage can be extensive ("haemorrhagic gastritis").
Chronic Gastritis
H. pylori–Associated Gastritis (Type B)
Helicobacter pylori is a gram-negative, flagellated, spiral bacterium that survives in the gastric mucus by producing urease, which generates an alkaline microenvironment. It is the strongest single risk factor for peptic ulcer disease and a WHO-classified carcinogen implicated in gastric adenocarcinoma and MALT lymphoma.
Key facts:
- Found in 60–80% of gastric ulcers and ~95% of duodenal ulcers
- Causes chronic active gastritis in almost 100% of infected persons
- Primarily affects the antrum
- Strains expressing CagA protein carry highest cancer/MALT lymphoma risk
Morphology:
- H. pylori resides in mucus overlying foveolar cells — concentrated in the antrum
- Neutrophilic infiltration of lamina propria ± pit abscesses
- Dense subepithelial plasma cells; lymphoid aggregates with germinal centres (induced MALT)
- Intestinal metaplasia (goblet cells + columnar absorptive cells) → precancerous
- Hyperplastic and inflammatory polyps
Diagnosis: H&E/Giemsa/Warthin-Starry silver stain; rapid urease test; urea breath test; stool antigen; serology; PCR
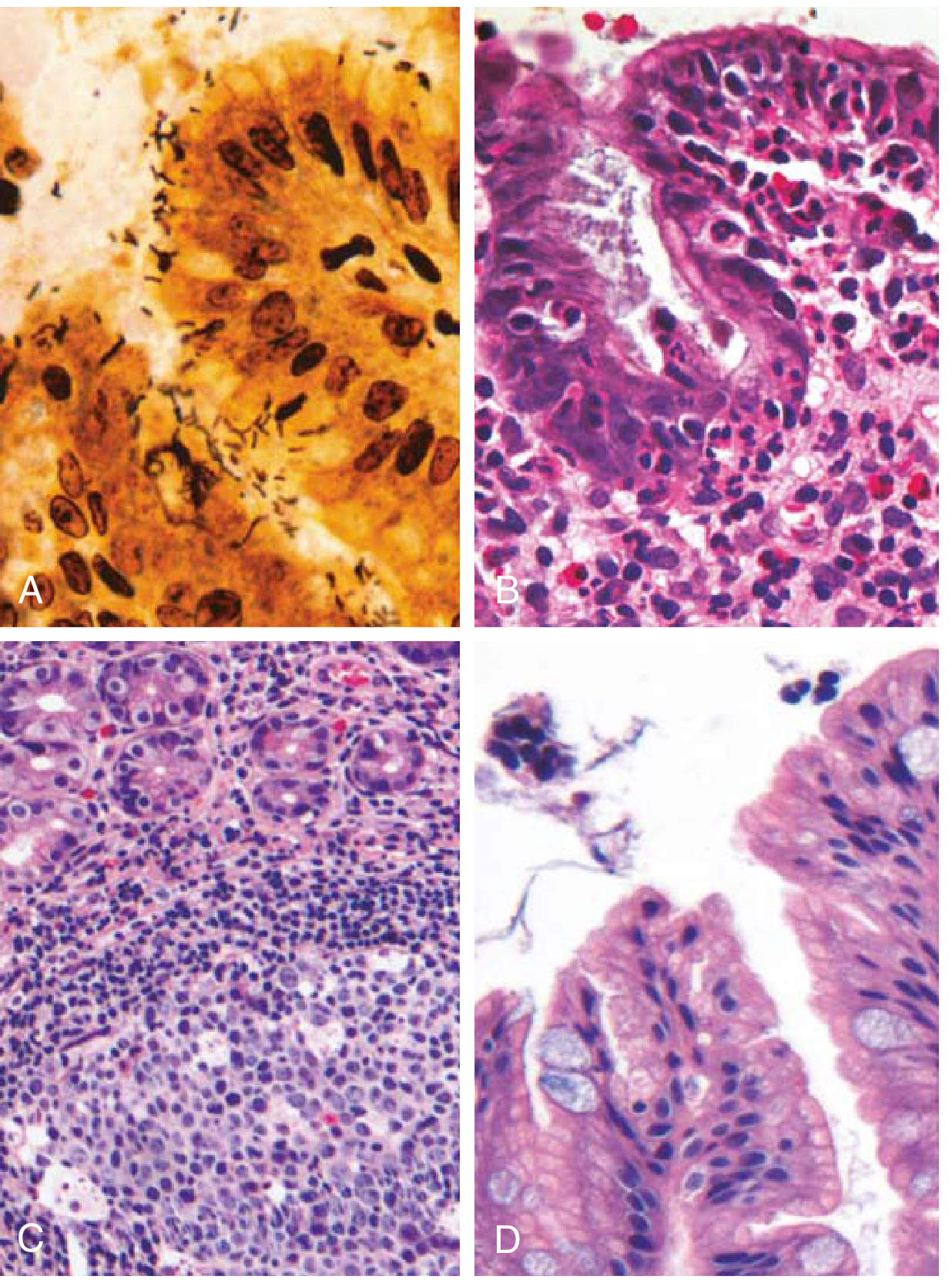
Autoimmune Gastritis (Type A)
Accounts for <10% of chronic gastritis. Caused by CD4+ T-cell–mediated and antibody-mediated destruction of parietal cells targeting H⁺,K⁺-ATPase (the proton pump) and intrinsic factor. Antibodies to H⁺,K⁺-ATPase and intrinsic factor are present in up to 80% of patients.
Features:
| Feature | H. pylori Gastritis | Autoimmune Gastritis |
|---|---|---|
| Location | Antrum | Body & fundus |
| Inflammatory infiltrate | Neutrophils, subepithelial plasma cells, germinal centres | Lymphocytes, macrophages (deeper, gland-centred) |
| Acid production | Increased → slightly decreased | Decreased (achlorhydria) |
| Gastrin | Normal → markedly increased | Markedly increased (hypergastrinemia) |
| Serology | Anti-H. pylori antibodies | Anti-parietal cell (H⁺,K⁺-ATPase, intrinsic factor) |
| Sequelae | PUD, adenocarcinoma, MALT lymphoma | Pernicious anaemia, adenocarcinoma, carcinoid tumour |
| Associations | Lower socioeconomic status | Hashimoto thyroiditis, T1DM, Addison disease |
Morphology: Diffuse body/fundus atrophy with thinned mucosa; loss of rugal folds; parietal and chief cell loss; intestinal metaplasia; endocrine (ECL) cell hyperplasia → risk of gastric carcinoid tumours
Clinical: Pernicious anaemia (megaloblastic, B12 deficiency); atrophic glossitis; peripheral neuropathy. Median age at diagnosis 60 years, slight female predominance.
Other Forms of Gastritis
- Eosinophilic gastritis: Dense eosinophilic infiltrate (antrum/pylorus), often with peripheral eosinophilia and raised IgE; associated with food allergy (milk, soy in children), parasites, systemic sclerosis
- Lymphocytic gastritis: Predominantly adult females; 40% linked to coeliac disease; marked increase in intraepithelial T-lymphocytes; endoscopic "varioliform" appearance
- Granulomatous gastritis: Contains granulomas; causes include Crohn disease (most common in Western populations), sarcoidosis, mycobacteria, fungi, CMV, H. pylori
2. Peptic Ulcer Disease (PUD)
PUD refers to chronic mucosal ulceration of the stomach or duodenum. Almost always associated with:
- H. pylori infection (chronic antral gastritis → ↑ acid + ↓ duodenal bicarbonate)
- NSAIDs (± corticosteroids; suppress prostaglandin synthesis → ↓ mucosal protection)
- Cigarette smoking
- Rarer: Zollinger-Ellison syndrome, CMV/HSV infection, cocaine (vasoconstriction), cirrhosis
Risk factors:
- H. pylori infection
- NSAIDs (especially in elderly >60 yrs)
- Smoking (synergises with H. pylori)
- COPD, psychological stress
- Zollinger-Ellison syndrome
Epidemiology: Incidence is falling with declining H. pylori prevalence; however, NSAID-associated PUD in elderly is rising. Duodenal ulcers are ~4× more common than gastric ulcers.
Morphology of a peptic ulcer:
- Round-to-oval punched-out defect with sharply demarcated, perpendicular walls ("cliff-like" edges)
- 4 histological zones from surface inward:
- Necrotic debris (acute inflammatory exudate)
- Non-specific fibrinoid necrosis
- Granulation tissue (vascular and fibroblastic)
- Collagenous scar (fibrosis) incorporating blood vessels with thickened walls (endarteritis obliterans)
- Actively inflamed margins; regenerating epithelium at edges
Complications:
- Haemorrhage (most common; posterior duodenal ulcers erode the gastroduodenal artery)
- Perforation (most dangerous; anterior ulcers; peritonitis)
- Obstruction (pyloric scarring → gastric outlet obstruction)
- Penetration (into adjacent organs: pancreas, liver)
- Malignant transformation (rare in gastric ulcers; duodenal ulcers do NOT undergo malignant change)
3. Gastric Polyps
| Type | Key Features |
|---|---|
| Hyperplastic polyps | Most common; elongated foveolar glands, inflamed stroma; associated with H. pylori and autoimmune gastritis; malignant potential very low |
| Fundic gland polyps | Common in PPIs users and FAP (APC mutation); cystic dilation of fundic glands; very low malignant potential |
| Gastric adenomas | Usually solitary antral lesions; all exhibit dysplasia (low or high grade); high-grade = nuclear atypia, cribriform structures; precursor to adenocarcinoma |
4. Gastric Adenocarcinoma
The most common gastric malignancy, comprising >90% of all gastric cancers.
Epidemiology
- High incidence in Japan, Chile, Costa Rica, Eastern Europe (up to 20× higher than North America)
- Incidence in the US has fallen ~85% since 1930 (↓ H. pylori prevalence, ↓ salt/smoked food, ↑ refrigeration)
- Exception: Gastric cardia adenocarcinoma is rising (linked to Barrett oesophagus, obesity, GERD)
Molecular Subtypes
The Cancer Genome Atlas (TCGA) classifies gastric adenocarcinoma into four groups:
- EBV-positive (~9%): Frequent PIK3CA mutations, DNA hypermethylation, extreme amplification of PD-L1/PD-L2
- MSI-high (~22%): Hypermutated; common in older women; MLH1 silencing by methylation; better prognosis
- Chromosomally unstable (CIN) (~50%): TP53 mutations, KRAS, ERBB2 amplification; predominates at gastroesophageal junction
- Genomically stable (GS) (~20%): Enriched for diffuse type; CDH1 (E-cadherin) and RHOA mutations; worst prognosis
Hereditary Gastric Cancer
- Hereditary diffuse gastric cancer: Loss-of-function mutations in CDH1 (E-cadherin) → up to ~30% of hereditary diffuse cases
- Lynch syndrome: Mismatch repair gene mutations → MSI → intestinal-type gastric cancer risk
Morphological Classification (Lauren)
| Type | Intestinal | Diffuse |
|---|---|---|
| Gross pattern | Bulky, polypoid or ulcerating mass | Infiltrating, thickened wall (linitis plastica) |
| Histology | Gland-forming; cohesive cells | Poorly cohesive/signet-ring cells; no glands |
| CDH1 | Preserved | Lost (E-cadherin loss) |
| Molecular | Often CIN, MSI, or EBV | Often GS |
| Pathogenesis | H. pylori → intestinal metaplasia → dysplasia | Often H. pylori or CDH1 mutations |
| Prognosis | Slightly better | Worse |
Signet-ring cells: Neoplastic cells with large mucin vacuoles displacing the nucleus to the periphery — pathognomonic of diffuse type.
Linitis plastica ("leather bottle stomach"): Extensive submucosal fibrosis from diffuse infiltration creates a shrunken, rigid stomach.
Spread and Metastases
- Virchow node: Left supraclavicular sentinel lymph node
- Sister Mary Joseph nodule: Periumbilical lymph node involvement
- Krukenberg tumour: Metastases to ovaries (bilateral; signet-ring cells within ovarian stroma)
- Blumer shelf: Deposits in the pouch of Douglas (palpable on rectal exam)
- Left axillary lymph nodes (Irish node)
Staging and Prognosis
- Early gastric cancer (EGC): Confined to mucosa ± submucosa, regardless of lymph node status → 90–95% 5-year survival
- Advanced cancer (involving muscularis propria or beyond) → poor prognosis; <20% of cases detected early in Western countries
- Workup: Endoscopy with biopsy; EUS (most reliable for T-staging); MDCT for locoregional + distant staging; PET-CT for metastases. CEA and CA-125 are of limited utility.
5. Gastric MALT Lymphoma
- Low-grade extranodal marginal zone B-cell lymphoma
- Arises from induced MALT secondary to H. pylori infection (especially CagA⁺ strains)
- Early-stage disease can regress completely with H. pylori eradication (antibiotic + PPI)
- Higher-grade transformation can occur; may acquire t(11;18) translocation — these do NOT respond to H. pylori eradication alone and require chemotherapy/rituximab
6. Other Gastric Tumours
| Tumour | Notes |
|---|---|
| Gastrointestinal stromal tumour (GIST) | Most common mesenchymal gastric tumour; driven by activating KIT (CD117) or PDGFRA mutations; spindle-cell morphology; treated with imatinib (KIT inhibitor) |
| Gastric carcinoid (neuroendocrine tumour) | Arises from ECL cells; associated with hypergastrinemia (autoimmune gastritis, MEN-1, Zollinger-Ellison); generally low-grade |
| Gastric lymphoma | Second most common gastric malignancy after adenocarcinoma |
Summary of Key Pathogenetic Relationships
H. pylori infection
├─→ Chronic antral gastritis
│ ├─→ Peptic ulcer disease
│ ├─→ Intestinal metaplasia → Dysplasia → Intestinal-type adenocarcinoma
│ └─→ MALT → MALT lymphoma
└─→ Diffuse gastritis
└─→ Diffuse-type adenocarcinoma (via additional CDH1 loss)
Autoimmune gastritis (Type A)
├─→ Achlorhydria → Pernicious anaemia
├─→ Intestinal metaplasia → Adenocarcinoma
└─→ ECL hyperplasia → Gastric carcinoid
Key sources: Robbins & Kumar Basic Pathology, Chapter 13 | Robbins, Cotran & Kumar Pathologic Basis of Disease, Chapter 17 | Grainger & Allison's Diagnostic Radiology | Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.