Weight loss 13kgs in 6months, headache for 1month, MRI brain contrast suggestive of neurocysticercosis, CSF ADA-19.58 U/L
neurocysticercosis CSF adenosine deaminase diagnosis
neurocysticercosis diagnosis treatment albendazole praziquantel
neurocysticercosis MRI brain cystic lesion
CSF ADA adenosine deaminase normal value tuberculosis meningitis vs neurocysticercosis differential diagnosis
Clinical Analysis
The MRI Finding: Neurocysticercosis
MRI Stages of NCC
| Stage | MRI Appearance |
|---|---|
| Vesicular | Cyst with CSF-like fluid; small hyperintense scolex = "hole-with-dot" sign (pathognomonic) |
| Colloidal vesicular | Cyst with ring/nodular enhancement; perilesional edema |
| Granular nodular | Smaller cyst, ring/nodular enhancement, less edema |
| Calcified nodular | Hyperdense calcification, no enhancement |
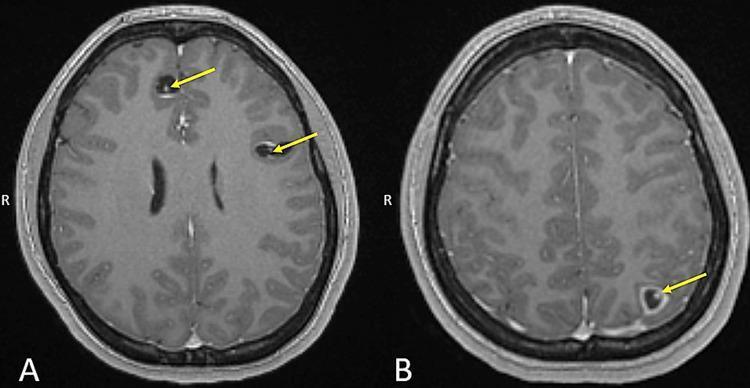
The Critical Question: CSF ADA of 19.58 U/L
What Elevated CSF ADA Means
CSF ADA Reference Values in Meningitis Studies
| Condition | Mean CSF ADA (U/L) |
|---|---|
| Tuberculous meningitis | 28.34 ± 14.83 |
| Neurobrucellosis | 8.71 ± 5.83 |
| Purulent (bacterial) meningitis | 6.18 ± 2.54 |
| Aseptic (viral) meningitis | 3.43 ± 3.48 |
| Cutoff for TBM | >10–12.4 U/L |
Interpreting the Full Clinical Picture
Two Diagnoses May Coexist — or NCC May Be a Red Herring
| Feature | Points Toward |
|---|---|
| 13 kg weight loss over 6 months | Active tuberculosis (constitutional/systemic TB) |
| Headache for 1 month | TBM or NCC-related raised ICP |
| MRI brain with cystic lesions | NCC (or rarely TB tuberculoma) |
| CSF ADA 19.58 U/L (elevated) | Strongly suggests TBM |
Key Clinical Reasoning
- NCC alone does not elevate CSF ADA significantly — studies confirm CSF ADA in aseptic/parasitic meningitis is low (3–8 U/L range).
- 13 kg weight loss in 6 months is a constitutional symptom far more typical of active tuberculosis (miliary or pulmonary TB) than of isolated NCC.
- The combination of elevated CSF ADA + significant constitutional weight loss + headache should prompt aggressive TB workup even if MRI suggests NCC.
- TB and NCC can co-exist in endemic populations (immunosuppression from TB increases susceptibility to CNS parasitosis, and both are endemic in similar geographic regions).
Recommended Workup
To Confirm/Exclude TBM
- CSF: CBNAAT/GeneXpert MTB/RIF Ultra (most sensitive), AFB smear & culture, cell count & differential (lymphocytic pleocytosis), protein, glucose
- CSF ADA >10 U/L supports TBM (this patient's value of 19.58 is significant)
- Serum LDH, CSF lactate
- CSF cytology (rule out lymphoma)
To Confirm NCC
- Serum ELISA (EITB) for anti-cysticercal antibodies — highly specific
- Check for extraneural cysts: fundoscopy (mandatory before antiparasitic therapy — to rule out ocular cysticercosis), subcutaneous nodules
- Stool examination for T. solium proglottids/eggs (in household contacts)
To Investigate Weight Loss
- Chest X-ray / CT chest — miliary TB, cavitary disease
- Sputum AFB × 3 / CBNAAT
- HIV serology (co-infection common, must be ruled out)
- IGRA (Quantiferon-TB Gold) or Mantoux test
- CBC (lymphopenia in TB, eosinophilia in parasitosis)
- Serum albumin (nutritional status)
Management Principles
If TBM is Confirmed (or High Suspicion)
- Intensive phase (2 months): HRZE (Isoniazid + Rifampicin + Pyrazinamide + Ethambutol)
- Continuation phase (7–10 months for TBM): HR
- Adjunctive dexamethasone (0.4 mg/kg/day tapering over 6–8 weeks) — reduces mortality and neurological sequelae
- Monitor for drug-induced hepatotoxicity (LFTs at baseline and 2 weeks)
For NCC (if confirmed active/viable cysts)
- Albendazole (15 mg/kg/day in 2 divided doses × 8–30 days) OR Praziquantel (50–100 mg/kg/day × 15–30 days)
- Concomitant corticosteroids (dexamethasone) to blunt inflammatory response from dying cysts — this overlap is favorable if TBM is being treated concurrently
- Anticonvulsants if seizures present
- Surgery for obstructive hydrocephalus or intraventricular cysts
- Caution: Anti-parasitic drugs are contraindicated before ruling out ocular cysticercosis (can cause retinal detachment)
⚠️ Important: If both TBM and NCC are present, dexamethasone/corticosteroids serve dual purposes (adjunctive in TBM + anti-inflammatory in NCC). However, anti-TB drugs should take priority; antiparasitic therapy can be introduced once TBM treatment is established.
Summary
| Finding | Interpretation |
|---|---|
| MRI — NCC pattern | Confirmed parasitic CNS infection (vesicular/colloidal stage) |
| CSF ADA 19.58 U/L | Elevated — strongly suggests co-existing TBM |
| 13 kg weight loss/6 months | Systemic TB most likely; rule out HIV/malignancy |
| Headache 1 month | Raised ICP from either/both |
Csf cell count 0-2 lymphocytes, sugar-144mg/dl, protein-168mg/dl, fundus examination- normal, esr-25mm/hr, crp-negative, mantoux -negative, ct chest- normal
CSF ADA elevated neurocysticercosis normal cell count protein normal glucose raised differential diagnosis
CSF protein 168 mg/dl glucose 144 mg/dl raised serum glucose interpretation blood CSF ratio
neurocysticercosis CSF normal findings parenchymal diagnosis
neurocysticercosis parenchymal CSF normal protein glucose cell count weight loss 13kg differential
Updated Clinical Re-Analysis
CSF Analysis — Detailed Interpretation
| Parameter | This Patient | Interpretation |
|---|---|---|
| Cell count | 0–2 lymphocytes | Normal / acellular |
| Glucose | 144 mg/dL | Mirrors hyperglycemia — not low |
| Protein | 168 mg/dL | Elevated (normal: 15–45 mg/dL) |
| ADA | 19.58 U/L | Elevated above 10 U/L cutoff |
The CSF Glucose — Critical Reinterpretation
⚠️ This is an important insight: the CSF glucose of 144 is not evidence against meningitis — it reflects the patient's glycemic state. A simultaneous fasting blood glucose must be checked to calculate the CSF:blood glucose ratio.
Does This CSF Profile Fit TBM?
| TBM Criterion | Expected | This Patient |
|---|---|---|
| CSF cells | >100/mm³, lymphocytic | 0–2 — absent |
| CSF glucose | <40 mg/dL or ratio <0.5 | Normal ratio (high due to hyperglycemia) |
| CSF protein | Elevated (usually >150 mg/dL) | 168 mg/dL — mildly elevated |
| Clinical ESR | Usually elevated | 25 mm/hr — normal |
| CRP | Usually elevated | Negative |
| Mantoux | Often positive | Negative |
| CT chest | Often abnormal | Normal |
Does the Elevated CSF ADA (19.58 U/L) Still Matter?
- Fungal meningitis
- CNS sarcoidosis
- Autoimmune encephalitis (GFAP-astrocytopathy)
- Lymphoproliferative disease/CNS lymphoma
- Neurocysticercosis (inflammatory stage)
- NCC itself — degenerating cysts trigger T-lymphocyte activation even with minimal pleocytosis in parenchymal disease
- CNS lymphoma — must now move up the differential given 13 kg weight loss + elevated CSF protein + elevated ADA + acellular CSF
- Carcinomatous meningitis — similar profile
The CSF Protein (168 mg/dL) — What It Tells Us
- Spinal block (Froin's syndrome)
- Guillain-Barré syndrome
- CNS lymphoma / leptomeningeal malignancy
- Chronic meningitis (e.g., fungal, NCC with basal involvement)
- Diabetic polyradiculopathy (relevant given the hyperglycemia)
- NCC (protein elevation in 40% of cases, even with normal cell counts)
Revised Differential Diagnosis
| Priority | Diagnosis | Supporting Evidence | Against |
|---|---|---|---|
| 1st | Parenchymal NCC (active/colloidal stage) | MRI findings, elevated CSF protein, CSF ADA (T-cell response), compatible epidemiology | Weight loss unexplained by NCC alone |
| 2nd | CNS lymphoma / leptomeningeal malignancy | 13 kg weight loss, elevated protein, elevated ADA, acellular CSF | MRI shows cystic NCC-like lesions (not typical lymphoma) |
| 3rd | Disseminated malignancy with CNS involvement | 13 kg constitutional weight loss | Normal CT chest |
| 4th | Fungal meningitis (Cryptococcus) | Acellular CSF, elevated protein, elevated ADA, weight loss if immunocompromised | No HIV mentioned, normal cells |
| 5th | TBM | (Previously considered) | Effectively excluded by acellular CSF, normal ESR/CRP, negative Mantoux, normal chest |
What Explains the 13 kg Weight Loss?
- Intestinal taeniasis (T. solium adult tapeworm in gut) — NCC and intestinal tapeworm coexist in 10–15% of cases; tapeworm can cause malabsorption, anorexia, and weight loss
- Undetected malignancy — must be excluded
- HIV/immunocompromised state — predisposes to both NCC and opportunistic infections
- Diabetes mellitus — the very high CSF glucose (144 mg/dL) and likely high serum glucose suggests possible uncontrolled DM, which causes weight loss
- Occult systemic infection elsewhere
Immediate Next Steps
Mandatory
- Simultaneous serum glucose — to calculate CSF:blood ratio and confirm/exclude hyperglycemia as cause of high CSF glucose
- HIV serology (ELISA + confirmatory Western blot) — weight loss + NCC is a classic HIV presentation
- Serum anti-cysticercal antibodies (ELISA/EITB) — confirm NCC diagnosis
- Stool examination — proglottids/ova of T. solium (intestinal taeniasis co-existing)
- HbA1c + fasting glucose — assess for uncontrolled diabetes
To Rule Out Malignancy (Given 13 kg weight loss + acellular CSF + elevated protein)
- CSF cytology (ideally repeat with large volume, 10 mL) — to detect leptomeningeal malignancy
- Serum LDH, β2-microglobulin
- CT abdomen-pelvis — occult malignancy (lymphoma, GI tumor)
- CSF flow cytometry if lymphoma suspected
To Rule Out Fungal Meningitis
- CSF India ink stain
- CSF cryptococcal antigen (CrAg)
- CSF fungal culture
Management of Confirmed Parenchymal NCC
- Albendazole 15 mg/kg/day (max 800 mg/day) in 2 divided doses × 8–28 days — preferred over praziquantel (better CNS penetration, less interaction with dexamethasone)
- Dexamethasone 0.1 mg/kg/day × 2 weeks — to blunt the inflammatory response as cysts die (prevents seizures from peri-cystic edema)
- Anticonvulsants if seizures occur
- No antiparasitic therapy if only calcified cysts (dead cysts — no benefit)
Summary
| Previous Concern | New Verdict |
|---|---|
| Elevated CSF ADA → TBM? | Unlikely — acellular CSF, normal ESR/CRP, negative Mantoux, normal chest CT exclude TBM |
| CSF glucose 144 → hyperglycorrhachia? | No — reflects patient's serum hyperglycemia; ratio likely normal |
| CSF protein 168 → meningitis? | Yes — NCC-related protein elevation; check for CNS lymphoma |
| 13 kg weight loss → ?TB | Re-investigate: HIV, DM, intestinal taeniasis, occult malignancy |
Hiv -negative
disseminated cysticercosis weight loss 13kg MRI brain neurocysticercosis CSF protein elevated diagnosis workup
disseminated cysticercosis weight loss cachexia clinical features
Clinical Synthesis — HIV Negative
What Explains the 13 kg Weight Loss in a HIV-Negative NCC Patient?
1. Disseminated Cysticercosis (Most Important to Exclude)
- Myositis and progressive muscle wasting → weight loss
- Subcutaneous nodules (palpable)
- "Rice-grain" calcifications on plain X-ray of limbs/trunk
2. Uncontrolled Diabetes Mellitus
- Progressive weight loss (glucosuria, catabolic state)
- Increased susceptibility to parasitic infections
- Elevated CSF protein (diabetic polyradiculopathy/neuropathy)
3. Intestinal Taeniasis (Co-existing Tapeworm)
- Malabsorption
- Nausea, reduced appetite
- Gradual weight loss over months
4. Occult Malignancy (Must Still Be Excluded)
- CSF cytology — leptomeningeal carcinomatosis
- CT abdomen-pelvis — lymphoma, GI/pancreatic malignancy
- Serum LDH, CA-125, CEA, AFP, PSA (age/sex-appropriate markers)
Revised Diagnostic Picture
| Finding | Most Likely Explanation |
|---|---|
| MRI brain — NCC pattern | Parenchymal NCC (vesicular/colloidal stage) |
| CSF glucose 144 mg/dL | Hyperglycaemia (likely DM) — not meningitis |
| CSF protein 168 mg/dL | NCC-related protein elevation ± diabetic polyradiculopathy |
| CSF ADA 19.58 U/L | T-cell response to degenerating NCC cysts |
| CSF cells 0–2 | Parenchymal NCC (acellular CSF is typical for parenchymal disease — 50% of cases have completely normal CSF) |
| 13 kg weight loss | Uncontrolled DM and/or disseminated cysticercosis and/or intestinal taeniasis |
| Normal ESR, CRP, Mantoux, CT chest | Effectively excludes active TB |
| HIV negative | Excludes immunocompromise as cause |
Immediate Priority Actions
| Investigation | Why |
|---|---|
| HbA1c + fasting blood glucose | Explain CSF glucose 144, weight loss, and ↑ susceptibility to NCC |
| Serum anti-cysticercal antibodies (EITB/ELISA) | Confirm NCC (specificity ~100%, sensitivity 98%) |
| Whole-body plain X-ray (thighs, arms, trunk) | Screen for disseminated cysticercosis (rice-grain calcifications) |
| Stool microscopy × 3 | Intestinal taeniasis co-infection |
| CSF cytology | Rule out leptomeningeal malignancy |
| CT abdomen-pelvis | Screen for occult malignancy |
| Blood glucose (simultaneous with LP record) | Calculate CSF:blood glucose ratio |
| CBC with differential | Eosinophilia supports parasitic infection |
Treatment Plan (Once DM and Malignancy Excluded)
| Drug | Dose | Duration |
|---|---|---|
| Albendazole | 15 mg/kg/day in 2 divided doses | 28 days (preferred) |
| Dexamethasone | 0.1 mg/kg/day (tapering) | 2–4 weeks — to cover inflammatory reaction from dying cysts |
| Anticonvulsants | If seizures develop | Until imaging shows resolution |
If DM is confirmed: glycaemic control is essential before and during antiparasitic therapy — steroid use will worsen hyperglycaemia and requires close glucose monitoring.
If disseminated cysticercosis confirmed: treatment is the same but requires longer and potentially repeated cycles of albendazole under close monitoring, as mass die-off of cysts can provoke severe inflammatory reactions.
Bottom Line
Hba1c-8.9, FBS-230, PPBS-320, urine sugar-3+, xray thighs and arms- normal
Case Solved
HbA1c 8.9% | FBS 230 mg/dL | PPBS 320 mg/dL | Urine sugar 3+ | X-ray limbs — Normal
Final Unified Diagnosis
How DM Explains Everything in This Case
| Clinical Finding | Explanation by DM |
|---|---|
| Weight loss 13 kg / 6 months | Uncontrolled DM — osmotic diuresis, glucosuria, catabolic state, insulin deficiency → protein/fat breakdown |
| CSF glucose 144 mg/dL | Passive equilibration with serum glucose (~230–320 mg/dL); CSF:blood ratio = ~0.6 → normal ratio, confirms no glucose consumption by infection |
| CSF protein 168 mg/dL | Diabetic neuropathy/polyradiculopathy causes blood-brain barrier disruption → protein leak; also NCC-related |
| CSF ADA 19.58 U/L | T-cell response to degenerating NCC cysts (not TBM) |
| CSF cells 0–2 | Parenchymal NCC: cysts are walled off within brain parenchyma — 50% of parenchymal NCC has completely normal CSF |
| Normal ESR, CRP | No systemic bacterial/TB infection |
| Normal Mantoux, CT chest | TB excluded |
| Normal fundus | No ocular cysticercosis — safe to give antiparasitic therapy |
| Normal X-ray limbs | No disseminated cysticercosis |
CSF Glucose Re-interpreted Definitively
| Parameter | Value |
|---|---|
| CSF glucose | 144 mg/dL |
| FBS (approximate at time of LP) | ~230 mg/dL |
| CSF : blood glucose ratio | 144/230 = 0.63 → Normal (normal: 0.6) |
| Interpretation | No hypoglycorrhachia — glucose is not being consumed; no active meningitis |
The Complete Pathophysiological Story
Poorly controlled T2DM (HbA1c 8.9%)
↓
Impaired cell-mediated immunity (T-cell dysfunction in DM)
↓
Susceptibility to Taenia solium cysticercus CNS invasion
↓
Parenchymal NCC → headache, elevated CSF protein, elevated ADA
↓
Osmotic diuresis + glucosuria + catabolism
↓
13 kg weight loss over 6 months
Final Diagnosis Summary
| # | Diagnosis | Status |
|---|---|---|
| 1 | Parenchymal Neurocysticercosis | ✅ Confirmed (MRI contrast, elevated ADA, compatible CSF) |
| 2 | Type 2 Diabetes Mellitus — poorly controlled | ✅ Confirmed (HbA1c 8.9%, FBS 230, PPBS 320, glucosuria 3+) |
| 3 | Tuberculous meningitis | ❌ Excluded (acellular CSF, normal ESR/CRP/Mantoux/CT chest) |
| 4 | Disseminated cysticercosis | ❌ Excluded (normal limb X-rays, normal fundus) |
| 5 | HIV | ❌ Excluded |
| 6 | Occult malignancy | Low probability — CT abdomen if weight loss persists post-treatment |
Treatment Plan
1. Antiparasitic Therapy (NCC)
| Drug | Dose | Duration |
|---|---|---|
| Albendazole | 15 mg/kg/day in 2 divided doses (typically 800 mg/day in adults) | 28 days |
| Dexamethasone | 0.1 mg/kg/day (6–8 mg/day), taper over 2 weeks | Given concurrently to suppress peri-cystic inflammation |
Albendazole is preferred over praziquantel: better CNS penetration, not affected by dexamethasone (praziquantel levels are reduced by corticosteroids and rifampicin).
2. Diabetes Management — Critical Priority
⚠️ Dexamethasone will worsen hyperglycaemia significantly. Blood glucose monitoring is mandatory 2–4 times daily during steroid co-administration.
| Step | Action |
|---|---|
| Initiate/optimise insulin therapy | Basal-bolus insulin preferred during steroid use (steroids blunt oral agents) |
| Daily glucose monitoring | FBS + PPBS during albendazole + dexamethasone course |
| Dietary counselling | Carbohydrate restriction, regular meals |
| Target HbA1c | <7.5% (moderate target during active illness) |
| Long-term | Transition to oral agents (Metformin ± others) once steroids stopped and glucose stabilised |
3. Symptomatic
- Analgesics (paracetamol) for headache during cyst die-off inflammatory phase
- Anticonvulsants (levetiracetam or phenytoin) if seizures occur during treatment
- Seizure precautions — advise patient, restrict driving
4. Follow-up
| Timeline | Action |
|---|---|
| 2 weeks | Blood glucose review, LFTs (albendazole is hepatotoxic) |
| 4 weeks | End of albendazole — repeat MRI brain with contrast |
| 3 months | Repeat HbA1c, reassess weight |
| MRI at 3 months | Assess cyst resolution/calcification |
Key Teaching Points from This Case
- CSF glucose must always be interpreted against simultaneous serum glucose — never in isolation. A CSF glucose of 144 is meaningless without knowing the blood glucose.
- Parenchymal NCC has normal CSF in 50% of cases — acellular CSF does not exclude NCC.
- Elevated CSF ADA is not pathognomonic for TBM — it reflects T-cell activation from any cause, including NCC.
- Uncontrolled DM is the single unifying diagnosis here — explains weight loss, high CSF glucose, impaired immunity predisposing to NCC, and elevated CSF protein.
- Dexamethasone + antiparasitic therapy in a diabetic — steroid-induced hyperglycaemia is predictable and must be pre-emptively managed with insulin.
Wt-64kgs
Dosing Calculations — Weight 64 kg
1. Albendazole
| Calculation | Value |
|---|---|
| 15 mg × 64 kg | = 960 mg/day |
| Standard adult cap | 800 mg/day (manufacturer's recommended maximum) |
| Prescribed dose | 800 mg/day (400 mg twice daily with fatty meal) |
| Duration | 28 days |
Albendazole absorption is significantly enhanced when taken with a fatty meal (e.g., milk, peanut butter) — increases bioavailability 5-fold. Counsel patient specifically.
2. Dexamethasone
| Calculation | Value |
|---|---|
| 0.1 mg × 64 kg | = 6.4 mg/day |
| Prescribed dose | 6 mg/day (practical rounding) |
| Route | Oral or IV |
| Duration | Start with albendazole, taper over 2 weeks after completing antiparasitic course |
| Week | Dexamethasone dose |
|---|---|
| Weeks 1–4 (with albendazole) | 6 mg/day |
| Week 5 | 4 mg/day |
| Week 6 | 2 mg/day |
| Week 7 | 1 mg/day → stop |
3. Albendazole Pre-treatment Weight Note
Diabetes Management at 64 kg
Insulin (Required during Dexamethasone course)
| Calculation | Value |
|---|---|
| Starting dose | 0.2 units/kg/day (conservative start during active illness) |
| 0.2 × 64 kg | = ~12–14 units glargine/detemir at night |
| Titrate | Increase by 2 units every 3 days if FBS >130 mg/dL |
- Add rapid-acting insulin (aspart/lispro) 4–6 units before main meals, titrated to PPBS
- PPBS target: <180 mg/dL during illness
Blood Glucose Monitoring Plan
| Time | Check |
|---|---|
| Fasting | Daily |
| 2 hours post-breakfast | Daily (steroids peak effect is morning) |
| Pre-dinner | Daily |
| 2 hours post-dinner | Alternate days |
⚠️ Dexamethasone particularly spikes post-lunch and afternoon glucose. Watch for afternoon hyperglycaemia even if fasting glucose appears controlled.
Complete Prescription Summary
| Drug | Dose | Frequency | Duration | Instructions |
|---|---|---|---|---|
| Albendazole | 400 mg | Twice daily | 28 days | With fatty meal |
| Dexamethasone | 6 mg | Once daily (morning) | 6 weeks total (tapering) | With food; monitor glucose |
| Insulin glargine | 12–14 units | Bedtime | Duration of steroid course → reassess | Titrate per FBS |
| Rapid insulin (if needed) | 4–6 units | Before main meals | As above | Titrate per PPBS |
| Pantoprazole | 40 mg | Once daily | Duration of steroids | GI protection with dexamethasone |
| Anticonvulsant (if seizure) | Levetiracetam 500 mg | Twice daily | Until imaging clear | Preferred in DM (no hepatic induction) |
Follow-up Schedule
| Visit | What to Check |
|---|---|
| Day 14 | LFTs (albendazole hepatotoxicity), blood glucose, HbA1c trend |
| Day 28 | End of albendazole — LFTs, blood glucose, symptom review |
| Week 8 | MRI brain with contrast (assess cyst resolution) |
| Month 3 | HbA1c (target improvement from 8.9%), weight check, reassess insulin need |
Expected Outcome
- NCC cysts → collapse → calcify over 2–6 months on MRI
- Headache should resolve within 2–4 weeks
- Weight should recover once glycaemic control is achieved — weight loss in DM is reversible with treatment
- Target: regain 5–8 kg over next 3 months as glucose normalises