Salter Harris classification
Reading File
Finding Sources
Searching Images
Salter Harris classification physeal fracture types
Reading File
Loading Image
Reading File
Reading File
Here is a comprehensive overview of the Salter-Harris classification of growth plate (physeal) fractures:
Salter-Harris Classification of Growth Plate Fractures
The physeal plate is a cartilaginous structure at the ends of long bones adjacent to the metaphysis. It is responsible for longitudinal bone growth and, during childhood, is weaker than surrounding bone and the attached ligaments/tendons — making it susceptible to fracture. The Salter-Harris system classifies these injuries based on the anatomical path of the fracture line.
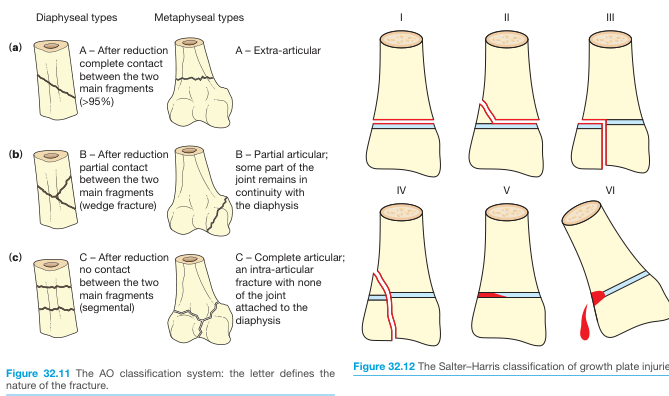
Type I — Through the Physis
- Fracture passes entirely through the physeal cartilage, separating the epiphysis from the metaphysis with no bone fragment.
- Nondisplaced injuries may appear normal on X-ray — diagnosis is clinical (point tenderness over the growth plate).
- MRI or stress views may be needed to confirm.
- Displaced injuries reduce easily (periosteum remains intact).
- Prognosis: Excellent — full growth potential preserved.
Type II — Through Physis + Metaphysis (most common, ~75%)
- Fracture extends through the physis and exits through the metaphysis, creating the classic Thurston-Holland fragment.
- Extraarticular. Periosteum remains intact on the metaphyseal side.
- Usually manageable with closed reduction.
- Prognosis: Good — growth disturbance is uncommon.
Type III — Through Physis + Epiphysis
- Fracture extends through the physis and exits through the epiphysis into the joint.
- Intraarticular — risks post-traumatic arthritis if not anatomically reduced.
- Open reduction often necessary to restore joint surface congruity.
- Prognosis: Good if blood supply to fragment is intact; poor if vascularity is disrupted.
Type IV — Through Metaphysis + Physis + Epiphysis
- Fracture crosses all three zones: metaphysis, physis, and epiphysis.
- Intraarticular and highly unstable.
- ORIF (open reduction and internal fixation) is typically required to maintain alignment across the physis.
- Prognosis: Guarded — high risk of physeal bar formation and growth disturbance if not perfectly reduced.
Type V — Crush Injury of the Physis (rarest)
- An axial compressive load crushes the physis between the epiphysis and metaphysis.
- May appear normal on initial X-ray — often diagnosed retrospectively when growth disturbance becomes apparent.
- Prognosis: Poor — high rate of premature physeal closure and resultant limb length discrepancy or angular deformity.
Type VI (Rang extension)
- Involves loss of a portion of the physis, typically due to a shearing or avulsion mechanism.
- Not in the original Salter-Harris description; added later by Rang.
- Results in a peripheral physeal bar and angular growth deformity.
Memory Aid — "SALTR"
| Type | Mnemonic | Structure involved |
|---|---|---|
| I | Slip | Physis only |
| II | Above | Physis + Metaphysis |
| III | Lower | Physis + Epiphysis |
| IV | Through | Metaphysis + Physis + Epiphysis |
| V | Ram (crush) | Compression of physis |
Key Clinical Points
- Growth plate injuries must always be considered in skeletally immature patients — ligaments are stronger than the physis, so "sprains" in children are often physeal fractures.
- Always palpate directly over the growth plate; tenderness there is a fracture until proven otherwise.
- Higher Salter-Harris numbers generally correlate with greater risk of growth disturbance, premature physeal closure, angular deformity, and limb length discrepancy.
- Complications that should be discussed with the patient/family: growth delay, growth arrest, and joint incongruity.
— Textbook of Family Medicine, 9e, p. 810–811 | Bailey and Love's Short Practice of Surgery, 28e, p. 444
Green stick frctures
Finding Sources
Finding Sources
Searching Images
greenstick fracture pediatric bone X-ray
Reading File
Reading File
Loading Image
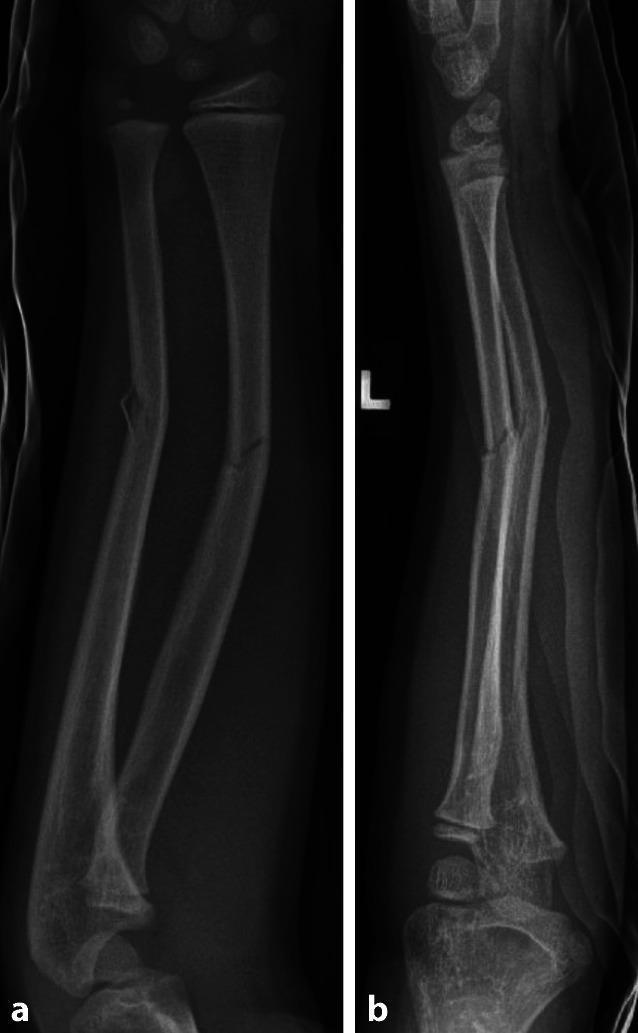
Greenstick Fractures
Definition & Mechanism
A greenstick fracture is an incomplete fracture unique to children, named after what happens when you try to snap a green (living) tree twig: the tension side cracks while the compression (concave) side merely buckles without fully breaking.
The key biomechanical features:
- Cortical disruption and periosteal tearing on the convex (tension) side
- Intact periosteum and buckled cortex on the concave (compression) side
- The bone bends and partially fractures rather than breaking completely through
Why Only in Children?
Pediatric bone has a higher collagen-to-mineral ratio than adult bone, making it more flexible and porous. This allows the bone to deform plastically rather than fracture completely. The thick, strong periosteum further prevents full displacement.
Radiographic Appearance
Greenstick fractures appear as:
- A fracture line/cortical break on one side of the bone only
- Bowing or angulation of the bone without a complete fracture line visible on the other side
- Soft tissue swelling is usually present
They can be subtle — careful inspection of the cortical contour is essential. Two orthogonal views (AP + lateral) are mandatory.
Comparison with Related Pediatric Fracture Patterns
| Fracture Type | Cortex Involved | Key Feature |
|---|---|---|
| Torus (buckle) | One cortex buckled, none disrupted | Most stable; compression side only deforms |
| Greenstick | One cortex cracked + opposite cortex buckled | One level worse than torus; partial break |
| Complete fracture | Both cortices disrupted | Full break through bone |
| Plastic deformation (bowing) | No visible cortical break | Diffuse deformation of entire diaphysis |
Clinical Features
- More stable and less painful than complete fractures — the intact periosteal hinge limits displacement
- Typically occurs in the forearm (radius/ulna) following a fall on an outstretched hand (FOOSH), but also seen in tibia, fibula, clavicle
- Angulation is common but significant displacement is rare
Management
- Closed reduction is usually sufficient — the intact periosteal hinge acts as a guide for reduction
- After reduction, the fracture is immobilized in a cast (typically long arm cast for forearm fractures)
- Remodeling capacity in children is excellent, so small residual angulation is often acceptable
- In some cases (especially unstable or significantly angulated fractures), the greenstick may need to be completed (intentionally converted to a complete fracture) to allow proper realignment and prevent re-angulation in cast
Prognosis
Generally excellent. The thick periosteum and robust remodeling potential of growing bone mean these fractures heal reliably with conservative management.
— Tintinalli's Emergency Medicine, p. 949 | Pfenninger and Fowler's Procedures for Primary Care, 3e
Sunderlands clasifictaion of nerve injury
Finding Sources
Finding Sources
Searching Images
Sunderland classification peripheral nerve injury degrees
Reading File
Reading File
Sunderland Classification of Nerve Injury
Sunderland expanded Seddon's classic 3-tier system into 5 grades (later extended to 6 by Mackinnon) by subdividing neurotmesis based on the specific connective tissue layers disrupted. Understanding nerve anatomy is essential to this classification.
Nerve Anatomy (layers, inside → out)
Axon → Endoneurium → Perineurium → Epineurium
- Endoneurium — surrounds individual axons
- Perineurium — surrounds each fascicle
- Epineurium — surrounds the entire nerve trunk
Sunderland Grades
| Grade | Seddon Equivalent | Structures Injured | Prognosis / Recovery |
|---|---|---|---|
| I | Neurapraxia | Myelin only (focal demyelination); axon intact | Full spontaneous recovery, often within hours–6 weeks |
| II | Axonotmesis | Axon + myelin; endoneurium, perineurium, epineurium intact | Full recovery — intact endoneurial tubes guide regeneration |
| III | Axonotmesis | Axon + myelin + endoneurium disrupted; perineurium & epineurium intact | Incomplete/variable recovery — intrafascicular scarring may impede regeneration |
| IV | Axonotmesis / Neurotmesis | Axon + myelin + endoneurium + perineurium disrupted; epineurium intact | Poor spontaneous recovery — surgical intervention usually required |
| V | Neurotmesis | Complete transection of all neural and connective tissue elements (including epineurium) | No spontaneous recovery — surgical repair mandatory |
| VI (Mackinnon) | Mixed | Combination of different grades within the same nerve trunk | Variable — reflects the complexity of high-energy injuries |
Detailed Notes on Each Grade
Grade I (Neurapraxia)
- Focal conduction block at the site of injury
- No Wallerian degeneration
- Axons structurally intact — only myelin is disrupted
- Classic example: Saturday night palsy (radial nerve compression at spiral groove) → wrist drop that resolves spontaneously
Grade II
- Axon is disrupted → Wallerian degeneration occurs distally
- Endoneurial tubes remain intact → provide a perfect scaffold for regenerating axons to reach target organs
- Complete and predictable recovery; rate ~1 mm/day axonal regeneration
Grade III
- Endoneurial disruption allows intrafascicular fibrosis and misdirection of regenerating axons
- Perineurium intact maintains fascicular architecture
- Recovery is incomplete; degree depends on extent of internal scarring
Grade IV
- Only the epineurium holds the nerve together as a gross structure
- Severe internal disorganization — fascicles are obliterated
- Surgical exploration nearly always required; nerve-in-continuity but non-functional (neuroma-in-continuity)
Grade V
- Complete nerve transection — no continuity whatsoever
- Surgical repair (primary neurorrhaphy or nerve grafting) is required
- Outcome depends on level of injury, gap length, time to repair, and patient age
Grade VI (Mackinnon)
- Seen in complex injuries (e.g., stretch + partial laceration)
- Different fascicles within the same nerve have different degrees of injury
- Requires careful intraoperative mapping before deciding on management
Seddon vs Sunderland — Correlation
Seddon Sunderland
─────────────────────────────────
Neurapraxia → Grade I
Axonotmesis → Grades II, III, IV
Neurotmesis → Grade V (+ VI)
Key Clinical Points
- Grades I–II: Observe; spontaneous recovery expected
- Grade III: Observation ± surgery depending on recovery progress at 3 months (EMG/NCS helps)
- Grades IV–V: Surgical intervention required — repair, grafting, or nerve transfer
- Wallerian degeneration (anterograde axon + myelin breakdown distal to injury) begins in Grade II and above
- EMG/nerve conduction studies are critical at 3–6 weeks post-injury to help grade severity and predict prognosis
- The higher the injury level (proximal lesions), the worse the outcome — regenerating axons must travel farther to reach denervated muscle, which atrophies irreversibly if reinnervation is delayed beyond ~18–24 months
— Bradley and Daroff's Neurology in Clinical Practice | Sabiston Textbook of Surgery, 21e
Wallerian degeneration
Finding Sources
Finding Sources
Searching Images
Wallerian degeneration nerve axon regeneration diagram
Reading File
Loading Image
Reading File
Wallerian Degeneration
Wallerian degeneration is the process of anterograde (distal) axon and myelin breakdown that occurs after a peripheral nerve axon is severed or severely injured (Sunderland grades II–V). Named after Augustus Waller, who first described it in 1850.
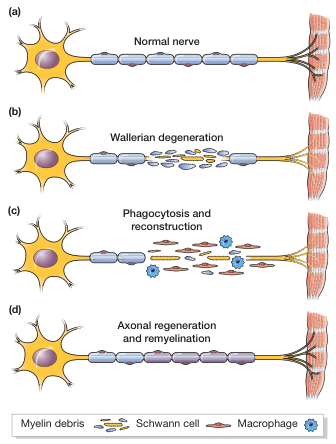
Phases of Wallerian Degeneration
1. Distal Segment Changes (begins within hours)
- Immediately after axonal injury, retrograde and anterograde axoplasmic transport is disrupted
- Rapid influx of extracellular calcium ions triggers activation of calcium-dependent proteases (calpains), which destroy the axonal cytoskeleton
- Within 24–48 hours: the axon distal to the injury site begins to fragment and disintegrate
- Myelin sheath surrounding the distal axon breaks down into ovoids of myelin debris
- Schwann cells dedifferentiate — they shift from a myelin-producing role to a "repair cell" role, driven by upregulation of c-Jun protein
- Macrophages are recruited (within a few days) and, together with activated Schwann cells, phagocytose the axon and myelin debris
2. Proximal Segment Changes
- A limited degree of retrograde degeneration extends proximally from the injury site — typically only up to the first node of Ranvier
- The cell body undergoes chromatolysis:
- Rough endoplasmic reticulum (Nissl substance) breaks up and disperses
- Cell nucleus becomes eccentrically displaced
- Gene expression shifts from axon maintenance → protein synthesis for regeneration
- Very proximal injuries (e.g., avulsion near the spinal cord) can cause apoptosis of the cell body itself, precluding any recovery
Bands of Büngner
After debris clearance, Schwann cells proliferate and align within the original basal lamina tubes to form longitudinal columns called bands of Büngner. These serve as the physical scaffold guiding regenerating axon sprouts from the proximal stump toward the target organ.
Nerve Regeneration After Wallerian Degeneration
| Mechanism | When it contributes | Context |
|---|---|---|
| Remyelination | Weeks–months | Grade I (neurapraxia) — no Wallerian; Schwann cells divide and remyelinate |
| Collateral sprouting | 3–6 months | Partial injury — intact adjacent axons sprout from nodes of Ranvier or terminals to reinnervate denervated fibres |
| Axon regeneration | 6–24 months | Complete/severe injury — growth cone advances from proximal stump along bands of Büngner |
- The growth cone (the advancing tip of the regenerating axon) extends via filopodia and lamellipodia
- Axon regeneration rate: approximately 1–3 mm/day (classically ~1 mm/day clinically)
- Guided by neurotropism — neurotrophins (NGF, BDNF), adhesion molecules (cadherins, laminins, immunoglobulin superfamily factors) secreted by Schwann cells
Why Timing Matters
- Denervated muscle undergoes progressive atrophy — after ~18–24 months of denervation, motor endplates and muscle fibres become irreversibly fibrotic
- This creates a race: the regenerating axon must reach the target muscle before the window for reinnervation closes
- More proximal injuries have worse outcomes because axons must regenerate over longer distances
Wallerian Degeneration vs. Other Responses to Nerve Injury
| Pathological Response | What happens | Seen in |
|---|---|---|
| Segmental demyelination | Myelin lost, axon intact | Mild compression (neurapraxia) |
| Wallerian degeneration | Axon + myelin breakdown distal to injury | Grades II–V (axonotmesis, neurotmesis) |
| Axonal degeneration | Distal ("dying back") axon loss | Metabolic/toxic neuropathies (diabetes, renal failure) |
Electrodiagnostic Correlates
- Immediately after injury, distal nerve stimulation can still evoke a response (axon physically present but disconnected)
- Wallerian degeneration renders the distal segment electrically inexcitable after ~5–7 days (motor fibres) to ~10–11 days (sensory fibres)
- EMG: fibrillations and positive sharp waves appear 2–3 weeks after injury, confirming denervation
— Bradley and Daroff's Neurology in Clinical Practice | Bailey and Love's Short Practice of Surgery, 28e
Classify fractures. How do fractures heal. Radiographic appearance of a long bone fracture
Finding Sources
Finding Sources
Searching Images
classification of fractures types diagram long bone
Finding Sources
Finding Sources
Searching Images
fracture healing stages callus formation radiograph
Reading File
Reading File
Reading File
Reading File
Here is a comprehensive overview of all three topics:
1. Classification of Fractures
Fractures are classified across multiple parameters. The complete description of any fracture should address all of them.
A. Skin Integrity
| Type | Definition |
|---|---|
| Closed | Skin intact over the fracture |
| Open (compound) | Fracture communicates through a skin breach — contaminated by definition; risk of osteomyelitis |
B. Fracture Pattern / Geometry
| Pattern | Description | Mechanism |
|---|---|---|
| Transverse | Fracture line perpendicular to the long axis | Direct blow |
| Oblique | Fracture line at an angle to the long axis | Combined bending + axial force |
| Spiral | Fracture winds around the shaft | Rotational/torsional force |
| Longitudinal | Fracture runs along the long axis | Axial split |
| Comminuted | >2 fragments | High-energy or crushing mechanism |
| Segmental | Isolated mid-segment between two fracture lines | High-energy |
| Impacted | One fragment driven into the other | Axial compression |
| Avulsion | Fragment pulled off by a tendon or ligament | Sudden muscular contraction |
| Stress (fatigue) | Repeated submaximal loading — no single traumatic event | Overuse |
| Pathological | Fracture through diseased bone (tumor, infection, osteoporosis) | Minimal/no trauma |
C. Incomplete Fractures (Pediatric)
| Type | Description |
|---|---|
| Torus (buckle) | One cortex buckles, none fully disrupted |
| Greenstick | Tension-side cortex cracks, compression-side buckles |
| Plastic deformation | Diffuse bowing — no visible fracture line |
D. Displacement & Alignment
- Nondisplaced — fragments in anatomic alignment; no reduction needed but may need immobilization
- Displaced — described by direction of the distal fragment relative to proximal
- Angulated — described by the direction the apex points (e.g., apex anterior = apex volar)
- Shortened — fragments overlap (bayonet apposition)
- Rotated — fragment has twisted on its long axis
- Distracted — fragments pulled apart
E. Articular Involvement
- Intraarticular — fracture line extends into the joint surface → risk of post-traumatic arthritis; anatomic reduction mandatory
- Extraarticular — fracture does not enter the joint
F. Anatomical Location Within the Bone
- Diaphyseal (shaft) — middle portion
- Metaphyseal — flared ends near the growth plate
- Epiphyseal — the articular end; physeal fractures in children classified by Salter-Harris
G. Special Pediatric: Salter-Harris (growth plate fractures)
Types I–V based on involvement of physis, metaphysis, and epiphysis (covered in prior session).
H. Open Fracture Severity: Gustilo-Anderson Classification
| Grade | Key Features |
|---|---|
| I | <1 cm wound, clean, minimal soft tissue injury |
| II | >1 cm wound, moderate soft tissue injury |
| IIIA | Extensive soft tissue injury but adequate coverage possible |
| IIIB | Extensive soft tissue loss, periosteal stripping, bony exposure |
| IIIC | Associated arterial injury requiring repair for limb salvage |
2. How Fractures Heal
Fractures heal by either primary (direct) or secondary (indirect) mechanisms.
Primary (Direct) Bone Healing
- Occurs when fragments are rigidly fixed with anatomical compression (e.g., compression plating)
- Requires absolute stability — no micromotion
- Osteoclastic cutting cones cross the fracture line, creating resorption canals → filled immediately by osteoblasts
- Results in direct Haversian remodelling with no external callus formation
- Radiographically: fracture line gradually disappears without visible callus
Secondary (Indirect) Bone Healing (most common)
Occurs with non-rigid fixation (casts, intramedullary nails, external fixators). Proceeds through 4 stages:
Stage 1 — Haematoma / Inflammation (hours–days)
- Fracture disrupts periosteum, endosteum, and surrounding soft tissues → fracture haematoma forms
- Entrapped erythrocytes in a fibrin network at the fracture ends
- Bone necrosis occurs at fractured ends
- Platelets degranulate → release cytokines (TNF-α, IL-1, IL-6, IL-11, IL-18)
- Neutrophils arrive first → followed by macrophages
- Fibroblasts and capillaries proliferate (granulation tissue formation)
- Mesenchymal stem cells migrate from periosteum and bone marrow
Stage 2 — Soft Callus (days–weeks)
- Granulation tissue is replaced by fibrocartilaginous soft callus
- Chondroblasts differentiate from periosteal stem cells
- Callus stabilises the fracture ends and grows even without perfect apposition
- Radiographically: fracture line still visible; soft tissue swelling
Stage 3 — Hard Callus (weeks–months)
- Two processes occur simultaneously:
- Intramembranous ossification — osteoblasts from periosteum deposit new bone around the outer surface of the callus, forming a bony sheath
- Endochondral ossification — the fibrocartilage within the callus calcifies, then is replaced by woven bone
- Together these form a hard (bony) callus — a fusiform, radio-opaque mass bridging the fracture
- Radiographically: callus appears as irregular radiodense fusiform mass bridging the fracture; original fracture line becomes less distinct
Stage 4 — Remodelling (months–years)
- Osteoclasts and osteoblasts remodel the woven bone callus into lamellar bone
- The medullary canal is restored
- Bone returns toward its original cortical architecture
- In children: remodelling can correct significant angular deformity
- Radiographically: callus shrinks, cortex reforms, medullary canal re-establishes
Factors Affecting Fracture Healing
| Promotes healing | Impairs healing |
|---|---|
| Youth | Advanced age |
| Good blood supply | Poor vascularity / avascular bone |
| Adequate immobilisation | Excessive movement / instability |
| Adequate apposition | Large fracture gap |
| Cancellous bone | Cortical bone (slower) |
| Adequate nutrition (Ca²⁺, Vit C, Vit D) | Malnutrition |
| Infection-free wound | Infection |
| — | NSAIDs (COX-2 inhibition reduces callus) |
| — | Corticosteroids, smoking |
3. Radiographic Appearance of a Long Bone Fracture
Acute Fracture (Day 0–2)
- Lucent (dark) fracture line through the cortex
- Loss of cortical continuity
- Soft tissue swelling adjacent to the fracture
- Joint effusion if intraarticular
- Fracture pattern, displacement, angulation, and comminution visible
Early Healing (Week 1–2)
- Fracture line may appear slightly wider as bone ends are resorbed
- Soft tissue swelling persists
- No callus visible yet (too early to mineralise)
Soft Callus Phase (Week 2–4)
- Still no definite mineralisation
- Periosteal elevation may be visible as a thin line lifted off the cortex
Hard Callus Phase (Week 4–12)
- Periosteal new bone and external callus appear as hazy, irregular, radiodense material surrounding the fracture site — the classic "fluffy" or "fusiform" density bridging the fracture
- Fracture line begins to blur and become indistinct
- Internal callus (endosteal) also visible as density filling the medullary canal
Remodelling Phase (Months–Years)
- Callus progressively shrinks and becomes more dense and regular
- Fracture line disappears
- Cortex reforms
- Medullary canal re-establishes
- Eventually the fracture site may be radiographically undetectable
Key Radiographic Principles for Any Long Bone Fracture
- Always obtain two orthogonal views (AP + lateral minimum)
- Image the joint above and below the fracture to detect associated injuries
- Describe: bone involved; region (diaphysis/metaphysis/epiphysis); pattern; displacement; angulation (apex direction); comminution; articular involvement; open vs closed; physeal involvement in children
— Sabiston Textbook of Surgery, 21e | Histology: A Text and Atlas (Pawlina) | Textbook of Family Medicine, 9e | Rockwood and Green's Fractures in Adults, 10e
This is a shared conversation. Sign in to Orris to start your own chat.