Familiarization of students with the structure of the ENT service. Mastering methods of examination of ENT organs. Working with the frontal reflector. Clinical anatomy, phy siology, methods of examination of the and paranasal sinuses. Acute and chronic diseases of the nose. Trauma of the nose. Epistaxis. Anterior and posterior tamponade. Hematoma and abscess of the nasal septum
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Reading File
Reading File
Now I have comprehensive information to compile a full educational summary. Let me synthesize everything from the sources.
ENT Service: Nose & Paranasal Sinuses — Complete Study Guide
1. Structure of the ENT Service & Working with the Frontal Reflector
The ENT (otolaryngology) service is organized around diseases of the ear, nose, throat, head, and neck. Examination requires a systematic approach with specialized instruments.
The Frontal Reflector (Head Mirror)
- A concave mirror worn on the clinician's forehead, with a central perforation for the examiner's eye
- Reflects ambient light (from a lamp positioned behind/to the side of the patient) into the examined cavity
- Used for all ENT examinations: nasal, oral, pharyngeal, and laryngeal
- The examiner looks through the central hole, keeping both eyes open, allowing binocular depth perception
- The light source is placed ~30 cm behind and to the right of the patient; the mirror is angled to direct the reflected beam into the field of examination
General ENT Physical Examination Sequence
- External inspection (deformity, swelling, asymmetry)
- Palpation (tenderness, masses, lymph nodes)
- Speculum/endoscopic examination of the cavity
- Cranial nerve assessment when indicated
2. Clinical Anatomy of the Nose
External Nose
The external nose is composed of:
- Bony framework: nasal bones (paired), frontal processes of the maxilla, nasal spine of the frontal bone
- Cartilaginous framework: upper lateral cartilages (ULC), lower lateral cartilages (LLC, "alar cartilages"), septal cartilage
- The keystone area is the critical articulation between the nasal bones and the upper lateral cartilages — trauma here leads to the "open roof deformity"
Nasal Septum
The septum has three main components:
- Perpendicular plate of the ethmoid (posterosuperior bony)
- Vomer (posteroinferior bony)
- Quadrangular cartilage (anterior cartilaginous)
The septomaxillary ligament joins the septum to the anterior nasal spine; growth forces here influence maxillary development.
Nasal Cavity
Each nasal cavity contains three turbinates (conchae): inferior, middle, and superior. Beneath each turbinate is the corresponding meatus:
- Inferior meatus: drains the nasolacrimal duct
- Middle meatus: drains the maxillary, frontal, and anterior ethmoid sinuses (via the ostiomeatal complex)
- Superior meatus: drains the posterior ethmoid and sphenoid sinuses (sphenoethmoid recess)
Blood Supply — Critical for Epistaxis
The nasal cavity receives its blood supply from both internal and external carotid systems:
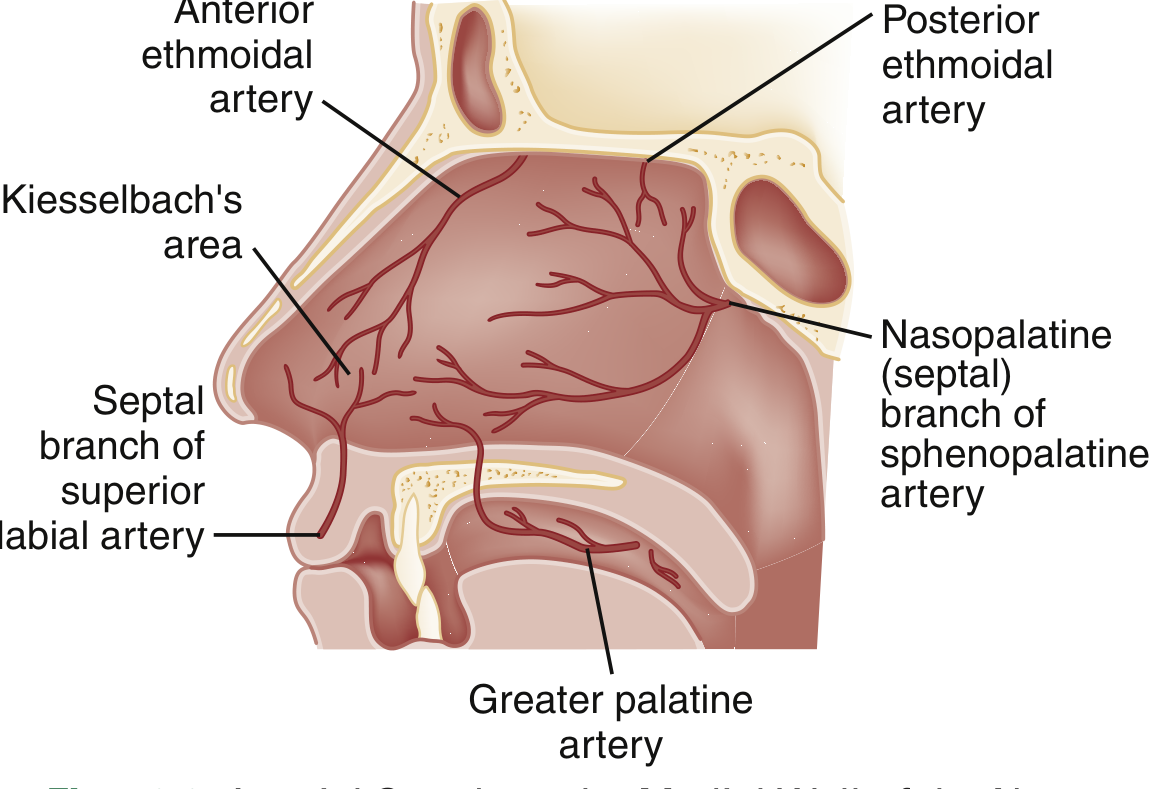
- Sphenopalatine artery (external carotid → maxillary): supplies turbinates laterally, posterior and inferior septum
- Anterior and posterior ethmoidal arteries (internal carotid → ophthalmic): supply superior mucosa
- Superior labial artery (external carotid → facial): supplies anterior mucosal septum
- Greater palatine artery: supplies inferior nasal floor
Kiesselbach's plexus (Little's area): anastomotic network on the anteroinferior septum where all four arteries meet — the most common site of epistaxis (~90% of cases).
3. Paranasal Sinuses — Anatomy & Development
| Sinus | Drains Into | Visible on X-ray | Notes |
|---|---|---|---|
| Maxillary | Middle meatus | At birth | Largest sinus; first to develop (10 weeks' gestation) |
| Ethmoid | Middle & superior meatus | After age 2 | Basal lamella of middle turbinate separates anterior/posterior cells |
| Frontal | Middle meatus (frontal recess) | ~6 years | Absent in Down syndrome (30%) |
| Sphenoid | Sphenoethmoid recess | After 6–7 years | Dangerous proximity to optic nerve, ICA, cavernous sinus |
Examination Methods
- Anterior rhinoscopy: nasal speculum + frontal reflector; assess mucosa, septum, turbinates
- Posterior rhinoscopy: small mirror placed in oropharynx to visualize nasopharynx, choanae
- Nasal endoscopy: rigid (0°, 30°, 70° telescopes) or flexible fiberoptic endoscope — gold standard for evaluating the ostiomeatal complex
- Transillumination: shining a light into a darkened sinus to assess air content — low specificity, rarely used
- Imaging: CT scan (gold standard for sinusitis/anatomy); MRI for soft tissue/fungal disease; plain X-ray has limited utility
4. Physiology of the Nose
- Warming and humidification of inspired air (via the mucosa and turbinate vascular plexus)
- Filtration: mucociliary clearance; nasal hairs (vibrissae) trap large particles
- Olfaction: olfactory epithelium in the superior nasal vault (cranial nerve I)
- Nasal cycle: alternating congestion and decongestion of each side (~every 2–4 hours), regulated by the autonomic nervous system
- Resonance: contributes to vocal quality
5. Rhinoscopy — Examination Technique
Anterior rhinoscopy (nasal speculum):
- Patient sits upright, head neutral
- Gently insert nasal speculum, open blades vertically
- First position (head neutral): examine nasal floor, inferior turbinate, anterior septum
- Second position (head tilted back 30°): examine middle turbinate and meatus
- Key findings: mucosal color and swelling, septal deviation, polyps, discharge, foreign bodies, masses
Posterior rhinoscopy (mirror technique):
- Warm a small nasopharyngeal mirror to prevent fogging
- Depress the tongue, introduce the mirror behind the uvula
- Illuminate with the frontal reflector
- Visualize: choanae, posterior end of the turbinates, Eustachian tube orifices, adenoids, nasopharyngeal wall
6. Acute & Chronic Rhinitis
Acute Rhinitis (Common Cold)
- Viral etiology (rhinovirus most common); lasts 7–10 days
- Symptoms: watery rhinorrhea → mucopurulent discharge, nasal obstruction, sneezing, low-grade fever
- Treatment: symptomatic (saline irrigation, decongestants, analgesics); antibiotics not indicated
Chronic Rhinitis
- Allergic rhinitis: IgE-mediated; pale, boggy, bluish turbinates; watery rhinorrhea; treat with intranasal corticosteroids, antihistamines
- Vasomotor rhinitis: non-allergic, triggered by temperature/humidity/odors
- Hypertrophic rhinitis: irreversible turbinate enlargement, often from chronic inflammation
- Atrophic rhinitis (ozena): progressive atrophy of mucosa and turbinates; foul-smelling crusts; may be associated with Klebsiella ozaenae
Nasal Polyps
- Benign, grape-like mucosal outgrowths arising from the ethmoid sinuses or middle meatus
- Associated with: chronic sinusitis, aspirin sensitivity (Samter's triad: asthma + aspirin sensitivity + polyps), cystic fibrosis (suspect CF in children <10 years with polyps)
- Appear pale, glistening, non-tender (distinguished from turbinates by lack of pain on probing)
- Treatment: intranasal corticosteroids; endoscopic polypectomy for refractory cases
7. Acute & Chronic Sinusitis
Acute Sinusitis (Rhinosinusitis)
Definition: Inflammation of ≥1 paranasal sinus lasting <4 weeks.
Etiology:
-
90% viral (rhinovirus, influenza)
- Bacterial superinfection in <2% (most common: S. pneumoniae, H. influenzae, M. catarrhalis)
Symptoms: Purulent nasal discharge, facial pain/pressure/fullness, hyposmia/anosmia, fever, maxillary toothache, postnasal drip
Examination: Direct rhinoscopy reveals mucopurulent discharge in middle meatus; tenderness over affected sinuses
Antibiotic criteria (all must be met): persistent symptoms >10 days without improvement, or severe symptoms (fever ≥39°C + purulent discharge >3–4 days), or worsening symptoms after initial improvement ("double-sickening")
- First-line: amoxicillin-clavulanate for 5–7 days
Complications (rare but serious): orbital cellulitis/abscess, osteomyelitis (Pott's puffy tumor of frontal bone), meningitis, intracranial abscess, cavernous sinus thrombosis
Invasive fungal sinusitis: emergency in immunocompromised patients (uncontrolled diabetes, transplant recipients); caused by Mucorales or Aspergillus; requires urgent endoscopic biopsy and gadolinium-enhanced MRI
Chronic Sinusitis
Definition: Symptoms lasting >12 weeks
Treatment:
- Intranasal corticosteroids (mometasone, fluticasone, budesonide rinses)
- Saline irrigation (high-volume washes more effective)
- Short courses of oral corticosteroids for polypoid disease
- Macrolide antibiotics (3 months) for chronic sinusitis without polyps (modest evidence)
- Endoscopic sinus surgery (ESS) for patients failing medical therapy — restores patency of the ostiomeatal complex
8. Nasal Trauma & Nasal Fracture
Nasal bones are the most commonly fractured facial bones.
Clinical features:
- Epistaxis, swelling, tenderness, deformity
- Periorbital ecchymosis without other orbital injury is suggestive of nasal fracture
- Nasal bone mobility — virtually diagnostic; assess by grasping the nasal dorsum and rocking the pyramid
- Always examine for septal hematoma (an emergency)
Diagnosis: Clinical; plain films rarely change management. Ultrasound is an alternative. CT reserved for suspected intracranial injury or complex facial fractures.
Management:
- Exclude associated injuries and septal hematoma
- Topical vasoconstrictors + anterior rhinoscopy to assess septum
- Closed reduction: indicated for significant displacement/obstruction — best performed at 5–10 days (after swelling resolves, before fibrous union)
- Open rhinoplasty/septorhinoplasty for complex cases
9. Epistaxis
Classification
- Anterior epistaxis (~90%): arises from Kiesselbach's plexus; typically unilateral, manageable in outpatient/ED
- Posterior epistaxis (~10%): involves the sphenopalatine artery; more severe, bilateral; predominantly in older adults with comorbidities; requires inpatient management
Etiology
- Local: trauma (nose-picking), mucosal dryness (winter/dry air), rhinitis, topical nasal sprays, septal deviation
- Systemic: anticoagulants/antiplatelets, bleeding disorders (von Willebrand disease, hemophilia), thrombocytopenia, hereditary hemorrhagic telangiectasia (HHT/Osler-Weber-Rendu), hypertension (associated with persistent bleeding, though causal link not established)
Initial Management
- Assess airway, hemodynamics, tissue perfusion
- Patient leans forward (prevents swallowing of blood)
- Patient blows nose to clear clots
- Apply bilateral pressure on the cartilaginous septum for 10–15 minutes (nose clip superior to manual pressure alone)
- Topical vasoconstrictor: oxymetazoline 0.05% (2 sprays) before applying pressure — improves hemostasis and facilitates examination
- Tranexamic acid is an effective adjunct (decreases immediate and delayed bleeding)
Cautery
- If a bleeding point is identified: chemical cautery with silver nitrate — circumferential cauterization around the vessel, then over it
- Applied to one side of the septum at a time to avoid perforation
- Indication: anterior epistaxis with visible bleeding point
Anterior Nasal Tamponade (Packing)
Indicated when pressure and cautery fail.
- Absorbable materials: oxidized regenerated cellulose (Surgicel), gel foam — expand on contact with blood; no removal needed
- Non-absorbable ribbon gauze: petroleum jelly (Vaseline) gauze layered in a "stacking" fashion from the floor up, using bayonet forceps; removed at 48–72 hours
- Nasal tampons/sponges (Merocel): compressed foam inserted dry and expands with saline or blood; easy to insert; removed after 48 hours
- Balloon catheters: dedicated epistaxis balloons (e.g., Rapid Rhino) with a PVA fabric covering — inflate anterior balloon first
- Prophylactic antibiotics: routine use is not recommended with anterior packing
Posterior Nasal Tamponade (Packing)
Indicated for posterior epistaxis failing anterior packing.
Traditional method (gauze pack):
- A Foley catheter (12–14 Fr) or dedicated posterior balloon catheter is passed through the affected nostril to the nasopharynx
- The balloon is inflated with saline (5–10 mL) to form a posterior tampon
- The catheter is then pulled anteriorly until resistance is felt (seating the balloon in the choana)
- An anterior pack is then placed in the ipsilateral naris
- The catheter is secured with an umbilical clamp over a dental roll at the nostril — care taken not to apply pressure to the nasal ala
- Patient requires hospitalization and monitoring (risk of hypoxia, bradycardia from vagal stimulation — "nasopulmonary reflex")
Refractory epistaxis: angiographic embolization of the sphenopalatine artery or surgical ligation (endoscopic sphenopalatine artery ligation)
10. Hematoma of the Nasal Septum
Pathophysiology: Mechanical trauma ruptures perichondrial blood vessels → blood accumulates in the submucoperichondrial space → cartilage is stripped of its blood supply → risk of avascular necrosis
- Occurs in 0.8–1.6% of nasal trauma cases
- More common in males and children (mucoperichondrium is less adherent)
Clinical features:
- Bilateral, purple/dusky, fluctuant, soft swellings on the septum
- Nasal obstruction (often complete)
- Pain (disproportionate to the injury)
- Widened septum on anterior rhinoscopy
Management — emergency:
- Prompt incision and drainage under local/general anaesthesia
- Incision placed on the convex side of the swelling, along the floor of the nasal cavity
- Thorough drainage of clot and irrigation
- Quilting sutures or bilateral nasal packing to obliterate the dead space and prevent re-accumulation
- Follow-up in 24–48 hours to check for re-accumulation
Untreated consequences: fibrocartilaginous replacement → saddle nose deformity, or secondary infection → septal abscess
11. Abscess of the Nasal Septum
Pathophysiology: Most commonly results from an infected hematoma. Other causes: nasogastric tube injury, furunculosis, sinusitis (direct extension from sphenoiditis), dental infection, post-septoplasty
Causative organisms:
- S. aureus (70%) — including MRSA
- H. influenzae, group A beta-hemolytic streptococcus, S. pneumoniae, K. pneumoniae
Clinical features:
- Widened, tense, painful septal swelling (bilateral)
- Fever, nasal obstruction, erythema of the nasal vestibule
- Fluctuance on palpation
Management:
- Prompt operative incision and drainage
- Culture-directed antibiotic therapy (empiric: anti-staphylococcal coverage; IV for severe cases)
- Bilateral nasal packing to prevent re-accumulation
Complications (life-threatening if delayed):
- Saddle nose deformity (cartilage destruction)
- Nasal valve collapse
- Facial cellulitis, sepsis
- Orbital extension
- Cavernous sinus thrombosis (via ophthalmic veins)
- Intracranial extension: epidural abscess, meningitis, intracranial abscess
Key point: Cavernous sinus thrombosis is the most feared intracranial complication, occurring via retrograde spread through the valveless facial and ophthalmic veins.
Summary Table: Epistaxis Management
| Step | Anterior | Posterior |
|---|---|---|
| First-line | Direct pressure 10–15 min + oxymetazoline | Same |
| Adjunct | Tranexamic acid | Tranexamic acid |
| If bleeding found | Silver nitrate cautery | — |
| Packing | Anterior tampon/gauze/balloon | Posterior balloon + anterior pack |
| Setting | Outpatient/ED | Inpatient (monitoring required) |
| Prophylactic antibiotics | Not routinely recommended | Considered (prolonged packing) |
| Refractory | Embolization or surgery | Sphenopalatine artery ligation/embolization |
Sources: Rosen's Emergency Medicine — Concepts and Clinical Practice; Tintinalli's Emergency Medicine; Harrison's Principles of Internal Medicine 22e; K.J. Lee's Essential Otolaryngology; Cummings Otolaryngology Head and Neck Surgery
continue
Asking for Preferences
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
ENT Service — Continued: Ear, Pharynx & Larynx
Part II: The Ear
12. Clinical Anatomy of the Ear
External Ear
Auricle (Pinna)
- Elastic cartilage covered by skin; the lobule contains only adipose tissue (no cartilage)
- Key landmarks: helix, antihelix, tragus, antitragus, concha, scapha, triangular fossa
- Perichondrium adheres tightly to the cartilage — this is why auricular hematomas are surgical emergencies (stripped perichondrium = cartilage necrosis)
External Auditory Canal (EAC)
- ~35 mm total length; 1/3 cartilaginous (lateral) + 2/3 bony (medial, derived from the tympanic bone)
- The canal makes an S-shaped curve: to examine the tympanic membrane in adults, pull the auricle posterosuperiorly to straighten the canal; in children <3 years, pull posteroinferiorly (canal is more horizontal)
- Skin of the bony canal is thin and adherent — trauma causes exquisite pain
- Cerumen (ear wax): produced by modified apocrine glands in the outer cartilaginous canal; slightly acidic (pH 5–6), providing antibacterial protection
Middle Ear
Tympanic Membrane (TM)
- Four layers: squamous epithelium (outer), radiating fibrous layer, circular fibrous layer, mucosal layer (inner)
- Total area: 70–80 mm²; vibrating surface: ~55 mm²
- Divided into:
- Pars tensa (larger, inferior portion): has all four layers; the taut, vibrating portion
- Pars flaccida (Shrapnell's membrane) (smaller, superior): lacks fibrous layers — prone to retraction pockets and cholesteatoma formation
- Key landmarks visible on otoscopy: light reflex (cone of light, anteroinferior), handle (manubrium) of malleus, lateral process of malleus, umbo (tip of manubrium)
Ossicular Chain
- Malleus: head, neck, manubrium, anterior process, lateral (short) process — handle attached to TM
- Incus: body, short process, long process (lenticular process) — articulates with stapes
- Stapes: head, neck, anterior crus, posterior crus, footplate (oval window)
- Footplate: ~1.41 × 2.99 mm
- Articulates with oval window via annular ligament (syndesmotic joint)
- Ossicular chain amplifies sound by approximately 25–30 dB via mechanical advantage (area ratio TM:oval window ≈ 14:1 + lever action)
Middle Ear Muscles
- Tensor tympani (CN V₃ — trigeminal): inserts on medial surface of manubrium; tenses the TM; attenuates low frequencies
- Stapedius (CN VII — facial): inserts on posterior neck of stapes; pulls stapes posteriorly; dampens loud sounds (acoustic reflex); paralysis → hyperacusis in facial nerve palsy
Prussak Space: bounded by the lateral malleal fold and Shrapnell's membrane — key site of pars flaccida cholesteatoma development
Eustachian Tube
- 17–18 mm at birth → ~35 mm in adults
- At birth: horizontal orientation → adult: 45° incline (pharyngeal orifice 15 mm lower than tympanic orifice)
- Cartilaginous (medial 24 mm) + bony (lateral 11 mm) portions; narrowest at the isthmus (junction)
- Functions: pressure equalization, drainage of middle ear secretions, protection from nasopharyngeal secretions
- Opens with yawning/swallowing (tensor veli palatini muscle, CN V₃)
- Children have a shorter, more horizontal tube → higher risk of otitis media
Inner Ear
- Cochlea: 2.75 turns; contains the organ of Corti (hair cells on the basilar membrane); tonotopic organization — high frequencies at base, low frequencies at apex
- Vestibular apparatus: 3 semicircular canals (superior, posterior, horizontal) + utricle + saccule; detect angular and linear acceleration
- Round window membrane: separates middle ear from scala tympani — allows fluid displacement when stapes pushes oval window
- Blood supply: labyrinthine artery (branch of AICA or basilar artery) — end artery with no collateral circulation → susceptible to ischemia
13. Physiology of Hearing
Sound transmission pathway (air conduction):
Sound waves → EAC → TM → malleus → incus → stapes → oval window → perilymph (scala vestibuli) → basilar membrane vibration → hair cell depolarization → cochlear nerve (CN VIII) → brainstem → auditory cortex
Bone conduction: sound vibrates skull → directly stimulates cochlea (bypasses middle ear)
Acoustic impedance matching: the middle ear overcomes the mismatch between air (low impedance) and cochlear fluid (high impedance) via:
- Area ratio: TM surface >> oval window (~17:1)
- Lever action of ossicles (~1.3:1)
- Combined gain: ~25–30 dB
14. Examination of the Ear
Otoscopy
- Inspect auricle and periauricular area for deformity, swelling, erythema, fistulae
- Palpate tragus (pain → otitis externa), mastoid process (pain → mastoiditis)
- Pull auricle posterosuperiorly (adults) to straighten the canal
- Select appropriate speculum (largest that fits comfortably)
- Insert otoscope gently; assess EAC (skin, discharge, foreign body, cerumen)
- Examine TM: color, translucency, position, light reflex, landmarks, perforation, retraction pockets, middle ear effusion (air-fluid level, bubbles behind TM)
- Pneumatic otoscopy: attach insufflation bulb — insufflation causes normal TM to move laterally (outward); absent movement = middle ear effusion or perforation
Normal TM appearance: pearly grey/translucent, intact, good light reflex at the 5-o'clock position (right ear)
Tuning Fork Tests
Preferred fork: 512 Hz (avoids somatosensory vibration from low frequencies; avoids masking from ambient noise at high frequencies). Strike gently to avoid overtones.
| Test | Method | Normal | Conductive Loss | Sensorineural Loss |
|---|---|---|---|---|
| Weber | Stem on midline skull | Midline (equal bilaterally) | Lateralizes to the poorer (affected) ear | Lateralizes to the better ear |
| Rinne | Mastoid, then at ear canal | AC > BC (Positive Rinne) | BC > AC (Negative Rinne) | AC > BC (Positive Rinne, but both reduced) |
| Bing | Mastoid, occlude canal | Louder with occlusion (Positive) | No change with occlusion (Negative) | Louder with occlusion (Positive) |
| Schwabach | Compare BC duration to examiner | Equal duration | Patient hears longer (Prolonged) | Patient stops sooner (Diminished) |
Rinne interpretation:
- Positive Rinne (AC > BC): normal or sensorineural loss
- Negative Rinne (BC > AC): conductive hearing loss (air-bone gap ≥15–20 dB)
Weber + Rinne combined logic:
- Weber → right + Negative Rinne right → right conductive loss
- Weber → right + Positive Rinne on both sides → left sensorineural loss
15. Classification of Hearing Loss
| Type | Mechanism | Tuning Fork | Audiogram |
|---|---|---|---|
| Conductive | Outer/middle ear pathology blocks sound transmission | Negative Rinne; Weber to affected ear | Air-bone gap >15 dB |
| Sensorineural | Cochlea or CN VIII damage | Positive Rinne (both reduced); Weber to better ear | AC and BC both reduced equally |
| Mixed | Both components | Negative Rinne; air-bone gap + overall threshold elevation | Reduced BC with additional air-bone gap |
Common causes:
- Conductive: cerumen impaction, otitis media with effusion, TM perforation, ossicular chain disruption, otosclerosis
- Sensorineural: presbycusis (age-related, high-frequency), noise-induced, Ménière's disease, ototoxic drugs (aminoglycosides, cisplatin, loop diuretics), acoustic neuroma
16. Otitis Externa (OE)
Definition: Infection/inflammation of the external auditory canal skin.
Epidemiology: Affects 3–10% of the population; peak in summer ("swimmer's ear").
Pathophysiology: Cerumen removal, water (raises canal pH, macerates skin) → breakdown of protective acidic barrier → bacterial overgrowth
Predisposing factors: excessive cleaning, cotton swabs, hearing aids, swimming, eczema/psoriasis, diabetes
Causative organisms:
- Pseudomonas aeruginosa (most common overall)
- Staphylococcus aureus
- Peptostreptococcus, Bacteroides fragilis (mixed)
- Fungal (<10%): Aspergillus niger, Candida spp. — more common in tropical climates, presenting with pruritus > pain
Clinical features:
- Otalgia (severe, exacerbated by traction on the auricle or pressure on the tragus — distinguishes OE from otitis media)
- Pruritus, aural fullness, otorrhea
- Canal erythema and edema (may occlude the lumen)
Treatment:
- Canal debridement/cleaning (remove debris/cerumen)
- Topical antibiotic ± steroid drops: ciprofloxacin/dexamethasone (Ciprodex) — superior to neomycin-polymyxin-hydrocortisone against Pseudomonas
- If canal too swollen for drops: wick/ribbon gauze insertion first
- Oral antibiotics if infection spreads beyond the canal
- Fungal OE: 2% acetic acid drops ± steroid; clotrimazole drops/powder
Necrotizing (Malignant) Otitis Externa:
- Life-threatening osteomyelitis of skull base
- ~90% in immunocompromised (diabetes mellitus, HIV, chemotherapy)
- Organism: Pseudomonas aeruginosa virtually always
- Starts in EAC → spreads to bone → facial nerve palsy, mastoiditis, meningitis, death
- Treatment: prolonged IV antipseudomonal antibiotics + surgical debridement; high mortality
17. Auricular Hematoma
- Blunt trauma (wrestlers, boxers) shears perichondrium from cartilage → blood collects in the subperichondrial space
- Cartilage depends entirely on perichondrium for nutrition → avascular necrosis → fibrosis and neocartilage formation → "cauliflower ear" (permanent deformity)
- Distinguished from ecchymosis by fluctuant swelling with loss of auricular landmarks
- Treatment: prompt incision and drainage + dental roll compression sutures to obliterate the dead space and prevent re-accumulation; follow-up at 24 hours
18. Otitis Media
Acute Otitis Media (AOM)
Definition: Acute onset of middle ear effusion (MEE) + signs/symptoms of infection.
Epidemiology: Most common reason for pediatric antibiotic prescriptions; 93% of children have ≥1 episode by age 7; peak incidence 6–24 months.
Pathophysiology: Eustachian tube dysfunction (abnormal compliance + delayed innervation of tensor veli palatini) → nasopharyngeal organisms colonize middle ear
Etiology:
- Streptococcus pneumoniae (25–40%)
- Haemophilus influenzae (10–30%)
- Moraxella catarrhalis (2–15%)
- Viral URT infection commonly precedes AOM
Diagnostic criteria (ALL required):
- Moderate-to-severe bulging of the TM, OR new-onset otorrhea not due to OE
- Mild bulging + recent-onset ear pain (tugging, rubbing) or intense TM erythema
- Presence of middle ear effusion (MEE) — confirmed by pneumatic otoscopy, tympanometry, or visualization of air-fluid level
Treatment:
- Observation (watchful waiting): appropriate for children ≥2 years with non-severe, unilateral AOM
- Antibiotics: indicated for age <6 months (all), bilateral AOM, severe symptoms (otalgia ≥48h or temp ≥38.5°C), otorrhea, or children with craniofacial abnormalities
- First-line: amoxicillin (80–90 mg/kg/day)
- Penicillin allergy or failure after 48–72h: amoxicillin-clavulanate
- Myringotomy: drainage of middle ear for severe pain or failure to respond
Otitis Media with Effusion (OME / "Glue Ear")
- MEE without acute infection signs; often follows AOM or occurs with Eustachian tube dysfunction
- Leading cause of acquired hearing loss in children
- Most resolve spontaneously within 3 months
- Persistent OME >3 months with hearing loss: tympanostomy tube (grommet) insertion
Chronic Otitis Media (COM)
- Hallmark: persistent TM perforation with intermittent otorrhea
- May be associated with cholesteatoma (keratinizing squamous epithelium in the middle ear — destroys bone, erodes ossicles)
- COM complications:
- Intratemporal: hearing loss (conductive or sensorineural), TM perforation, mastoiditis, facial nerve palsy, labyrinthitis
- Intracranial: meningitis, epidural/subdural abscess, brain abscess, sigmoid sinus thrombosis, otitic hydrocephalus
Mastoiditis (Complication of AOM)
- Signs: AOM on otoscopy + local mastoid inflammation (pain, erythema, tenderness, auricular protrusion/displacement anteroinferiorly)
- Posterior-superior EAC skin edema often present
-
50% of cases in children ≤4 years
- Coalescent mastoiditis: pus collection with bone destruction → surgical emergency
- Treatment: IV antibiotics ± cortical mastoidectomy; CT scan to assess extent
19. Tympanic Membrane Perforation
Acute (traumatic):
- Causes: barotrauma (slap to ear, blast injury, diving, air travel), direct trauma (cotton bud), temporal bone fracture
- Symptoms: sudden ear pain, hearing loss, tinnitus, otorrhea (if infected)
- Examination: otoscopy reveals the perforation (describe location: central vs. marginal, quadrant)
- Treatment: keep dry; most heal spontaneously within 6–8 weeks (90% of AOM perforations heal without intervention); paper patch if slow to close; myringoplasty/tympanoplasty for persistent perforation
Marginal vs. central perforations:
- Central (pars tensa, not reaching the annulus): typically benign, lower risk of cholesteatoma
- Marginal/attic (involves the annulus or pars flaccida): higher risk of cholesteatoma formation
Part III: The Pharynx & Larynx
20. Clinical Anatomy of the Pharynx
The pharynx is a muscular tube extending from the skull base to the level of C6, divided into:
| Region | Boundaries | Key Structures |
|---|---|---|
| Nasopharynx | Skull base → soft palate | Choanae, adenoids, Eustachian tube orifices, fossa of Rosenmüller |
| Oropharynx | Soft palate → epiglottis | Palatine tonsils, posterior tongue (base), soft palate, posterior pharyngeal wall |
| Hypopharynx (Laryngopharynx) | Epiglottis → C6 | Piriform sinuses, posterior pharyngeal wall, postcricoid region |
Waldeyer's ring: the ring of lymphoid tissue encircling the pharyngeal inlet — adenoids (nasopharynx), palatine tonsils (oropharynx), lingual tonsils (tongue base), and smaller lymphoid deposits
Muscles: pharyngeal constrictors (superior, middle, inferior) form the posterior/lateral wall; open during swallowing via coordinated relaxation
21. Clinical Anatomy of the Larynx
Located at C3–C6; the larynx serves as the entrance to the lower airway and is the organ of phonation.
Cartilages:
- Thyroid cartilage: largest; forms the laryngeal prominence (Adam's apple); greater in males due to androgen effect
- Cricoid cartilage: only complete ring; at the level of C6; the narrowest part of the pediatric airway (subglottis)
- Epiglottis: leaf-shaped elastic cartilage; attached to the thyroid cartilage anteriorly; deflects food into the piriform sinuses during swallowing
- Arytenoids (paired): pivot on the cricoid, control vocal fold tension and abduction/adduction
- Corniculate & cuneiform cartilages (minor)
Glottic levels:
- Supraglottis: epiglottis, aryepiglottic folds, false vocal cords (vestibular folds), ventricles
- Glottis: true vocal cords (vocal folds) and anterior/posterior commissures
- Subglottis: below true cords to inferior margin of cricoid
Innervation (both from CN X — Vagus):
- Superior laryngeal nerve (SLN): internal branch (sensory above cords) + external branch (motor to cricothyroid — the only muscle NOT innervated by RLN)
- Recurrent laryngeal nerve (RLN): motor to all intrinsic laryngeal muscles except cricothyroid; sensory below the cords
- Left RLN loops around the arch of the aorta → longer course → more vulnerable to mediastinal pathology
22. Examination of the Pharynx & Larynx
Indirect laryngoscopy (mirror laryngoscopy):
- Patient sits upright, leans forward ("sniffing position"), tongue protruded
- Warm a laryngeal mirror (size 4–6) to prevent fogging
- Grasp tongue with gauze; place mirror against soft palate without touching posterior wall
- Illuminate with frontal reflector
- Ask patient to phonate "eee" — vocal folds adduct (assess movement); breathe — cords abduct
- Assess: epiglottis, aryepiglottic folds, arytenoids, true and false vocal cords, subglottis (partially)
Direct laryngoscopy: under general anaesthesia; definitive airway assessment and therapeutic access
Flexible fiberoptic nasolaryngoscopy: gold standard in office setting; passed transnasally; allows dynamic assessment of vocal fold motion, swallowing, and airway anatomy without significant patient discomfort
23. Acute Pharyngitis & Tonsillitis
Etiology: majority are viral
- Viral: rhinovirus, adenovirus, EBV (infectious mononucleosis), influenza
- Bacterial: Group A β-hemolytic Streptococcus (GABHS / Streptococcus pyogenes) — the only form routinely requiring antibiotics
- Other bacteria: S. pneumoniae, H. influenzae, N. gonorrhoeae
GABHS pharyngitis:
- Peak: late winter/early spring; incubation 2–5 days
- Symptoms: sudden-onset sore throat, odynophagia, fever, chills; headache, abdominal pain
- Signs: tonsillar exudate, anterior cervical lymphadenopathy (tender), scarlet fever rash (diffuse erythematous rough rash, spares perioral area), soft palate petechiae, strawberry tongue, Pastia lines (rash in flexural creases)
McIsaac Score (modified Centor criteria):
| Criterion | Points |
|---|---|
| Temp >38°C | +1 |
| No cough | +1 |
| Tender anterior cervical nodes | +1 |
| Tonsillar swelling/exudate | +1 |
| Age 3–14 years | +1 |
| Age 15–44 years | 0 |
| Age ≥45 years | −1 |
- Score 0–1: antibiotics not indicated
- Score 2–3: rapid antigen test (RADT) then treat if positive
- Score 4–5: treat empirically
Diagnosis: throat culture (gold standard, 96% sensitivity on sheep blood agar); RADT (sensitivity 60–80%, specificity 90%)
Treatment:
- Penicillin V (orally, 10 days) or amoxicillin — drug of choice
- Penicillin allergy: erythromycin or first-generation cephalosporin
- Never use ampicillin/amoxicillin alone if EBV suspected — causes maculopapular rash in ~90% of EBV patients
- Goals: shorten illness, prevent complications (rheumatic fever, peritonsillar abscess, post-streptococcal glomerulonephritis)
24. Peritonsillar Abscess (PTA)
The most common deep neck space infection in adults.
Pathophysiology: Pus accumulates between the tonsil capsule and the superior constrictor muscle.
Clinical features:
- "Hot potato voice" (muffled, plum-in-mouth quality)
- Trismus (spasm of medial pterygoid)
- Uvular deviation toward midline (away from the abscess)
- Unilateral tonsillar bulging + soft palate swelling
- Severe dysphagia, drooling, dehydration
Complications: dehydration, airway obstruction, parapharyngeal abscess (rupture through superior constrictor), bacteremia, mediastinitis (descending necrotizing mediastinitis — life-threatening), aspiration pneumonia, cavernous sinus thrombosis
Treatment:
- Surgical drainage: needle aspiration (at the point of maximal fluctuation in the superior pole) or incision and drainage — both equally effective
- Empiric antibiotics: IV penicillin (or amoxicillin-clavulanate for polymicrobial cover); clindamycin if penicillin allergy
- Rehydration
- Tonsillectomy (quinsy tonsillectomy): performed acutely if recurrent PTA or if airway compromise
Indications for tonsillectomy (Paradise/AAO-HNS criteria):
- ≥7 episodes in 1 year, OR ≥5/year for 2 years, OR ≥3/year for 3 years
- Peritonsillar abscess
- Hypertrophy causing obstructive sleep apnea
- Suspicion of malignancy
- Diphtheria carrier state
25. Hoarseness & Laryngeal Diseases
Hoarseness = abnormal vocal quality; caused by disruption of normal vocal fold vibration.
Acute hoarseness (onset <2 weeks): viral laryngitis, vocal abuse, acute GERD, intubation injury — usually self-limiting
Chronic hoarseness (>2 weeks): must be investigated with laryngoscopy
- Vocal cord nodules ("singer's nodules"): bilateral, at junction of anterior 1/3 and posterior 2/3; from vocal abuse → voice rest + speech therapy
- Vocal cord polyps: unilateral; smoking, vocal abuse; requires laryngoscopy ± microsurgery
- Vocal cord paralysis (see below)
- Malignancy (laryngeal squamous cell carcinoma) — must be excluded
Causes of hoarseness (summary table):
| Category | Examples |
|---|---|
| Acute | Viral laryngitis, vocal hemorrhage, GERD, trauma/intubation, conversion disorder |
| Chronic | Malignancy, nodules, polyps, papillomas, paralysis, chronic laryngitis (smoking/reflux) |
Acute Laryngitis
- Most commonly viral (rhinovirus, parainfluenza, influenza)
- Hoarseness, voice loss, mild sore throat, cough
- Treatment: voice rest, steam inhalation, hydration, avoid irritants; antibiotics not routinely indicated
- Performers: short course of systemic corticosteroids may restore voice rapidly
Vocal Cord Paralysis
- Unilateral RLN palsy: hoarse, breathy voice; risk of aspiration; usually compensated by contralateral cord
- Causes of left RLN palsy: lung cancer (Pancoast), mediastinal lymphadenopathy, aortic aneurysm, cardiac enlargement, thyroid surgery, esophageal cancer
- Causes of right RLN palsy: thyroid/neck surgery, upper lobe right lung tumour, thyroid cancer
- Bilateral RLN palsy: stridor, respiratory distress — may require tracheostomy
- Diagnosis: flexible nasolaryngoscopy; CT neck and chest to identify cause
- Treatment: speech therapy, medialization thyroplasty (type I), or injection laryngoplasty for unilateral palsy
Croup (Laryngotracheobronchitis)
- Most common cause of acute stridor in children (6 months – 5 years)
- Etiology: parainfluenza virus (most common)
- Subglottic edema → "steeple sign" on AP neck X-ray
- "Barking cough," inspiratory or biphasic stridor
- Treatment: dexamethasone (single oral dose — effective even in mild croup); nebulized budesonide or IM dexamethasone if unable to tolerate oral; nebulized racemic epinephrine for severe cases
Epiglottitis
- Bacterial: Haemophilus influenzae type b (now rare post-Hib vaccination); S. pneumoniae, Klebsiella
- Sudden high fever, severe sore throat, drooling, muffled voice, tripod positioning, inspiratory stridor
- "Thumbprint sign" on lateral neck X-ray (swollen epiglottis)
- Airway emergency — do not examine the throat in a restless child (can precipitate complete obstruction)
- Management: secure airway first (intubation in OR or ICU); IV antibiotics (cefotaxime or ceftriaxone)
26. Summary: ENT Examination Instruments & Techniques
| Examination | Instrument | Key Technique |
|---|---|---|
| External ear, EAC, TM | Otoscope (+ pneumatic bulb) | Pull auricle posterosuperiorly (adult); posteroinferiorly (child <3 years) |
| Nasal cavity (anterior) | Nasal speculum + frontal reflector | 2 positions: head neutral, then tilted back 30° |
| Nasopharynx | Nasopharyngeal mirror + frontal reflector | Warm mirror; depress tongue; place behind uvula |
| Oropharynx | Tongue depressor + torch/frontal reflector | Depress tongue; inspect tonsils, posterior wall, uvula |
| Larynx (indirect) | Laryngeal mirror + frontal reflector | Warm mirror; patient phonates "eee" and breathes |
| Larynx (direct) | Rigid laryngoscope (GA) or flexible fiberscope | Flexible scope via nasal route — no anaesthesia needed |
| Hearing | Tuning fork 512 Hz | Weber (midline skull), Rinne (mastoid vs. ear canal), Bing, Schwabach |
Sources: K.J. Lee's Essential Otolaryngology; Textbook of Family Medicine 9e; Rosen's Emergency Medicine; Tintinalli's Emergency Medicine; Cummings Otolaryngology Head and Neck Surgery; Harrison's Principles of Internal Medicine 22e
Acute and chronic diseases of the nose. Trauma of the nose. Epistaxis. Anterior and posterior tamponade. Hematoma and abscess of the nasal septum in detailed explanation medically
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Loading Image
I now have comprehensive material from multiple authoritative sources. Here is the detailed medical guide.
Acute & Chronic Diseases of the Nose, Nasal Trauma, Epistaxis, Tamponade, Hematoma & Abscess of the Nasal Septum
PART 1 — ACUTE DISEASES OF THE NOSE
1.1 Acute Viral Rhinitis (Common Cold)
Definition: Acute self-limiting viral infection of the sinonasal mucosa.
Epidemiology: Extremely common; incidence peaks in autumn/winter. Risk factors include families with multiple children, daycare, healthcare work, stress, underlying allergic rhinitis, and low humidity.
Etiology: Rhinovirus and coronavirus together account for ~50% of cases. Other pathogens include parainfluenza virus, RSV, adenovirus, influenza, enterovirus, and echovirus. Transmission occurs by contact with or aerosolization of infectious secretions.
Pathophysiology: The virus infects ciliated respiratory epithelium of the nasal mucosa → inflammatory cytokine release → vasodilation, mucosal edema, hypersecretion → nasal obstruction and rhinorrhea. Secondary bacterial superinfection occurs in 0.5–2% of cases.
Clinical Features:
- Acute-onset watery rhinorrhea → progressing to mucopurulent discharge over days
- Sneezing, nasal congestion, pharyngitis, cough
- Olfactory loss (hyposmia), headache, malaise, low-grade fever
- Conjunctivitis and mild cervical lymphadenopathy
- Duration: 7–10 days (self-limiting)
Diagnosis: Clinical; serologic, tissue culture, or PCR assays are available but rarely necessary in routine practice.
Treatment:
- Supportive: rest, adequate hydration, steam inhalation
- Topical decongestants (oxymetazoline 0.05%) — short-term use only (≤5 days) to avoid rebound congestion (rhinitis medicamentosa)
- Systemic decongestants (pseudoephedrine)
- Mucolytics, saline nasal irrigations
- Analgesics/antipyretics for fever and myalgia
- Antibiotics: not indicated unless bacterial superinfection develops
1.2 Acute Bacterial Rhinitis
Pathophysiology: Usually secondary to an infected viral rhinitis, often part of bacterial rhinosinusitis when infection spreads to the paranasal sinuses.
Organisms: Streptococcus pneumoniae, Haemophilus influenzae, S. aureus, Bordetella pertussis, Chlamydia, diphtheria bacilli (rare).
Clinical Features: Similar to viral rhinitis but with more prominent yellow-green purulent discharge, longer duration, and more significant systemic symptoms (higher fever, malaise).
Diagnosis: Anterior rhinoscopy reveals mucopurulent secretion in the nasal cavity and possibly the middle meatus. Culture and sensitivity if antibiotic-resistant organisms suspected.
Treatment:
- Antibiotic therapy targeted at likely organisms: amoxicillin-clavulanate is a common first-line choice
- Nasal irrigation, decongestants, mucolytics
1.3 Acute Rhinosinusitis
Definition: Inflammation of the nasal cavity and one or more paranasal sinuses lasting <4 weeks.
Etiology: >90% viral; bacterial superinfection in <2% (most common: S. pneumoniae, H. influenzae, M. catarrhalis).
Symptoms: Purulent nasal discharge, facial pain/pressure/fullness, nasal obstruction, hyposmia/anosmia, postnasal drip, fever, maxillary toothache, halitosis.
Antibiotic prescribing criteria — ALL must be met:
- Symptoms persisting >10 days without improvement, OR
- Severe symptoms: fever ≥39°C + purulent discharge for ≥3–4 days, OR
- "Double-sickening": initial improvement followed by worsening
First-line antibiotic: Amoxicillin-clavulanate (5–7 days for adults); add intranasal corticosteroids, decongestants, saline rinses.
Dangerous complications (rare but critical):
- Orbital cellulitis/abscess (most common complication of ethmoid sinusitis)
- Pott's puffy tumor (osteomyelitis of frontal bone)
- Meningitis, intracranial abscess
- Cavernous sinus thrombosis — presents with proptosis, chemosis, ophthalmoplegia, fever, and headache
PART 2 — CHRONIC DISEASES OF THE NOSE
2.1 Chronic Allergic Rhinitis
Definition: IgE-mediated chronic inflammatory nasal disease triggered by allergen exposure in a sensitized individual.
Classification:
- Seasonal (hay fever): triggered by pollens (grass, trees, ragweed), occurring at specific times of year
- Perennial: triggered year-round by indoor allergens (dust mites, animal dander, mold spores)
Immunopathology — Two-Phase Response:
Phase 1 — Early Phase (begins within 5–15 minutes of allergen exposure):
- Allergen bridges IgE molecules on sensitized mast cells → mast cell degranulation
- Released mediators: histamine, heparin, tryptase, PGD₂, leukotrienes (LTC₄, LTD₄, LTE₄), platelet-activating factor (PAF)
- Effects: sneezing, rhinorrhea, nasal pruritus, conjunctival itching
Phase 2 — Late Phase (begins 4–8 hours later):
- Cytokine-mediated recruitment of eosinophils, neutrophils, basophils
- Perpetuates and amplifies inflammation
- Symptoms: nasal blockage, increased mucus secretion, nasal hyperresponsiveness
Clinical Features:
- Nasal: congestion, watery/clear rhinorrhea, pruritus, sneezing, postnasal drip, hyposmia
- Ocular: pruritus, lacrimation (allergic conjunctivitis)
- Associated: "allergic salute" (rubbing the nose upward with the palm), "allergic shiners" (infraorbital dark circles from venous pooling), nasal crease from repeated rubbing
Examination: Pale, bluish, boggy, edematous inferior turbinates; watery secretions; cobblestoning of posterior pharyngeal wall; may find nasal polyps
Diagnosis:
- Skin prick test or specific serum IgE (RAST) to identify allergens
- Nasal smear: eosinophilia supports allergic etiology
Treatment:
- Allergen avoidance (most important — dust mite covers, air filtration, pet removal)
- Intranasal corticosteroids (mometasone, fluticasone) — most effective single agent; reduce all nasal symptoms; first-line for moderate-severe disease
- Antihistamines (cetirizine, loratadine, fexofenadine) — H₁ blockers; reduce sneezing, rhinorrhea, pruritus; less effective for congestion
- Intranasal antihistamines (azelastine) — rapid onset
- Leukotriene receptor antagonists (montelukast) — adjunctive
- Decongestants (oral pseudoephedrine or short-course intranasal oxymetazoline) — for congestion
- Allergen immunotherapy (subcutaneous or sublingual) — modifies the underlying immune response; consider in patients with inadequate response to pharmacotherapy
2.2 Vasomotor (Non-allergic) Rhinitis
Definition: Chronic nasal symptoms (congestion, rhinorrhea) not caused by allergy, infection, or structural disease; also called non-allergic rhinopathy (NAR).
Pathophysiology: Postulated autonomic imbalance with parasympathetic predominance → vasodilation and mucosal edema. There is no IgE-mediated mechanism, no eosinophilia.
Triggers: Temperature/humidity changes, barometric pressure changes, strong odors (perfume, cooking smells), tobacco smoke, pollutants, alcohol, exercise.
Clinical Features: Predominantly female adults; clear watery rhinorrhea and nasal congestion triggered by non-allergic stimuli; absence of nasal/ocular pruritus (helps differentiate from allergic rhinitis).
Treatment: Avoidance of triggers; intranasal ipratropium bromide (anticholinergic — reduces rhinorrhea); intranasal corticosteroids; saline irrigations.
2.3 Atrophic Rhinitis (Ozena)
Definition: Progressive atrophy of the nasal mucosa and underlying bone (turbinate resorption), causing paradoxical nasal obstruction despite a wide nasal cavity.
Classification:
- Primary atrophic rhinitis: endemic in developing countries (subtropical and temperate climates); unknown etiology but bacterial involvement suspected
- Organisms: Klebsiella ozaenae, S. aureus, Proteus mirabilis, E. coli
- Secondary atrophic rhinitis: follows trauma, excessive turbinate surgery (turbinectomy), irradiation, or granulomatous disease (more common in developed countries, less severe and progressive); related to "empty nose syndrome" after over-resection of turbinate tissue
Histopathology:
- Squamous metaplasia of the respiratory epithelium (loss of cilia)
- Glandular atrophy (loss of mucus secretion)
- Diffuse endarteritis obliterans (obliterative small vessel changes)
Clinical Features:
- Foul-smelling yellow/green crusts (fetor ozenae) — the patient is often unaware due to anosmia
- Anosmia (loss of smell) — from mucosal atrophy over the olfactory region
- Nasal obstruction (paradoxical — due to sensory loss, not mechanical blockage)
- Epistaxis from friable, dry, crusted mucosa
- Atrophy and fibrosis of the turbinates
Examination: Wide nasal cavities, large accumulation of dried green/brown crusts; turbinates reduced or absent.
Treatment:
- Medical: Frequent nasal saline irrigations (dissolve and remove crusts); topical antibiotics (e.g., chloramphenicol ointment); glucose-glycerin 25% nasal drops; oestrogen nasal drops (improve mucosal vascularity); systemic antibiotics for K. ozaenae (tetracyclines, ciprofloxacin)
- Surgical: Young's operation (surgical closure of the nasal vestibule to produce nasal rest and mucosal regeneration) — closed for 6–12 months then reopened; submucosal injection of fat/bone to reduce cavity size
2.4 Nasal Polyps
Definition: Benign, soft, pale, edematous, grape-like outgrowths of the sinonasal mucosa — not true neoplasms but the result of chronic mucosal inflammation.
Origin: Predominantly from the ethmoid sinuses and middle meatus region.
Associations:
- Chronic rhinosinusitis (most common association)
- Samter's triad (aspirin-exacerbated respiratory disease): nasal polyps + aspirin sensitivity + asthma — mediated by cysteinyl leukotrienes
- Cystic fibrosis: nasal polyps in a child <10 years should prompt CF screening
- Allergic fungal rhinosinusitis
- Churg-Strauss syndrome (eosinophilic granulomatosis with polyangiitis)
Pathology: Oedematous stroma with a chronic inflammatory infiltrate rich in eosinophils; goblet cell hyperplasia; no glands (distinguishes from normal mucosa).
Clinical Features:
- Bilateral progressive nasal obstruction
- Anosmia or hyposmia
- Clear or mucoid rhinorrhea, postnasal drip
- Chronic sinusitis symptoms
Examination: Grey/pale/glistening swellings visible in the middle meatus or prolapsing into the nasal cavity. Unlike turbinates, polyps are insensitive to probing (not painful), non-tender, and mobile.
Treatment:
- Intranasal corticosteroids — first-line; reduce polyp size; must be used long-term
- Short course of systemic corticosteroids — for rapid debulking of large polyps (oral prednisolone for 2–3 weeks)
- Endoscopic sinus surgery (ESS/FESS) — for polyps refractory to medical therapy; removes polyps and restores patency of the ostiomeatal complex
- Biologic therapy: dupilumab (anti-IL-4/IL-13 monoclonal antibody) — indicated for severe CRS with nasal polyps uncontrolled by surgery and steroids
2.5 Chronic Rhinosinusitis (CRS)
Definition: Symptoms of rhinosinusitis lasting >12 weeks without complete resolution.
Subtypes: CRS without nasal polyps (CRSsNP) vs. CRS with nasal polyps (CRSwNP) — different inflammatory endotypes.
Symptoms: Same cardinal symptoms as acute rhinosinusitis but prolonged: nasal obstruction, mucopurulent discharge, facial pressure/pain, and olfactory disturbance.
Treatment:
- Intranasal corticosteroids (high-volume budesonide rinses most effective for CRSwNP)
- High-volume saline irrigation (neti pot, sinus rinse bottles)
- Macrolide antibiotics × 3 months for CRSsNP (modest evidence, likely anti-inflammatory effect)
- Short-course systemic steroids for polyp exacerbations
- Endoscopic sinus surgery (ESS) for inadequate response to ≥12 weeks of medical therapy
2.6 Granulomatous Diseases of the Nose (Selected)
Granulomatosis with Polyangiitis (GPA, formerly Wegener's):
- Necrotizing granulomas of the respiratory tract + glomerulonephritis + systemic vasculitis
- Sinonasal involvement (85%): severe nasal crusting, epistaxis, rhinorrhea, secondary sinusitis
- Saddle nose deformity from septal cartilage/bone destruction
- Diagnosis: c-ANCA (anti-PR3 antibodies), biopsy showing necrotizing granulomas
- Treatment: cyclophosphamide + corticosteroids; rituximab for refractory disease
Rhinoscleroma:
- Chronic granulomatous disease due to Klebsiella rhinoscleromatis
- Endemic in Central America, Central Africa, Middle East
- Three stages: (1) Catarrhal — purulent rhinorrhea + crusting; (2) Granulomatous — painless masses, septal destruction; (3) Sclerotic — dense fibrotic scarring, nasal stenosis, anosmia
- Key histopathology: Mikulicz cells (large macrophages with intracellular bacilli) + Russell bodies (swollen plasma cells)
- Treatment: long-term tetracyclines or fluoroquinolones; surgical debridement
PART 3 — TRAUMA OF THE NOSE
3.1 Nasal Bone Fracture
Anatomy relevant to trauma: The nasal pyramid is formed by two nasal bones articulating with the frontal bone, the frontal processes of the maxilla, and the perpendicular plate of the ethmoid. The keystone area — where the nasal bones articulate with the upper lateral cartilages — is critical for structural integrity.

Mechanism:
- Lateral impact (most common): displaces one nasal bone laterally while the contralateral side buckles inward
- Frontal impact: depresses both nasal bones posteriorly, widening the dorsum; more likely to fracture the nasal septum
Clinical Features:
- Pain and tenderness over the nasal bridge
- Epistaxis (may be profuse)
- Swelling and ecchymosis over the nasal dorsum
- Periorbital ecchymosis ("raccoon eyes") without other orbital injury = highly suggestive of nasal fracture
- Nasal bone mobility: grasping the nasal dorsum between thumb and index finger and rocking the pyramid back and forth — virtually diagnostic of fracture
- Nasal obstruction (mucosal swelling, septal deviation, or hematoma)
- Crepitus on palpation
Internal examination (after topical vasoconstrictors and clot clearance):
- Mucosal lacerations
- Septal fracture or acute deviation
- Presence of a septal hematoma — must not be missed
Diagnosis:
- Clinical — the diagnosis is made at the bedside
- Plain radiographs: rarely change management; do not alter treatment decisions for isolated nasal fractures
- Ultrasound: comparable sensitivity/specificity to plain radiography; demonstrates cortical disruption; a useful bedside alternative
- CT scan: reserved for suspected intracranial injury, associated orbital fractures, or complex midfacial trauma (Le Fort fractures); not needed for isolated nasal fractures
Management:
- Immediate: control epistaxis, exclude septal hematoma, analgesics, ice application, elevation
- Closed reduction: indicated when there is significant cosmetic deformity or nasal obstruction
- Optimal timing: after swelling resolves (3–5 days post-injury) but before fibrous union forms (within 10–14 days in adults; 7–10 days in children)
- Performed under local or general anaesthesia; the nasal pyramid is manipulated back into position with a Walsham (internal) and Asch (septal) forceps
- Open rhinoplasty / septorhinoplasty: for complex injuries, delayed presentations, or inadequate closed reduction results — typically deferred 6 months for full healing
PART 4 — EPISTAXIS
4.1 Epidemiology & Blood Supply
Epistaxis is the most common ENT emergency. It has a bimodal age distribution — peaks in children (<10 years) and adults (>50 years). Incidence is higher in winter months in colder climates, due to dry indoor heating that desiccates nasal mucosa. Death from epistaxis is exceedingly rare but it can be distressing and occasionally life-threatening.
Nasal blood supply (see image from previous session):

| Artery | Origin | Area Supplied |
|---|---|---|
| Sphenopalatine | External carotid → maxillary | Turbinates laterally; posterior & inferior septum |
| Anterior ethmoidal | Internal carotid → ophthalmic | Superior nasal mucosa (medial & lateral) |
| Posterior ethmoidal | Internal carotid → ophthalmic | Superior nasal mucosa |
| Superior labial (septal branch) | External carotid → facial | Anterior mucosal septum & anterior lateral mucosa |
| Greater palatine | External carotid → maxillary | Nasal floor |
Kiesselbach's plexus (Little's area): The anastomosis of all four arteries on the anteroinferior nasal septum — the site of ~90% of all nosebleeds.
4.2 Classification
| Feature | Anterior Epistaxis | Posterior Epistaxis |
|---|---|---|
| Frequency | ~90% | ~10% |
| Site | Kiesselbach's plexus (anteroinferior septum) | Sphenopalatine artery (posterior nasal cavity) |
| Laterality | Usually unilateral | Often bilateral; blood flows down both sides |
| Severity | Usually mild to moderate | Often severe; can be life-threatening |
| Population | All ages; more common in children | Predominantly older adults with comorbidities |
| Management setting | Outpatient/ED | Inpatient |
4.3 Etiology
Local causes:
- Nose-picking (digital trauma) — most common in children
- Mucosal dryness and crusting (winter, heated air, nasal oxygen therapy)
- Upper respiratory infection (mucosal vasodilation and fragility)
- Allergic rhinitis, nasal polyps
- Nasal fracture or surgery (postoperative)
- Nasal foreign bodies
- Environmental irritants (cocaine insufflation — destroys septal mucosa and cartilage)
- Neoplasms (nasopharyngeal angiofibroma in adolescent males; inverted papilloma; carcinoma)
- Septal deviation causing turbulent airflow and mucosa desiccation
Systemic causes:
- Anticoagulant/antiplatelet therapy (warfarin, heparin, aspirin, clopidogrel, DOACs) — most significant risk factor for recurrent or severe bleeding
- Bleeding disorders: von Willebrand disease (most common inherited coagulopathy), hemophilia A/B
- Thrombocytopenia (ITP, liver disease, chemotherapy)
- Hereditary Hemorrhagic Telangiectasia (HHT / Osler-Weber-Rendu): autosomal dominant; telangiectasias on the nasal mucosa, lips, tongue; recurrent severe epistaxis; treat with laser photocoagulation or bevacizumab
- Hepatic disease (coagulopathy, thrombocytopenia)
- Vitamin K deficiency, folic acid deficiency
- Hypertension: causal link unproven, but associated with persistent and more severe bleeding
- Chronic vasoconstrictor overuse (rhinitis medicamentosa → mucosal atrophy)
4.4 Clinical Assessment
Immediate assessment:
- Airway, breathing, circulation — is the patient haemodynamically stable?
- Establish IV access and obtain bloods if severe: FBC, coagulation screen (PT/INR/APTT), group & save
- History: timing, frequency, duration, severity, which nostril first, blood in throat (posterior), trauma, medications (anticoagulants, antiplatelets, NSAIDs), bleeding history, comorbidities (hypertension, liver disease, haematologic malignancy), cocaine use
Physical examination:
- Patient leans forward (not backward — prevents swallowing blood and aspiration)
- Patient blows nose to clear clots
- Apply bilateral direct pressure on the cartilaginous (soft, lower) part of the nose for 10–15 minutes continuously — a nose clip is superior to manual pressure alone
- Apply oxymetazoline 0.05% (2 sprays) before pressure — vasoconstriction optimises haemostasis and facilitates examination
- After pressure: anterior rhinoscopy with nasal speculum and headlight — inspect septum (floor parallel to room floor, speculum opened vertically to expose the septum), turbinates, and posterior wall
- Look for the bleeding point — anterior (septal surface, Kiesselbach's area) vs. posterior (blood visible in nasopharynx, bilateral nasal flow)
Investigations (selectively):
- Routine labs not indicated for straightforward anterior epistaxis
- Coagulation studies + FBC for: anticoagulant use, severe/prolonged bleeding, liver disease, haematologic malignancy, suspected bleeding disorder
4.5 Management — Step-by-Step
Step 1: First Aid
- Lean forward, mouth open to breathe, spit blood out
- Pinch soft lower nose for 10–15 minutes without releasing
- Cold compress across the nasal bridge (causes reflex vasoconstriction)
- Oxymetazoline 0.05% or xylometazoline: topical vasoconstrictors — reduce mucosal blood flow, facilitate examination
Step 2: Topical Anaesthesia & Vasoconstriction
- 2% lidocaine + 1:1000 adrenaline (epinephrine) soaked pledgets placed in the nasal cavity for 5 minutes — achieves anaesthesia for cautery and reduces bleeding
- Alternatively: cocaine 4–10% solution (both anaesthetic and vasoconstrictive — used in some ENT units)
Step 3: Chemical Cautery (if bleeding point visible)
- Silver nitrate sticks — applied to the bleeding point under direct vision
- Technique:
- Secure haemostasis first before attempting cautery (cautery during active bleeding is ineffective — blood washes away the silver nitrate)
- Cauterise from the periphery to the centre, and from superior to inferior (blood running down renders inferior applications ineffective)
- Contact time ≤15 seconds per application (prolonged contact → septal damage)
- Cauterise only ONE side of the septum at a time — bilateral cauterisation at the same session risks septal perforation by depriving the cartilage of bilateral blood supply
- Electrocautery (bipolar diathermy): used under endoscopic visualisation in theatre for refractory cases
Step 4: Topical Haemostatic Agents
Used when bleeding cannot be precisely localised or cautery is unsuccessful:
- Absorbable gelatin sponge (Gelfoam) — placed on the bleeding site; absorbs and promotes clot formation
- Oxidised regenerated cellulose (Surgicel) — absorbable; provides a scaffold for clot
- Thrombin + gelatin matrix (Floseal) — topical thrombin activates the coagulation cascade directly at the site; effective even in anticoagulated patients
Step 5: Tranexamic Acid
- Mechanism: antifibrinolytic — inhibits fibrinolysis by blocking plasminogen activation → stabilises the clot
- Routes: topical (500 mg IV solution applied to a nasal pledget or atomised into the naris) or systemic IV
- Evidence: Moderate-quality evidence shows topical TXA significantly reduces bleeding at 10 minutes and re-bleeding at 7–10 days compared to standard care alone; no significant adverse effects
- Particularly superior to anterior nasal packing in patients taking antiplatelet drugs
4.6 Anterior Nasal Tamponade (Packing)
Indicated when pressure, cautery, and topical haemostatics fail.
Principle: Mechanical compression of the bleeding vessels against the nasal bones and cartilage.
Method 1 — Non-absorbable Ribbon Gauze (Vaseline/BIPP gauze)
Technique:
- Achieve adequate topical anaesthesia (2% lidocaine + adrenaline pledgets)
- Using Tilley's nasal dressing forceps (bayonet-shaped):
- Begin by placing the first layer along the floor of the nasal cavity (most important — blocks posterior flow)
- Layer sequentially superiorly in an accordion/"stacking" fashion from floor to roof
- Each loop should fill ~1 cm of height before layering the next
- The pack should fill the entire nasal cavity from the floor to the roof
- Use BIPP (bismuth iodoform paraffin paste) gauze for antiseptic properties, or Vaseline (petroleum jelly) gauze
- Removal at 48–72 hours (leave longer only if necessary)
Method 2 — Merocel Nasal Tampon (Compressed Foam)
- A compressed polyvinyl acetal (PVA) sponge inserted dry into the nasal cavity
- Expands when wetted with saline or blood to fill the cavity and compress bleeding vessels
- Alternatively coated with antibacterial substances
- Easy and rapid to insert; popular in the ED
- Removal at 48 hours: soak with saline first to loosen adherence to mucosa before withdrawing
Method 3 — Inflatable Balloon Tampon (e.g., Rapid Rhino)
- Made of PVA fabric coated with carboxymethylcellulose (a procoagulant hydrocolloidal material)
- Inserted dry, self-lubricating when moistened — additional lubricant is unnecessary and can wash off the procoagulant coating
- Inserted along the floor of the nose and inflated with air (not water — to avoid mucosal pressure necrosis)
- Easy insertion; lower rate of mucosal trauma than gauze packing
Post-packing care:
- Analgesics (packing is uncomfortable)
- Saline spray to moisturise packing
- Prophylactic antibiotics: Routine use is not recommended for anterior packing; antibiotics are only considered for prolonged packing (>72 hours) due to theoretical risk of toxic shock syndrome from S. aureus and sinusitis
- Follow-up in 48–72 hours for pack removal
4.7 Posterior Nasal Tamponade (Packing)
Indication: Posterior epistaxis that persists or recurs despite properly placed anterior packing; bilateral epistaxis; or bleeding that flows immediately into the throat on anterior examination.
Principle: The posterior balloon occludes the choanae (the posterior opening of the nasal cavity into the nasopharynx), preventing blood from flowing posteriorly. The anterior pack then applies pressure to the intervening nasal cavity.
Important: Posterior packing requires hospital admission and monitoring due to:
- Risk of hypoxia (nasopulmonary reflex — obstructed nasal airway reflexly triggers bronchospasm and hypoventilation; also reduces O₂ saturation by ~5%)
- Bradycardia from vagal stimulation
- Toxic shock syndrome if packing left in place >72 hours without antibiotic cover
Method 1 — Double-Balloon Catheter Device (Preferred, e.g., Epistat)
Technique:
- Apply adequate topical anaesthesia to both nasal cavities
- Insert the catheter along the floor of the nose (not upward — the floor is horizontal) through the affected side until the tip is visible in the oropharynx
- Inflate the posterior balloon first with the specified volume of saline (typically 5–10 mL), creating a posterior plug in the nasopharynx
- Pull the catheter anteriorly until resistance is felt — the posterior balloon seats into and occludes the choana
- Inflate the anterior balloon slowly until snug in the nasal cavity (inflate to patient tolerance)
- Apply a folded gauze pad over the nostril and secure the catheter with an umbilical clamp placed over the pad — do not allow the clamp to rest against the alar rim (causes pressure necrosis of the nasal ala)
Method 2 — Foley Catheter (when commercial device unavailable)
Technique:
- Use a 12–14 Fr Foley catheter with a 10–30 mL balloon
- Lubricate generously
- Pass the catheter along the nasal floor until the tip is visible in the oropharynx or until fully inserted
- Inflate the balloon with 5–7 mL of sterile water in the nasopharynx
- Pull anteriorly until the balloon seats at the choana, then add another 5–7 mL if needed (total 7–15 mL)
- Caution: Excessive inflation can cause pressure necrosis of the posterior nasal structures and the palate; do not over-inflate
- Place an anterior pack in the same nostril
- Secure the Foley with an umbilical clamp over a dental roll or gauze pad at the nostril
- A second Foley can be placed on the contralateral side for bilateral posterior haemorrhage
Monitoring requirements (inpatient):
- Continuous pulse oximetry
- Supplemental oxygen (nasal obstruction causes a ~5% drop in SpO₂)
- Cardiac monitoring (vagal bradycardia risk)
- Regular inspection of the alar rim for pressure necrosis
- IV antibiotics considered (prolonged packing)
- Removal at 72 hours (or sooner if pack becomes dislodged)
4.8 Refractory Epistaxis
When bilateral anterior packing and posterior tamponade fail to control bleeding:
-
Endoscopic sphenopalatine artery ligation (ESPAL): endoscopic identification and surgical clipping or cauterisation of the sphenopalatine artery at the sphenopalatine foramen — the definitive surgical procedure for posterior epistaxis; high success rate (>90%)
-
Angiographic embolisation: interventional radiology; catheter-based selective embolisation of the internal maxillary artery or sphenopalatine artery; success rate >80%; preferred in patients with bleeding diatheses or on anticoagulation that cannot be reversed, and when surgical risk is high
-
Anterior ethmoidal artery ligation: for bleeding from the superior nasal vault (ethmoidal epistaxis) — external or endoscopic approach
PART 5 — HEMATOMA OF THE NASAL SEPTUM
5.1 Definition & Pathophysiology
A septal haematoma is a collection of blood between the septal cartilage and its overlying mucoperichondrium (submucoperichondrial space).
Mechanism: Blunt nasal trauma creates a buckling stress on the septum that tears the submucosal blood vessels while the mucosa remains intact. Blood accumulates in the potential space between the perichondrium and the cartilage.
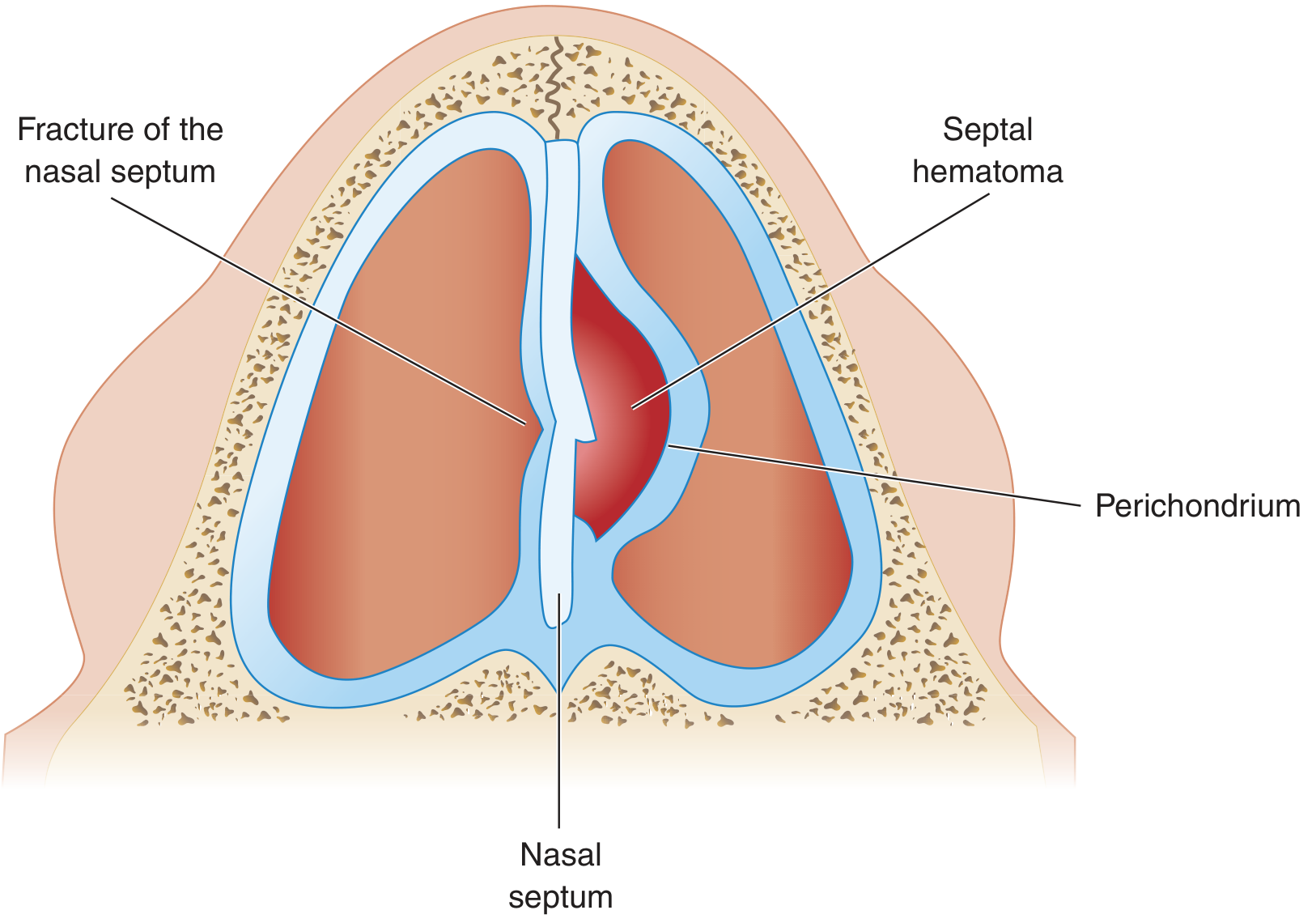
Why this is an emergency: The cartilage of the nasal septum is avascular — it depends entirely on the perichondrium for its blood and nutrient supply. When the haematoma lifts the perichondrium off the cartilage, the cartilage loses its blood supply → avascular (ischaemic) necrosis begins within hours to days.
Epidemiology:
- Occurs in 0.8–1.6% of nasal trauma cases
- More common in males and children (the mucoperichondrium is less adherent to the cartilage in children, making it easier to strip)
- Timing: may form immediately after trauma or more commonly within the first 24–72 hours post-injury
5.2 Clinical Features
| Feature | Description |
|---|---|
| Nasal obstruction | Usually complete bilateral obstruction even though it may begin unilaterally |
| Pain | Severe, often disproportionate to the injury; increases over the first 24 hours |
| Swelling | Widened septum, visible bilaterally on anterior rhinoscopy |
| Appearance | Bilateral bluish/purple-red, round, grape-like, smooth, fluctuant swellings on the septum |
| Response to vasoconstrictors | No reduction in size (distinguishes from turbinate swelling) |
| Fever | Present if infection has supervened (septal abscess forming) |
Examination technique: Insert gloved small fingers into each nostril and palpate the entire septum from front to back for swelling, fluctuation, or widening of the septal space. A normal septum is thin and smooth; a haematoma feels boggy and widened.
Key clinical distinction: A deviated septum appears asymmetric but is firm and bony; a septal haematoma is soft, fluctuant, and bilateral.
5.3 Treatment — Incision & Drainage
A septal haematoma is a surgical emergency. Every hour of delay increases the risk of cartilage necrosis.
Procedure for Incision and Drainage (based on Roberts & Hedges and Tintinalli):
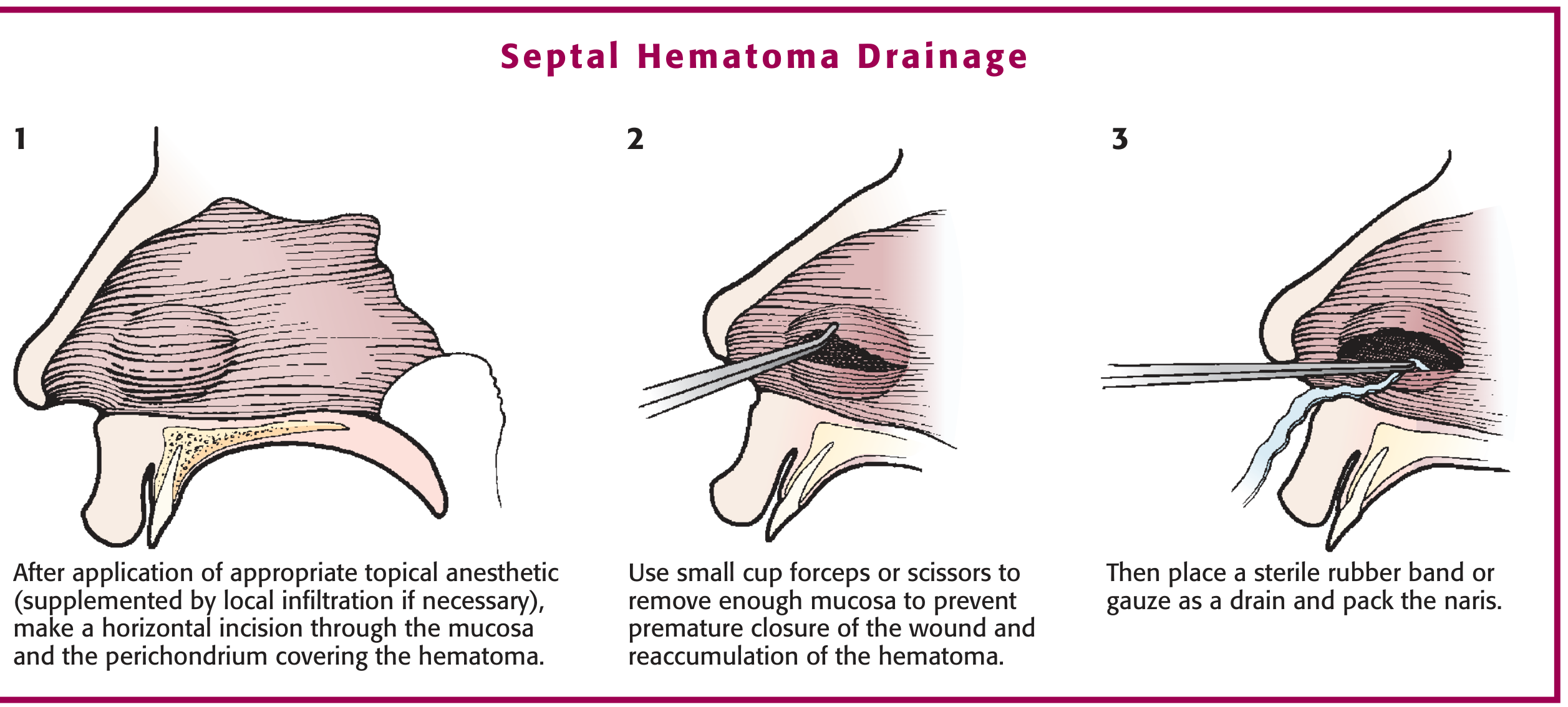
-
Positioning: Patient in the sniffing position; adequate anterior rhinoscopy setup with nasal speculum, light source (headlight or frontal reflector), suction, irrigation, and packing materials
-
Anaesthesia: Cotton pledgets soaked in 1:1 mixture of 4% lidocaine + 1:1000 adrenaline for 5 minutes; supplement with local infiltration (1% lidocaine + 1:100,000 adrenaline) if required
-
Sterile technique: Use sterile instruments; irrigate the nasal cavity to remove debris (sterile technique cannot be fully achieved in the nasal cavity but should be approached as closely as possible)
-
Incision: Make a small horizontal incision through the mucosa and perichondrium at the most dependent part of the haematoma (floor of the nasal cavity side), directly over the swelling — do not incise into the septal cartilage itself
-
Evacuation: Evacuate the clot using Frazier suction or forceps; use small cup forceps to remove enough mucosa to prevent premature closure of the wound and re-accumulation
-
Dead space obliteration: Place a sterile rubber band or gauze drain through the incision, then pack both sides with bilateral anterior nasal packs coated in topical antibiotic ointment (e.g., Mupirocin or Bacitracin) — this applies counter-pressure to both sides of the septum to prevent re-accumulation and keeps the septum midline
-
Post-procedure: Discharge with 24–48-hour follow-up to check for re-accumulation; if re-accumulation occurs, re-drain; some advocate quilting sutures (through-and-through sutures that quilt the mucoperichondrium back to the cartilage) as an alternative or adjunct to packing
-
Antibiotics: Prophylactic antibiotics are recommended given the risk of secondary infection (anti-staphylococcal coverage: amoxicillin-clavulanate or clindamycin)
5.4 Complications of Untreated Septal Haematoma
| Complication | Mechanism |
|---|---|
| Saddle nose deformity | Cartilage necrosis → collapse of the dorsal support of the nose |
| Nasal valve collapse | Loss of structural cartilaginous support → inspiratory collapse |
| Septal perforation | Ischaemia and necrosis of the cartilaginous septum |
| Septal abscess | Stagnant blood is an excellent culture medium → secondary bacterial infection |
| Orbital cellulitis/abscess | Contiguous spread |
| Cavernous sinus thrombosis | Retrograde spread via the ophthalmic and facial veins (valveless system) |
| Meningitis | Direct intracranial extension |
| Intracranial abscess | Epidural or subdural abscess from direct spread |
| Sepsis | Bacteraemia from an infected haematoma |
PART 6 — ABSCESS OF THE NASAL SEPTUM
6.1 Definition & Pathophysiology
A septal abscess is a collection of pus in the submucoperichondrial (or submucoperiosteal) space of the nasal septum. It is one of the most dangerous acute conditions in rhinology.
Pathogenesis: In the vast majority of cases, a septal abscess arises from secondary infection of a septal haematoma. Stagnant blood is an excellent culture medium, and bacteria from the nasal flora invade the haematoma, converting it to pus.
Other causes:
- Nasogastric or nasotracheal tube injury to the septum
- Furunculosis (nasal vestibule infection spreading posteriorly)
- Sinusitis (direct extension from sphenoiditis)
- Dental infection (odontogenic — through adjacent bone)
- Iatrogenic (post-septoplasty)
- Immunodeficiency states
Bacteriology:
- S. aureus — 70% (including MRSA; most common organism)
- Haemophilus influenzae
- Group A beta-haemolytic streptococcus (S. pyogenes)
- Streptococcus pneumoniae
- Klebsiella pneumoniae
- Enterobacteriaceae (Gram-negative)
- Mixed polymicrobial infections (including anaerobes) are common
6.2 Clinical Features
Symptoms:
- Bilateral nasal obstruction (often complete)
- Severe nasal pain — constant, throbbing, disproportionate
- Fever and systemic toxicity (high-grade, chills, rigors)
- Headache
- History of recent nasal trauma or recent upper respiratory infection
Signs:
- Widened, tense, bilateral septal swelling appearing purple/dusky/erythematous
- Fluctuance on palpation (pus under pressure)
- Erythema and oedema of the nasal vestibule and columella
- Tenderness on gentle palpation
- Fever, tachycardia
- In late cases: overlying skin erythema → spreading facial cellulitis
- No reduction in swelling after topical vasoconstrictors
Distinction from septal haematoma:
| Feature | Haematoma | Abscess |
|---|---|---|
| Time post-injury | Hours–3 days | Days–weeks |
| Fever | Absent | Present (often high-grade) |
| Systemic toxicity | Absent | Present |
| Swelling consistency | Fluctuant/boggy | Very tense, fluctuant |
| Skin/vestibule | Normal | Erythematous |
| Aspiration fluid | Blood | Pus |
6.3 Diagnosis
- Clinical — diagnosis is primarily clinical
- Aspiration: needle aspiration with a large-bore needle will yield pus, confirming the diagnosis and providing material for Gram stain and culture
- Blood cultures: if sepsis is suspected
- CT scan of nose and sinuses: maps the extent of involvement, assesses for extension into adjacent spaces (orbital, intracranial), identifies associated sinusitis, and guides surgical planning
- MRI: superior for soft tissue extension and intracranial involvement (gadolinium-enhanced)
6.4 Treatment
A septal abscess is a surgical emergency — delay in drainage leads to catastrophic complications.
1. Operative Incision and Drainage (I&D):
- Performed under local anaesthesia (for cooperative adults) or general anaesthesia (children, uncooperative patients, or extensive disease)
- An incision is made at the base of the septum (along the nasal floor) through both layers of mucoperichondrium
- The abscess cavity is fully opened, thoroughly evacuated, and irrigated with saline ± antiseptic solution
- Necrotic cartilage encountered during drainage is debrided
- A drain (rubber band or small Penrose drain) is placed to prevent premature closure
- Both nasal cavities are packed with antibiotic-coated packs to obliterate the dead space
2. Antibiotic Therapy:
- Start empirical IV antibiotics immediately — do not wait for culture results
- Empirical coverage: anti-staphylococcal (including MRSA cover if risk factors present):
- Standard: IV flucloxacillin (or cloxacillin) ± metronidazole for anaerobes
- MRSA risk or penicillin allergy: IV vancomycin or linezolid
- In immunocompromised: broad-spectrum cover (piperacillin-tazobactam)
- Adjust antibiotics based on culture and sensitivity results
- Duration: typically 10–14 days IV then oral; may need longer if intracranial complications
3. Supportive Care:
- IV fluids if dehydrated
- Analgesia (NSAIDs + opioids for severe pain)
- Antipyretics
- Monitoring of neurological status for early detection of intracranial complications
6.5 Complications
The complications of an untreated or inadequately treated septal abscess range from cosmetic to life-threatening:
Local complications:
- Saddle nose deformity: The most feared cosmetic complication. Cartilage destruction leads to loss of the dorsal nasal support, producing a characteristic concave profile of the nasal dorsum (the saddle). Correction requires rhinoplasty with cartilage grafting (rib or auricular cartilage).
- Nasal valve collapse: Loss of upper/lower lateral cartilage support causes inspiratory collapse of the nasal sidewall
- Septal perforation: Bilateral mucosal necrosis → communication between the two nasal cavities
Spreading infections:
- Facial cellulitis: Contiguous spread through the nasal skin and facial soft tissues
- Orbital cellulitis and abscess: Spread via the cribriform plate or frontal sinus
- Osteomyelitis of adjacent bones (maxilla, ethmoid, frontal)
Intracranial complications (via valveless venous system — ophthalmic veins → cavernous sinus):
- Cavernous sinus thrombosis: Presents with high fever, proptosis, chemosis (conjunctival oedema), ophthalmoplegia, decreased visual acuity, papilloedema, and signs of meningeal irritation — immediately life-threatening
- Meningitis: Headache, neck stiffness, photophobia, fever
- Epidural abscess
- Subdural empyema
- Brain abscess
- Septicaemia/sepsis with multi-organ failure
Key anatomical basis for intracranial spread: The valveless facial veins and anterior facial vein communicate directly with the ophthalmic veins, which drain into the cavernous sinus — a dural venous sinus at the base of the skull housing the internal carotid artery, oculomotor (III), trochlear (IV), ophthalmic and maxillary branches of trigeminal (V₁, V₂), and abducens (VI) nerves. Thrombophlebitis tracking to the cavernous sinus explains why an untreated nasal infection can produce blindness, ophthalmoplegia, meningitis, and death.
Summary Algorithm: Epistaxis Management
EPISTAXIS
│
├─ Assess ABC, haemodynamics, medications
│
├─ Patient leans forward + blows nose to clear clots
│
├─ Oxymetazoline 0.05% spray + pinch soft nose × 10–15 min
│
├─ Bleeding controlled? ──YES──► Observe × 30 min, educate, discharge
│
└─ Still bleeding?
│
├─ Topical lidocaine + adrenaline pledgets × 5 min
│
├─ Identify bleeding point?
│ ├─ YES → Silver nitrate cautery (periphery to centre, one side only)
│ └─ NO → Topical haemostatics (Gelfoam, Surgicel, Floseal) ± Tranexamic acid
│
├─ Still bleeding?
│ └─ ANTERIOR PACKING (Merocel / Rapid Rhino balloon / Vaseline gauze)
│
├─ Still bleeding OR posterior signs?
│ └─ POSTERIOR PACKING (Double balloon / Foley catheter)
│ → ADMIT + oxygen + monitoring
│
└─ Refractory?
└─ ENT consult → Endoscopic sphenopalatine artery ligation
OR angiographic embolisation
Sources: K.J. Lee's Essential Otolaryngology; Tintinalli's Emergency Medicine; Rosen's Emergency Medicine; Roberts & Hedges' Clinical Procedures in Emergency Medicine; Cummings Otolaryngology Head and Neck Surgery; Harrison's Principles of Internal Medicine 22e; Textbook of Family Medicine 9e
Mastering methods of examination of ENT organs. Working with the frontal reflector. Clinical anatomy, physiology, methods of examination of the nose and paranasal sinuses.
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Excellent — I now have rich, detailed content from multiple authoritative ENT textbooks. Here is the complete, detailed study guide.
Examination of ENT Organs — Frontal Reflector, Clinical Anatomy, Physiology & Examination Methods of the Nose and Paranasal Sinuses
SECTION 1: THE FRONTAL REFLECTOR AND ENT EXAMINATION SETUP
1.1 The Frontal Reflector (Head Mirror)
The frontal reflector is the traditional instrument central to all ENT clinical examinations. Before fiberoptic headlights became widely available, it was (and in many institutions still is) the standard light-directing device for examining the ear, nose, throat, and larynx.
Physical description:
- A circular, concave mirror approximately 10 cm in diameter
- Mounted on a headband worn on the examiner's forehead
- Has a central perforation (~1 cm diameter) through which the examiner looks with one eye (binocular vision is maintained)
- The concave surface focuses reflected light into a convergent beam at approximately 30–40 cm focal length
Light source:
- A lamp (60–100 W incandescent or LED) is positioned behind and to the side of the patient (typically at the patient's right shoulder, at head height)
- The examiner angles the mirror to reflect this beam precisely onto the structure being examined
Correct technique for using the frontal reflector:
- The examiner sits directly opposite the patient, at the same height
- The lamp is placed 30–40 cm to the patient's side, slightly behind the patient's head
- The mirror is tilted so the reflected light beam falls on the area of examination
- The examiner's right eye looks through the central hole with both eyes open — this achieves:
- Uniaxial illumination (light enters the cavity along the same axis as the examiner's gaze, eliminating shadows)
- Binocular depth perception
- The head is held still; the mirror is adjusted by tilting at its mounting joint
Why uniaxial illumination matters: When the light source and the line of sight are coaxial, shadows are cast directly away from the examiner — no dark areas are created within the examined cavity. A torch held at an angle creates shadows that obscure pathology.
Uses:
- Anterior and posterior rhinoscopy (nose)
- Otoscopy supplementation (external ear, canal inspection)
- Oropharyngoscopy (tonsils, posterior pharyngeal wall)
- Indirect laryngoscopy (with laryngeal mirror)
- Indirect nasopharyngoscopy (with nasopharyngeal mirror)
1.2 General Setup for ENT Examination
Room requirements:
- Partially darkened room (not fully dark) — reduces ambient light competition with the reflected beam
- Patient seated on an examination chair at the same height as the examiner
- All instruments at arm's reach before beginning
Equipment layout (per examination):
| Examination | Instruments Required |
|---|---|
| Nose (anterior) | Frontal reflector, nasal speculum (Thudichum's or Killian's), light source, tongue depressor |
| Nose (posterior) | Frontal reflector, nasopharyngeal mirror (sizes 2–4), spirit lamp or mirror-warmer, tongue depressor |
| Ear | Otoscope (or auriscope) with pneumatic bulb, frontal reflector, aural specula (3 sizes) |
| Throat | Frontal reflector, tongue depressor, good illumination |
| Larynx | Frontal reflector, laryngeal mirror (sizes 4–6), spirit lamp, gauze, tongue depressor |
Patient positioning:
- Upright, feet flat on the floor, back straight
- Head at the same level as the examiner's
- For nasal examination: head initially in neutral position; then tilted 30° backward for second-position rhinoscopy
SECTION 2: CLINICAL ANATOMY OF THE NOSE
2.1 External Nose — Landmarks and Framework
The external nose projects from the midface and forms the entrance to the nasal cavities. It consists of a bony upper third and a cartilaginous lower two-thirds.
Surface landmarks (from superior to inferior):
| Landmark | Definition |
|---|---|
| Nasion | The nasofrontal suture — the deepest point of the nasal bridge |
| Radix | The root of the nose; the area centred on the nasion extending to the level of the medial canthus |
| Rhinion | The bony-cartilaginous junction along the nasal dorsum (the "tip" of the bony nose) |
| Supratip break | A slight depression between the dorsum and the nasal tip lobule |
| Tip-defining points | The two highest points of the lower lateral cartilages — create the highlights visible on frontal view |
| Infratip break | The lobule-columella junction |
| Columella | The skin and soft tissue bridge between the two nostrils |
| Alar rim | The curved inferior edge of each nostril |
| Nasal sill | The nostril rim between the columella and the alar facial attachment |
Subcutaneous layers of the nose (mnemonic: SFDP):
- S — Superficial fatty layer (attached to dermis)
- F — Fibromuscular layer (nasal SMAS)
- D — Deep fatty layer (contains neurovascular structures)
- P — Periosteum/perichondrium
The optimal surgical dissection plane is between the deep fatty layer and the periosteum — avascular, minimal fibrosis.
Bony framework:
- Two paired nasal bones articulate superiorly with the frontal bone (at the nasion) and laterally with the frontal processes of the maxilla
- Posterior bony support: perpendicular plate of the ethmoid (contributes to the upper bony septum)
Cartilaginous framework:
- Upper lateral cartilages (ULC): fused to the caudal undersurface of the nasal bones; form the middle vault of the nose
- Lower lateral cartilages (LLC) (alar cartilages): C-shaped; have a medial crus (forming the columella), middle crus, and lateral crus (forming the alar rim)
- Quadrangular (septal) cartilage: the anterior cartilaginous septum; articulates superiorly with the perpendicular plate, inferiorly with the vomer
Keystone area: The critical zone where the nasal bones overlap and articulate with the upper lateral cartilages. This is the structural keystone of the nose — disruption here causes "open roof deformity."
Nasal tip support — Major supports:
- Size, shape, and resilience of the medial and lateral crura of the LLC
- Attachment of medial crura to the caudal septal cartilage
- Attachment of the upper lateral cartilage to the lower lateral cartilage (scroll area)
2.2 Internal Nasal Anatomy
The Nasal Septum
The nasal septum divides the nasal cavity into right and left halves. It has three components:
- Perpendicular plate of the ethmoid — posterior-superior bony component; articulates superiorly with the cribriform plate
- Vomer — posterior-inferior bony component; sits in the midline groove of the hard palate
- Quadrangular (septal) cartilage — the anterior cartilaginous component; articulates anteriorly with the nasal spine of the frontal bone and the nasal bones, posteriorly with the perpendicular plate, and inferiorly with the vomer
Mucoperichondrium/Mucoperiosteum: The mucosa of the septum is firmly adherent to the underlying perichondrium/periosteum, forming a single layer. This is the structure elevated during septal surgery (septoplasty) and which, when stripped by trauma, can accumulate blood to form a septal haematoma.
Columella: The anterior free edge of the septum is covered by the medial crura and skin to form the columella.
The Nasal Cavity — Walls and Contents
Each nasal cavity extends from the vestibule (at the nostrils) to the choanae (posterior opening into the nasopharynx).
Roof: Formed by the nasal bones anteriorly, the cribriform plate of the ethmoid centrally (where olfactory nerve filaments pass), and the body of the sphenoid posteriorly. This is the thinnest and most delicate part of the nasal cavity.
Floor: Formed by the palatine process of the maxilla anteriorly and the horizontal plate of the palatine bone posteriorly. It is flat and corresponds to the hard palate.
Medial wall: The nasal septum.
Lateral wall: The most anatomically complex wall — contains the turbinates (conchae), the meati, and the ostia of the paranasal sinuses.
2.3 The Turbinates (Conchae) — Detailed Anatomy
The turbinates are shelves of bone encased in erectile vascular mucosa capable of altering the nasal airway cross-sectional area.
Development: All turbinates originate from the ethmoid bone except the inferior turbinate, which is an independent facial bone derived from the maxilloturbinal.
| Turbinate | Origin | Size | Meatus Beneath It | Drains |
|---|---|---|---|---|
| Inferior | Maxilloturbinal — independent facial bone | Largest | Inferior meatus | Nasolacrimal duct (via Hasner valve, ~1 cm posterior to inferior turbinate face) |
| Middle | Second ethmoturbinal (ethmoid bone) | Medium | Middle meatus | Maxillary, frontal, anterior ethmoid sinuses (via ostiomeatal complex) |
| Superior | Third ethmoturbinal (ethmoid bone) | Small | Superior meatus + sphenoethmoidal recess | Posterior ethmoid and sphenoid sinuses |
| Supreme | Fourth/fifth ethmoturbinals | Rudimentary when present | — | — |
Middle turbinate (MT) — three-dimensional attachment (critical for surgical anatomy):
- Anterior portion: attached in the sagittal plane to the junction of the cribriform plate and the lateral lamella
- Middle portion (basal/ground lamella): attached in the coronal plane to the lamina papyracea — divides anterior from posterior ethmoid sinuses
- Posterior portion (horizontal attachment): attached in the axial plane to the crista ethmoidalis of the palatine bone, just anterior to the sphenopalatine foramen
Variations of the middle turbinate:
- Concha bullosa: pneumatized middle turbinate; present in ~1/3 of the population; can narrow the middle meatus and predispose to sinusitis
- Paradoxical middle turbinate: the lateral surface is convex rather than concave, narrowing the middle meatus
2.4 The Ostiomeatal Complex (OMC)
The ostiomeatal complex is the anatomical key to paranasal sinus drainage. It is a functional unit (not a single structure) in the middle meatus through which the maxillary, frontal, and anterior ethmoid sinuses drain.
Components of the OMC:
| Structure | Description |
|---|---|
| Uncinate process (UP) | Crescent-shaped bony plate from the ethmoid; forms the medial wall of the ethmoidal infundibulum; first lamella of the lateral nasal wall |
| Ethmoidal infundibulum | Three-dimensional space medial to the uncinate process; the final common drainage channel for the maxillary and anterior ethmoid sinuses |
| Hiatus semilunaris | The two-dimensional cleft between the uncinate process and the ethmoid bulla; the entrance to the infundibulum |
| Ethmoid bulla (EB) | The largest and most consistent anterior ethmoid air cell; forms the roof of the hiatus semilunaris; second lamella of the lateral nasal wall |
| Frontal recess | The most anterior and superior portion of the anterior ethmoid sinus; connects the frontal sinus to the middle meatus |
| Maxillary sinus ostium | Opens into the inferior aspect of the ethmoidal infundibulum |
Clinical importance: Mucosal oedema in the OMC (from allergy, viral rhinitis, or anatomical variants) obstructs drainage of multiple sinuses simultaneously → acute or chronic rhinosinusitis. This is the anatomical rationale for functional endoscopic sinus surgery (FESS).
2.5 Blood Supply of the Nasal Cavity
The nasal cavity has a dual blood supply — both internal and external carotid systems:
External carotid artery contributions (via maxillary artery → sphenopalatine artery):
- Sphenopalatine artery (SPA): enters through the sphenopalatine foramen (SPF) located in the superior meatus between the basal lamella of the MT and the superior turbinate
- Posterior septal branch: runs along the sphenoid rostrum; supplies the nasal septum; forms the vascular pedicle of the nasoseptal flap used in skull base reconstruction
- Posterior lateral nasal branch: supplies the middle and inferior turbinates
- Greater palatine artery (descending palatine → incisive foramen): supplies anteroinferior nasal cavity and nasal septum
- Pharyngeal branch of maxillary artery (palatovaginal artery): supplies posterosuperior nasal cavity and nasopharynx
- Superior labial artery (→ alar branch): supplies nasal vestibule and anterior nasal cavity
Internal carotid artery contributions (via ophthalmic artery → ethmoidal arteries):
- Anterior ethmoidal artery (AEA): enters via the anterior ethmoidal foramen; highly variable in its skull base relationship (may lie within the skull base, be dehiscent, or sit up to 4 mm below it in a mesentery — surgical hazard)
- Posterior ethmoidal artery (PEA): enters via the posterior ethmoidal foramen; smaller
Kiesselbach's plexus (Little's area): Confluence of the posterior septal branch of SPA, AEA, greater palatine artery, and alar branch of superior labial artery — all anastomose on the anteroinferior nasal septum. This is the site of ~90% of epistaxis.
Woodruff's plexus: Posterior inferior meatus venous plexus (previously thought arterial); formed by the posterior lateral nasal branch of SPA and the pharyngeal branch of IMA. Site of posterior epistaxis in elderly patients.
Venous drainage (important for understanding intracranial spread of infection):
- Anterior/posterior ethmoid veins → superior ophthalmic vein → cavernous sinus
- Sphenopalatine vein → pterygoid plexus
- Diploic veins of Breschet
- The entire system is valveless → retrograde spread of infection to the cavernous sinus is possible from nasal septal abscess, facial cellulitis, or sinusitis
2.6 Nerve Supply
Sensory innervation:
- Anterior ethmoid nerve (CN V₁ — ophthalmic → nasociliary): external nasal branch innervates the tip and ala; internal branch innervates anterior nasal septum and lateral wall
- Posterior nasal nerves (CN V₂ — maxillary → sphenopalatine ganglion): supply the posterior nasal cavity, turbinates, and septum; the nasopalatine nerve supplies the anterior hard palate
Olfactory innervation:
- Olfactory nerve (CN I): bipolar olfactory receptor neurons (ORNs) lie in the olfactory epithelium of the superior nasal vault (upper third of the septum, superior turbinate, and roof of the olfactory cleft); their unmyelinated axons pass through the cribriform plate (multiple foramina) and synapse in the olfactory bulb
Autonomic innervation:
- Parasympathetic (via CN VII → greater petrosal nerve → pterygopalatine ganglion → nasal branches): vasodilation and glandular secretion — dominant in vasomotor rhinitis
- Sympathetic (via deep petrosal nerve → pterygopalatine ganglion → nasal branches): vasoconstriction, reduction in secretions — mediates nasal decongestion with exercise
SECTION 3: PHYSIOLOGY OF THE NOSE
3.1 Functions of the Nose (Overview)
The nose serves five major physiological functions:
- Air conditioning (warming and humidification)
- Filtration and mucociliary clearance
- Olfaction
- Resonance (vocal quality)
- Immunological defence
3.2 Air Conditioning
Warming: The turbinate mucosa contains a venous erectile tissue plexus (cavernous sinusoids). Blood flow through these sinusoids warms incoming air to approximately 34°C by the time it reaches the nasopharynx, regardless of ambient temperature.
Humidification: The mucosa secretes 1–2 litres of mucus per day. The turbinate surface area is greatly expanded by their scrolled architecture → inspired air achieves ~95% relative humidity by the nasopharynx.
Nasal valve: The narrowest part of the nasal passage — the internal nasal valve — is formed by the angle between the caudal edge of the upper lateral cartilage and the nasal septum (normally 10–15°). This is where maximum airflow velocity occurs (Bernoulli principle — highest velocity at narrowest point creates negative pressure that tends to collapse the valve). The nasal valve accounts for ~50% of total upper airway resistance.
3.3 Nasal Airflow and Resistance
Key physical principles:
-
Bernoulli principle: Airflow velocity is greatest at the narrowest segment (nasal valve). Increased velocity → negative pressure → nasal valve collapse — this is the basis of nasal obstruction in valve incompetence.
-
Poiseuille's law: Resistance to airflow is inversely proportional to the fourth power of the radius. A small decrease in cross-sectional area causes a large increase in airway resistance. This explains why even modest turbinate hypertrophy or slight septal deviation causes disproportionate symptoms.
Airflow patterns:
- During inspiration: the main flow stream travels in the lower and middle airway (the space between the middle meatus and the nasal septum)
- During expiration: airflow is more uniformly distributed across inferior, middle, and olfactory regions; maximum velocity is lower
- Olfactory region receives relatively slow airflow during quiet breathing — important because slow laminar flow allows odorant molecules to diffuse to the olfactory epithelium
3.4 The Nasal Cycle
The nasal cycle is the physiological, cyclical alternation of mucosal congestion (vascular engorgement of the cavernous erectile tissue) between the right and left nasal cavities. It is controlled by the hypothalamus via alternating sympathetic tone.
- Cycle duration: approximately 2–4 hours (range 30 minutes to 6 hours)
- At any point, one side is relatively congested (higher resistance) and the other relatively decongested (lower resistance)
- Total nasal resistance remains approximately constant throughout the cycle
- May be abolished by: exercise, supine position (both sides congest), upper respiratory infection
- The cycle may serve an olfactory purpose: the differential flow rates between sides allow a wider range of odorants to be simultaneously detected (slow flow side better detects low-volatility odorants, fast flow side detects high-volatility odorants)
3.5 Mucociliary Clearance
The mucociliary transport system is the primary mechanical defence of the nasal and paranasal sinus mucosa.
Components:
- Ciliated columnar epithelium (respiratory epithelium): covers the nasal mucosa; each cell bears ~200 cilia (each ~6 μm long); cilia beat in a coordinated metachronal wave
- Mucus blanket: a bilayer system:
- Periciliary fluid (sol layer): low-viscosity layer in which cilia beat freely (~7 μm deep)
- Mucus gel (gel layer): the overlying viscoelastic layer that traps particles and microorganisms; propelled by the tips of cilia during their forward power stroke
Transport rate: Mucus travels at 2–10 mm/hour along the nasal mucosa; faster in the sinuses, directed toward natural ostia.
Direction of transport: All sinus mucus is transported toward the natural ostium of that sinus (even if an accessory ostium is present nearby). This is why surgical drainage must incorporate the natural ostium.
Secretory components:
- Seromucinous glands and goblet cells produce the two-layer mucus blanket
- Periciliary fluid contains: macrophages, neutrophils, basophils, eosinophils, mast cells, B and T lymphocytes, IgA, IgG, IgM, IgE, lysozyme, lactoferrin, and other antimicrobial proteins
Factors impairing mucociliary clearance:
- Viral infection (ciliary damage)
- Bacterial toxins
- Desiccation (dry air)
- Smoking (paralyses cilia)
- Primary ciliary dyskinesia (PCD/Kartagener syndrome — absent or dysfunctional dynein arms)
- Cystic fibrosis (abnormally thick mucus)
3.6 Olfaction
Olfactory epithelium (OE) location: Upper third of the nasal septum, superior turbinate surface, and roof of the olfactory cleft (between the septum and superior turbinate).
Cell types in the OE:
- Olfactory receptor neurons (ORNs): bipolar neurons; the only neurons in the human body that are regularly replaced throughout life; their dendrites extend to the surface and bear non-motile olfactory cilia that project into the overlying mucus
- Supporting (sustentacular) cells: metabolic support, phagocytosis of debris
- Basal cells: stem cells for regeneration of ORNs
- Bowman's glands: in the lamina propria; secrete the olfactory mucus
Olfactory transduction cascade:
- Odorant molecule diffuses into the olfactory mucus layer (~50 μm thick)
- Odorant binds to a specific olfactory receptor (OR) on ORN cilia — there are ~400 functional OR genes in humans (largest gene family)
- OR → Golf protein → adenylyl cyclase III → ↑cAMP → opens cAMP-gated cation channels → Na⁺/Ca²⁺ influx → depolarisation
- Ca²⁺ opens Ca²⁺-activated Cl⁻ channels → further amplification of depolarisation
- Action potential in the ORN axon → passes through cribriform plate foramina → olfactory bulb (glomeruli) → olfactory tract → primary olfactory cortex (piriform cortex, entorhinal cortex) → orbitofrontal cortex (conscious smell perception), limbic system (emotional/memory response)
Orthonasal vs. retronasal olfaction:
- Orthonasal: odorants enter via the nares during sniffing — the classic "smelling"
- Retronasal: during eating and swallowing, retrograde airflow carries volatile odorant molecules from the oropharynx into the nasal cavity via the nasopharynx — responsible for flavour perception (the sensation that food "tastes" different when you have a blocked nose is actually loss of retronasal olfaction)
SECTION 4: METHODS OF EXAMINATION OF THE NOSE
4.1 History Taking (Rhinological History)
A systematic history must precede examination:
Symptoms to enquire about:
- Nasal obstruction: unilateral or bilateral? Constant or intermittent? Which side? Worsened by position (suggests turbinate hypertrophy — worse lying on that side)?
- Rhinorrhoea: character (watery → allergy/CSF leak; mucoid → viral; purulent → bacterial); unilateral or bilateral; anterior or posterior (postnasal drip)?
- Sneezing: paroxysmal sneezing on waking → allergic rhinitis
- Smell disturbance: anosmia, hyposmia, parosmia (distorted smell), phantosmia (smell hallucinations)
- Facial pain/headache: location, timing, relationship to posture
- Nasal bleeding (epistaxis): frequency, severity, triggering factors, which nostril
- Crusting: location, colour, odour
- Associated ocular symptoms: itching, watering → allergic rhinoconjunctivitis
Relevant history: allergies, family history (atopy), recent URTI, medications (decongestants, anticoagulants, antihypertensives — ACE inhibitors cause rhinitis), occupational exposures, smoking and alcohol, trauma
4.2 External Examination
Before any speculum is inserted, inspect and palpate the external nose systematically:
- Inspection from the front: assess symmetry, dorsal width, tip projection, columellar show, alar shape, skin quality
- Inspection from the side (lateral profile): assess dorsal height (hump or saddle), nasolabial angle (normal 90–120° in women, 90–105° in men), tip definition
- Inspection from below (basal view): assess columellar-lobular relationship, nostril shape and symmetry, septal position at the base
- Palpation: assess nasal bone mobility (fracture), tenderness (sinusitis — maxillary and frontal sinus tenderness), skin texture and subcutaneous tissues
- Facial inspection: periorbital swelling (ethmoid/frontal sinusitis complication), orbital proptosis, "allergic shiners" (dark circles from venous stasis), "allergic crease" (transverse crease across the nose from repeated upward rubbing — the allergic salute)
- External nasal valve assessment: ask patient to breathe in through the nose — watch for alar rim collapse (inspiratory alar collapse = external valve insufficiency)
4.3 Anterior Rhinoscopy
Instrument: Nasal speculum — typically Thudichum's speculum (for outpatient use with frontal reflector) or Killian's long-bladed speculum (for deeper visualisation or procedural use).
Purpose: Examination of the anterior nasal cavity — vestibule, anterior septum, inferior and middle turbinates, nasal mucosa.
Technique — Step by Step:
Step 1 — Positioning:
- Patient sits upright, face level with the examiner
- Examiner positions the frontal reflector and directs the light beam into the nostril
- The floor of the nose must be parallel to the floor of the room — if the head tilts, only the superior structures are visible
Step 2 — Speculum insertion:
- Grasp the speculum in the left hand (for right-handed examiners) with the thumb on the screw and fingers controlling the blades
- Gently insert the blades into the nostril in the closed position
- Open the blades in the vertical direction (superiorly and inferiorly) — NOT laterally; lateral opening compresses the septum and obstructs the view
- Do not push the speculum too deeply (painful and may compress the anterior septum)
- The examiner's index finger rests on the patient's nose tip to steady the speculum
Step 3 — First position (head neutral):
- Examine the nasal floor (wide, flat surface)
- Inferior turbinate: colour (should be pink; pale/bluish in allergy; red/purple in vasomotor rhinitis; grey/pale in hypertrophic rhinitis)
- Anterior septal surface: deviation, perforations, ulcers, crusting, Kiesselbach's area (Little's area — anteroinferior septum, spider-web-like vessels)
- Inferior meatus: nasolacrimal duct orifice, any secretions
- Quality of nasal mucosa: colour, moisture, surface texture
Step 4 — Second position (head tilted back 30°):
- Tilting the head back allows the line of sight to pass over the inferior turbinate and visualise higher structures
- Now examine:
- Middle turbinate (its anterior end, colour, size, surface)
- Middle meatus: secretions here indicate sinusitis (purulent discharge tracking from the OMC)
- Any polyps (pale, gelatinous, grape-like masses in the middle meatus)
- Ostiomeatal region
Normal findings on anterior rhinoscopy:
- Pink, moist, smooth mucosa
- Patent airway bilaterally
- Nasal septum in the midline (mild deviations are common and often incidental)
- Inferior turbinate: pale pink, non-obstructing
- No discharge, polyps, masses, or perforation
Abnormal findings to document:
- Septal deviation (direction, severity, contact with lateral wall)
- Turbinate hypertrophy (grade on scale 1–4)
- Character and location of discharge (clear, mucoid, mucopurulent, bloody)
- Presence of polyps, granulations, masses, crusts
- Mucosal colour changes (pale/boggy = allergy; red = infection; pale/dry = atrophic rhinitis)
4.4 Posterior Rhinoscopy (Postnasal Mirror Examination)
Purpose: Examines the nasopharynx, posterior ends of the turbinates, choanae, Eustachian tube orifices, and the roof/posterior wall of the nasopharynx (where adenoids are located in children).
Instrument: Nasopharyngeal mirror (sizes 2–4 for adults; size 1–2 for children), spirit lamp or electric mirror warmer.
Technique — Step by Step:
Step 1 — Mirror preparation:
- Warm the mirror over a spirit lamp or warm water to prevent fogging during examination
- Test the mirror temperature on the back of your own hand to avoid burning the patient
- A thin smear of antifog solution can also be used
Step 2 — Patient preparation:
- Ask the patient to breathe quietly through the mouth (opens the velum and relaxes the pharynx)
- If the patient has a strong gag reflex: apply topical anaesthesia (lidocaine spray) to the posterior pharyngeal wall and soft palate
Step 3 — Mirror insertion:
- Depress the tongue with a tongue depressor held in the left hand — press firmly on the anterior two-thirds of the tongue (not the posterior third, which triggers gagging)
- Introduce the warmed mirror (held like a pencil in the right hand) into the oropharynx with the mirror face directed anterosuperiorly
- Pass the mirror behind the uvula and place it against the posterior surface of the soft palate, angled at approximately 45°
- Do not touch the posterior pharyngeal wall — this triggers the gag reflex
Step 4 — Illumination and examination:
- Direct the frontal reflector beam onto the mirror
- Gently rotate the mirror to systematically survey the nasopharynx
Structures visible on posterior rhinoscopy:
- Choanae (left and right): the posterior nasal openings; should be clear and unobstructed
- Posterior ends of the turbinates: inferior turbinate posterior end is large and rounded; middle turbinate posterior end is seen above
- Vomer: posterior free edge of the nasal septum
- Eustachian tube orifices (Rosenmüller's fossa area): one on each lateral nasopharyngeal wall — a slit-like opening surrounded by the torus tubarius (cartilaginous mound)
- Adenoid tissue (in children): lymphoid tissue on the posterior nasopharyngeal wall — assess size and surface
- Nasopharyngeal roof and walls: assess for masses, ulcers, asymmetry
Clinical significance: Unilateral otitis media with effusion in an adult must always prompt nasopharyngoscopy to exclude nasopharyngeal carcinoma obstructing the Eustachian tube.
4.5 Nasal Endoscopy (Diagnostic Rigid Endoscopy)
Nasal endoscopy represents a major advance in rhinologic diagnosis, providing direct, magnified, illuminated visualisation of structures inaccessible to the nasal speculum.
Equipment:
- Rigid endoscopes: most commonly 4 mm diameter (adult) and 2.7 mm (paediatric)
- Angles: 0° (straight ahead), 30° (most commonly used), 45° and 70° (for angled views)
- Connected to a camera + monitor for documentation and teaching
- Light source: xenon or LED fibre-optic
Patient preparation:
- Patient sits or lies supine
- Nasal cavities sprayed with a topical decongestant (oxymetazoline) to shrink mucosa and improve visualisation
- Topical local anaesthetic (2–4% lidocaine) applied on cotton pledgets
- Antifog solution applied to the lens
Three-pass diagnostic nasal endoscopy (stepwise systematic examination):
Pass 1 — Along the nasal floor:
- Endoscope inserted along the nasal floor medial to the inferior turbinate
- Examine: nasal floor, inferior turbinate (size, surface, colour), inferior meatus
- Advance to the nasopharynx: inspect choanae, posterior ends of turbinates, nasopharyngeal roof, both Eustachian tube orifices, adenoid pad (if present)
Pass 2 — Middle meatus (between inferior and middle turbinate):
- Withdraw slightly and redirect between the middle and inferior turbinates
- Examine: inferior portion of the middle meatus, anterior and posterior fontanelles, accessory maxillary ostia, uncinate process
- Advance medial to the middle turbinate: inspect the sphenoethmoidal recess, superior turbinate and meatus, sphenoid ostium (located medial to the inferior third of the superior turbinate, ~1.5 cm superior to the choana)
Pass 3 — Under the middle turbinate (withdrawal phase):
- As the scope is withdrawn anteriorly, rotate it laterally under the middle turbinate
- Inspect: posterior middle meatus, ethmoid bulla (bulla ethmoidalis), hiatus semilunaris, infundibular entrance
- Final withdrawal: inspect the middle turbinate head, uncinate process, and surrounding mucosa
Key findings to document at endoscopy:
- Mucosal oedema or polyposis
- Purulent or mucopurulent secretions (and their origin — OMC vs. sphenoethmoidal recess)
- Anatomical variants (concha bullosa, paradoxical MT, agger nasi hypertrophy)
- Septal deviations that impair access to the middle meatus
- Vascular assessment (Valsalva manoeuvre — expansion of a mass suggests vascular or intracranial connection)
- Masses or granulations (biopsy assessment — always obtain CT/MRI before biopsy of a sinonasal mass to exclude vascular tumour or intracranial connection)
Advantages over anterior rhinoscopy:
- Magnification and superior illumination
- Access to middle meatus and sphenoethmoidal recess — not visualisable with a speculum
- Ability to assess the natural ostia of sinuses
- Documentation with video/photography
- Gold standard for chronic rhinosinusitis diagnosis
SECTION 5: CLINICAL ANATOMY OF THE PARANASAL SINUSES
5.1 Overview
The paranasal sinuses are air-filled bony cavities that surround the nasal cavity and communicate with it through natural ostia. There are four pairs: maxillary, ethmoid, frontal, and sphenoid.
General functions of the paranasal sinuses:
- Reduce skull weight (pneumatisation)
- Contribute to resonance of the voice
- Extend the area of olfactory mucosa (debated)
- Humidify and warm air
- Act as a crumple zone protecting the brain from facial trauma
5.2 Maxillary Sinus
Anatomy:
- Largest of the paranasal sinuses; pyramidal in shape
- Boundaries:
- Superior wall: orbital floor (contains the infraorbital canal — dehiscent in 14%, causing vulnerability to sinusitis-related neuralgia)
- Inferior wall: alveolar and palatine processes of maxilla — the roots of the upper second premolar and first molar are frequently in close relation (or even protrude into) the sinus floor → odontogenic sinusitis
- Medial wall: lateral nasal wall (medial maxillary wall)
- Lateral wall: zygoma
- Anterior wall: facial surface of maxilla
- Posterior wall: separated from the pterygopalatine and infratemporal fossae
Natural ostium:
- Located in the superior part of the medial wall, opening into the inferior portion of the ethmoidal infundibulum at a 45° angle
- Shape: elliptical; ~3–5 mm in diameter
- Position within the infundibulum: superior third in 10%, middle third in 25%, inferior third in 65%
- Accessory ostia: present in ≥10% of patients — round openings through the anterior or posterior fontanelles (thin membranous areas medial to the uncinate process)
- Critical point: Mucus always flows toward the natural ostium regardless of accessory ostia — this is why surgical antrostomy must incorporate the natural ostium to be effective
Development: First sinus to develop (10 weeks' gestation); radiologically visible at birth; reaches adult size after eruption of permanent dentition (~18 years)
5.3 Ethmoid Sinuses
Anatomy: Not a single cavity but a labyrinth of 6–12 (average 7–8) small air cells divided by thin bony septa; total volume ~15 mL per side.
Anatomical boundaries:
- Lateral wall: lamina papyracea (paper-thin orbital plate of the ethmoid) — separates the ethmoid cells from the orbit; dehiscences are common → orbital complications of sinusitis
- Medial wall: nasal cavity (lateral nasal wall with turbinates)
- Superior wall: fovea ethmoidalis (orbital plate of the frontal bone) = the ethmoid roof, continuous with the anterior cranial fossa
- Medial to the fovea: lateral lamella of the cribriform plate — the thinnest bone in the skull base; fracture here during surgery → CSF leak
Keros classification (depth of olfactory fossa — related to lateral lamella length):
- Type I: 1–3 mm (second most common)
- Type II: 4–7 mm (majority of cases)
- Type III: 8–16 mm (rare; highest risk for CSF leak during sinus surgery)
Anterior ethmoid cells: drain via the OMC (middle meatus)
- Agger nasi cells (most anterior — landmark for frontal recess)
- Ethmoid bulla (most constant and largest anterior cell)
- Haller cells (infraorbital ethmoid cells — project along inferomedial orbit; can obstruct maxillary drainage)
Posterior ethmoid cells: drain into superior meatus
- Onodi cell (sphenoethmoidal cell): posterior ethmoid cell pneumatised posterolaterally over the sphenoid sinus; in ~30% of patients, the optic nerve and/or internal carotid artery project along its superolateral wall — major surgical hazard
5.4 Frontal Sinus
Anatomy:
- Paired sinuses within the frontal bone, separated by an often asymmetric intersinus septum
- Highly variable in size: absent in ~5%, rudimentary in 4%, large and expansive in the remainder
- Absent in Down syndrome and other craniofacial syndromes
Drainage via the frontal recess (not a true duct, but a 3-dimensional hourglass-shaped space):
- Boundaries of the frontal recess:
- Lateral: lamina papyracea
- Medial: middle turbinate
- Anterior: posterior wall of the agger nasi cell (when present)
- Posterior: anterior wall of the ethmoid bulla
- The recess tapers superiorly to the frontal ostium (narrowest point) then widens again within the sinus — hourglass configuration
Anterior ethmoidal artery: crosses the skull base in close relation to the posterior boundary of the frontal recess — at high risk of injury during frontal sinus surgery.
Development: Begins as mucosal invaginations at 4 months' gestation; secondary pneumatisation occurs from 6 months to 2 years of age; not radiologically visible until ~6 years; reaches adult size at ~20 years.
5.5 Sphenoid Sinus
Anatomy:
- The most posterior paranasal sinus; located within the body of the sphenoid bone
- Separated from its partner by an often asymmetric intersinus septum (the septum frequently deviates and attaches to the ICA — surgical hazard if the surgeon follows the septum expecting midline)
Pneumatisation classification:
- Conchal: absent pneumatisation (sinus filled with cancellous bone); rare
- Presellar: pneumatisation anterior to a vertical line through the tuberculum sellae (second most common)
- Sellar: pneumatisation to or behind the anterior wall of the sella turcica (most common)
- Postsellar: extensive pneumatisation posterior to the sella
Natural ostium:
- Opens into the sphenoethmoidal recess (medial to the superior turbinate, lateral to the nasal septum)
- Located approximately halfway to two-thirds up the anterior wall of the sinus
- Distance from anterior nasal spine: 6.2–8.0 cm (average 7.1 cm) at 30–34° angulation from the floor
- Always lower than the level of the maxillary sinus roof — following this endoscopic landmark avoids skull base penetration
Anatomical relationships (critically important — "sinus of danger"):
- Superior: pituitary gland (in the sella turcica), optic chiasm
- Lateral walls (depending on pneumatisation): internal carotid artery (ICA), cavernous sinus, cranial nerves III, IV, V₁, V₂, VI
- Superior and lateral wall: optic nerve (may be dehiscent in 4%)
- Posterior wall: basilar artery
- Floor: nasopharynx
Development: Secondary pneumatisation at 6–7 years; absent in Down syndrome.
SECTION 6: EXAMINATION METHODS FOR THE PARANASAL SINUSES
6.1 Sinus History Specific Points
- Facial pain/pressure: location and sinus correspondence (forehead → frontal; cheek/upper teeth → maxillary; behind/between the eyes → ethmoid; top of the head/vertex or retro-orbital → sphenoid)
- Postnasal drip, mucopurulent discharge from the nose
- Relationship to posture (maxillary sinus pain may worsen on bending forward)
- History of dental problems (odontogenic sinusitis)
- Seasonal pattern (allergic sinusitis)
- Response to previous treatments
6.2 Physical Examination of the Sinuses
Inspection:
- Inspect the skin overlying each sinus for erythema, swelling (oedema over the maxillary or frontal sinuses in acute sinusitis)
- Periorbital oedema (medial orbital wall — complication of ethmoid sinusitis)
- Facial asymmetry (suggests unilateral chronic disease or tumour)
Palpation and percussion:
- Maxillary sinuses: palpate and percuss over the anterior face of the maxilla (eminence below the zygoma, lateral to the nose) — tenderness suggests maxillary sinusitis
- Frontal sinuses: press the thumbs upward under the supraorbital ridge (against the floor of the frontal sinus) — tenderness suggests frontal sinusitis; do NOT press over the centre of the forehead (thin bone, unreliable)
- Ethmoid sinuses: tender to pressure medial to the medial canthus of the eye
- Sphenoid sinuses: inaccessible to direct palpation; symptoms of sphenoid sinusitis include vertex/retro-orbital headache, visual disturbance, and cranial nerve involvement
6.3 Transillumination
Technique:
- Performed in a completely darkened room
- A strong, focused light source (transilluminator or pen torch pressed firmly against the tissue) is used
Maxillary sinus transillumination:
- Place the light against the hard palate (intraoral) or against the skin overlying the infraorbital rim
- Observe the cheek for a dull red glow
- Compare both sides
Frontal sinus transillumination:
- Place the light against the medial supraorbital margin (floor of the frontal sinus)
- Observe for red glow above the eye
Interpretation:
- Normally aerated sinus: diffuse reddish glow
- Opacified sinus (mucus, pus, polyp, tumour): dull or absent glow
Limitations: Low sensitivity and specificity — not recommended as the sole diagnostic test. Results are unreliable in patients with thick facial tissues, very small sinuses, or asymmetric sinus pneumatisation. CT scanning is far superior.
6.4 Imaging of the Paranasal Sinuses
Plain X-rays (Sinus Series)
Historically used but now largely replaced by CT. Views include:
- Waters' view (occipito-mental): best for maxillary sinuses; patient faces the X-ray film with chin raised
- Caldwell's view (postero-anterior): best for frontal and ethmoid sinuses
- Lateral view: shows all sinuses in profile
Findings: opacification (total or partial), air-fluid levels (acute sinusitis), mucosal thickening (>6 mm significant in maxillary sinus)
Limitations: Cannot visualise the OMC or individual ethmoid cells; cannot differentiate tumour from secretions; significant false-positive and false-negative rates.
CT Scan (Standard of Care)
High-resolution CT of the paranasal sinuses is the gold standard imaging investigation for:
- Chronic rhinosinusitis
- Pre-surgical planning for endoscopic sinus surgery (ESS)
- Sinonasal tumours (bony involvement)
- Orbital and intracranial complications of sinusitis
Protocol: Coronal and axial slices (1–3 mm cuts); no IV contrast needed for chronic sinusitis (contrast added for complications or tumours)
CT findings in sinusitis: Mucosal thickening, opacification, air-fluid levels, ostiomeatal complex obstruction, bony erosion or remodelling
Imaging checklist before ESS (critical anatomical variants to identify):
- Skull base height and asymmetry (Keros classification)
- Lamina papyracea dehiscence
- Dehiscent optic nerve or ICA in sphenoid sinus
- Onodi cells
- Agger nasi cells (frontal recess anatomy)
- Concha bullosa
- Haller cells
- Accessory maxillary ostia
Important: CT should be performed at least 4 weeks after initiating medical therapy for the most recent sinusitis episode and at least 2 weeks after an acute URTI — to avoid false opacification from acute mucosal oedema confounding the surgical planning.
MRI
Advantages over CT:
- Superior soft tissue contrast — differentiates tumour from secretions (tumour enhances; mucus does not)
- Better assessment of intracranial extension, dural involvement, perineural spread
- Characterises mucus viscosity (T1/T2 signal intensity varies with water content)
- No ionising radiation
Indications in rhinology:
- Suspected sinonasal malignancy
- Intracranial complications of sinusitis
- Opacification adjacent to a skull base dehiscence (to exclude meningoencephalocele before biopsy)
- Allergic fungal rhinosinusitis characterisation
6.5 Special Tests for Nasal Function
Nasal airflow assessment:
| Test | Principle | Advantages | Disadvantages |
|---|---|---|---|
| Nasal inspiratory peak flow (NIPF) | Measures peak flow during maximal forced nasal inspiration | Portable, easy | Variable, influenced by lower airway, nasal valve collapse |
| Rhinomanometry | Pressure/flow curves at baseline and after decongestion; calculates nasal airway resistance | Functional test; both sides simultaneously | Time-consuming, cannot localise obstruction |
| Acoustic rhinometry | Sound waves reflected back from nasal cavity → cross-sectional area profile (rhinogram) | Fast; localises area of obstruction | Technically challenging; cannot measure beyond narrow apertures |
| Computational fluid dynamics (CFD) | Mathematical modelling of nasal airflow from CT/MRI data | Non-intrusive; simulates surgical outcomes | Time-consuming; unclear clinical correlation |
Smell testing (olfactometry):
- Sniffin' Sticks test: standardised odour sticks assess threshold, discrimination, and identification (TDI score)
- University of Pennsylvania Smell Identification Test (UPSIT): scratch-and-sniff booklet; reliable and validated
- Electro-olfactography: records electrical potentials from olfactory epithelium (research setting)
- Olfactory evoked potentials: records CNS response to olfactory stimulation
Summary Table: ENT Examination Instruments for the Nose
| Examination | Instrument | Key Points |
|---|---|---|
| External | Inspection + palpation | Lighting, patient cooperation, systematic |
| Anterior rhinoscopy | Thudichum/Killian speculum + frontal reflector | Open blades vertically; 2 head positions |
| Posterior rhinoscopy | Nasopharyngeal mirror + frontal reflector | Warm mirror; do not touch posterior wall |
| Nasal endoscopy | 0°/30° rigid endoscope + decongestant | 3 passes; systematic; decongestion first |
| Sinus palpation | Fingers (thumbs for frontal) | Compare bilaterally; percussion |
| Transillumination | Transilluminator | Dark room; poor specificity — not for sole diagnosis |
| Imaging — sinusitis | CT (coronal + axial, 1–3 mm) | Wait 4 weeks post-treatment |
| Imaging — tumour/complication | MRI (gadolinium) | Superior soft tissue, intracranial extension |
| Airflow | Rhinomanometry, acoustic rhinometry | Pre/post-decongestion comparison |
| Smell | Sniffin' Sticks, UPSIT | Threshold + identification scores |
Sources: K.J. Lee's Essential Otolaryngology; Cummings Otolaryngology Head and Neck Surgery; Tintinalli's Emergency Medicine; Textbook of Family Medicine 9e; Harrison's Principles of Internal Medicine 22e
This is a shared conversation. Sign in to Orris to start your own chat.