Hirsutism
Reading File
Finding Sources
Searching PubMed
hirsutism management
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Loading Image
Here is a comprehensive overview of hirsutism drawn from multiple authoritative textbooks.
Hirsutism
Definition
Hirsutism is the excessive growth of terminal (coarse, pigmented) hair in women in an androgen-dependent, male-pattern distribution. It is distinct from hypertrichosis, which is a diffuse increase in fine, vellus hair that is androgen-independent (seen with hypothyroidism, anorexia nervosa, or medications such as minoxidil, diazoxide, phenytoin). — Fitzpatrick's Dermatology, p. 1585; Textbook of Family Medicine 9e, p. 1053
Epidemiology
- Affects approximately 5–15% of women of reproductive age
- Prevalence varies widely by ethnicity: 38% in Greece, 21% in Australia, 10.8% in Iran, 2% in Thailand
- Asian women have lower hair follicle density and are less likely to manifest hirsutism even with androgen excess
- Heaviest hair growth occurs in younger reproductive-age women — Fitzpatrick's Dermatology, p. 1585
Pathogenesis
Hair follicles cycle through three phases: anagen (growth), catagen (involution), and telogen (rest). In androgen-sensitive areas, androgens convert vellus hair to coarse terminal hair via:
- DHT (dihydrotestosterone) is the most potent androgen — produced from testosterone by 5α-reductase within the hair follicle
- DHT binds nuclear androgen receptors → activates genes responsible for vellus-to-terminal hair conversion
- Androgens originate from the ovaries (LH-driven) and adrenal glands (ACTH-driven)
- In idiopathic hirsutism, local end-organ hypersensitivity occurs despite normal circulating androgens — Fitzpatrick's Dermatology, p. 1585–1586
Causes
| Etiology | Key Features |
|---|---|
| Idiopathic hirsutism (most common) | Regular ovulation, normal or mildly elevated androgens, increased 5α-reductase activity |
| PCOS (most common secondary cause) | Chronic anovulation, insulin resistance, LH excess, raised testosterone/LH:FSH ratio; >50% of PCOS women have hirsutism |
| Non-classic CAH (21-hydroxylase deficiency) | Autosomal recessive; cortisol synthesis impaired → androgen precursors diverted |
| Androgen-secreting tumors | Abrupt/rapid onset; virilization prominent; ovarian or adrenal |
| Ovarian hyperthecosis / HAIR-AN syndrome | Hyperandrogenism + insulin resistance + acanthosis nigricans; 1–5% of hyperandrogenic women |
| SAHA syndrome | Seborrhea + Acne + Hirsutism + Androgenetic Alopecia |
| Hyperprolactinemia | Prolactin stimulates adrenal DHEA-S |
| Exogenous androgens | Anabolic steroids, danazol |
| Cushing syndrome | Cortisol excess with androgen co-secretion |
— Fitzpatrick's Dermatology, Table 90-1; Textbook of Family Medicine 9e, p. 1054
Assessment: Modified Ferriman-Gallwey (mFG) Score
Nine body areas are each scored 0–4 (0 = no hair, 4 = frankly virile):
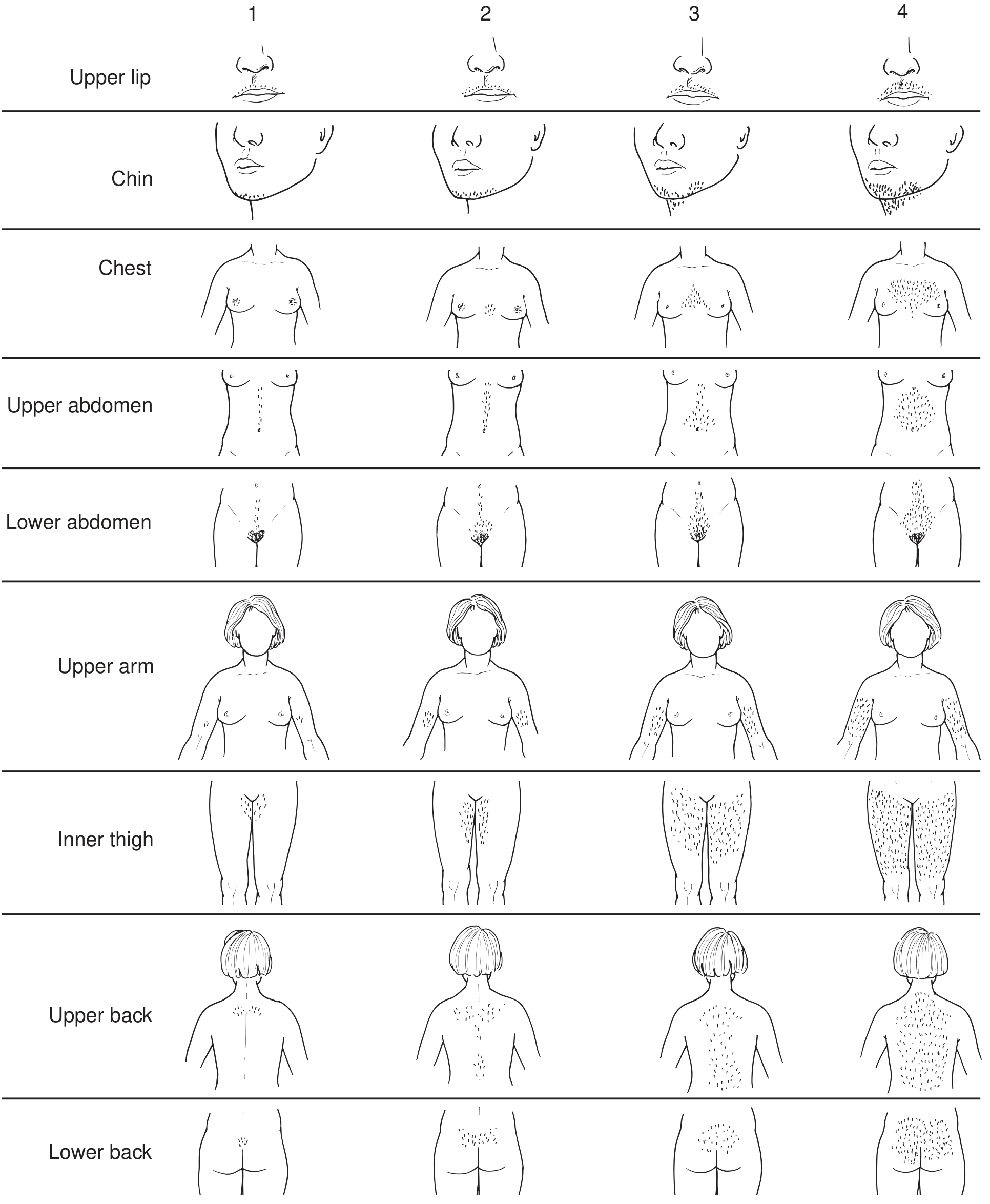
The nine areas: upper lip, chin, chest, upper abdomen, lower abdomen, upper arm, inner thigh, upper back, lower back
- Score ≤ 8: Normal
- Score 8–14: Mild hirsutism
- Score ≥ 15: Moderate–severe hirsutism
Ethnic and racial variation must be considered (e.g., Mediterranean women score higher at baseline). — Fitzpatrick's Dermatology, p. 1586; Textbook of Family Medicine 9e, p. 1053
Clinical Evaluation
History: onset (gradual vs. abrupt), menstrual irregularities, ethnic background, medications, weight changes, signs of virilization
Exam: upper lip > thighs > lower abdomen > upper back (most common distribution); also assess for acne, acanthosis nigricans, androgenic alopecia, clitoromegaly, voice deepening, breast atrophy
Red flags suggesting tumor: rapid onset, marked virilization
Laboratory workup:
- Total and bioavailable testosterone
- DHEA-S (adrenal androgen marker)
- 17-OH progesterone (morning, follicular phase) → screens for non-classic CAH
- Prolactin, LH/FSH ratio
- Fasting glucose/insulin if PCOS suspected
- 24-hour urine free cortisol or dexamethasone suppression test if Cushing's suspected
- Imaging (USS, CT, MRI): if testosterone >150 ng/dL or DHEA-S >700 μg/dL → suspect tumor
Women with mild-moderate hirsutism and regular cycles → likely idiopathic; hormone testing not mandatory. Hormone testing required in moderate-severe hirsutism or with menstrual irregularity. — Fitzpatrick's Dermatology, p. 1586; Harrison's 22e, p. 3185
Treatment
1. Non-pharmacologic (for all patients)
| Method | Notes |
|---|---|
| Bleaching | Cosmetic only |
| Shaving / chemical depilatory | Surface removal; shaving does NOT increase growth rate |
| Waxing | Temporary removal |
| Electrolysis | Effective permanent removal; skill-dependent |
| Laser / Intense Pulsed Light (IPL) | Best for large areas of pigmented terminal hair; photothermolysis of melanin → permanent reduction in many patients |
2. Pharmacologic
Response is not evident for 4–6 months; full effect may require 9–12 months due to hair cycle length.
| Drug | Mechanism | Dose | Notes |
|---|---|---|---|
| Combined OCP (1st line) | Suppresses LH → ↓ ovarian androgens; ↑ SHBG | Daily pill | Prefer low-androgenic progestins (norgestimate, drospirenone); ~20% improvement in hirsutism; 50% improvement in acne |
| Spironolactone | Competitive androgen receptor blocker + ↓ androgen synthesis + ↑ SHBG | 100–200 mg/day | Most studied antiandrogen; monitor K⁺/BP; avoid in pregnancy (feminizes male fetus); combine with OCP |
| Cyproterone acetate | Competes with DHT at androgen receptor; ↓ 5α-reductase | 50–100 mg days 1–15 + EE 50 μg days 5–26 | Not available in US; widely used in Europe/Canada; risk of liver dysfunction |
| Finasteride | 5α-reductase type 2 inhibitor | 2.5–5 mg/day | Limited efficacy (type 1 predominates in PSU); teratogenic |
| Dutasteride | Inhibits both 5α-reductase types 1 & 2 | Limited data | Potentially more efficacious than finasteride |
| Flutamide / Bicalutamide | Non-steroidal androgen receptor blocker | 62.5–250 mg BD / 25 mg/day | Risk of hepatotoxicity (flutamide); combine with OCP |
| GnRH agonists (e.g., leuprolide) | Suppress gonadotropins → ↓ ovarian androgens | 7.5 mg IM monthly + transdermal E₂ | Used for severe/refractory cases; add-back estrogen prevents osteoporosis |
| Glucocorticoids (e.g., dexamethasone 0.2–0.5 mg nightly) | Suppress adrenal androgen secretion | Low dose | Indicated specifically for CAH; not recommended as monotherapy for other causes |
| Eflornithine cream (13.9%) | Inhibits ornithine decarboxylase in hair follicle | Topical, twice daily | Slows facial hair growth; does not remove existing hair; effect reverses on stopping |
— Harrison's 22e, pp. 3184–3185; Fitzpatrick's Dermatology, pp. 1590–1591; Goodman & Gilman's, pp. 312–322
Associated Conditions
- PCOS: elevated cardiovascular and metabolic risk; insulin sensitizers (metformin) help treat underlying cause alongside hirsutism
- Non-classic CAH: OCPs first-line; add glucocorticoids if inadequate response
- HAIR-AN: insulin sensitizers are a cornerstone
- SAHA syndrome: present in ~18% of PCOS patients; associated with greater insulin resistance
Recent Evidence
A 2025 Cochrane systematic review (PMID 41288141) specifically evaluates cyproterone acetate for hirsutism, and a 2024 meta-analysis (PMID 38163998) updates evidence on inositol in PCOS-related hyperandrogenism. These do not fundamentally contradict current practice but refine the evidence base for specific agents.
Key clinical pearl: Treatment choice must be individualized. OCPs are first-line for most women not seeking conception. Antiandrogens (spironolactone, cyproterone) are added when OCP response is inadequate. All pharmacologic therapy requires 6–12 months for full assessment of benefit and should be combined with non-pharmacologic approaches.
This is a shared conversation. Sign in to Orris to start your own chat.