Pathology of lymphoma
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Searching Images
Reed-Sternberg cells Hodgkin lymphoma histology
Reading File
Reading File
Reading File
Searching Images
Burkitt lymphoma starry sky histology
Reading File
I now have comprehensive content from Robbins Pathology on lymphoma. Let me compile a thorough, well-organized answer covering the full pathology of lymphoma.
Pathology of Lymphoma
Lymphoma is a clonal malignant proliferation of lymphoid cells. It is broadly divided into Hodgkin lymphoma (HL) and Non-Hodgkin lymphoma (NHL), with further subdivision based on cell of origin, morphology, immunophenotype, and molecular genetics.
1. General Principles
Diagnosis Requires Tissue
Lymphoid neoplasia can be suspected clinically, but histologic examination of lymph nodes or other involved tissues is mandatory for diagnosis. Analysis of lineage-specific protein (marker) expression and genetic alterations complements morphologic study. Lymph node specimens must be sent fresh without formalin for optimal flow cytometry.
- Robbins Pathologic Basis of Disease, p. 555
Clonality and Antigen Receptors
Antigen receptor gene rearrangement generally precedes malignant transformation; all daughter cells of the transformed progenitor share the same antigen receptor gene configuration, producing identical surface immunoglobulins or T-cell receptors — the basis of clonality testing.
Molecular Pathogenesis
- AID (activation-induced cytidine deaminase): expressed in germinal center B cells to drive class switching and somatic hypermutation. Misregulation causes translocations (e.g., MYC::IGH in Burkitt) and BCL6 mutations.
- V(D)J recombinase errors: can juxtapose proto-oncogenes to antigen receptor gene regulatory elements, underling rearrangements in both B- and T-cell neoplasms.
- Viruses: HTLV-1 (adult T-cell leukemia/lymphoma); EBV (30–40% of HL, Burkitt lymphoma — 90% endemic, 20% sporadic); HHV8 (primary effusion lymphoma).
- Chronic inflammation: H. pylori → gastric MALT lymphoma; gluten-sensitive enteropathy → intestinal T-cell lymphoma.
- Robbins, pp. 553–555
2. WHO Classification Overview
Hodgkin Lymphoma (HL)
| Subtype | Notes |
|---|---|
| Classic HL – Nodular Sclerosis | Most common (40–70%); mediastinal, supraclavicular nodes |
| Classic HL – Mixed Cellularity | |
| Classic HL – Lymphocyte-Rich | |
| Classic HL – Lymphocyte-Depleted | |
| Nodular Lymphocyte-Predominant HL (NLPHL) | Distinct biology; "popcorn cells" |
Non-Hodgkin Lymphoma (NHL) — Mature B-Cell Neoplasms (selection)
- Chronic lymphocytic leukemia / Small lymphocytic lymphoma (CLL/SLL)
- Follicular lymphoma (FL)
- Mantle cell lymphoma (MCL)
- Marginal zone lymphoma (MZL) / MALT
- Diffuse large B-cell lymphoma (DLBCL)
- Burkitt lymphoma (BL)
- Lymphoplasmacytic lymphoma
- Plasma cell myeloma
NHL — Mature T-Cell & NK-Cell Neoplasms (selection)
- Peripheral T-cell lymphoma (NOS)
- Anaplastic large cell lymphoma (ALCL), ALK+/ALK−
- Adult T-cell leukemia/lymphoma (HTLV-1)
- Extranodal NK/T-cell lymphoma
- Hepatosplenic T-cell lymphoma
Precursor Neoplasms
- T/B-lymphoblastic lymphoma/leukemia (ALL)
Robbins, p. 555; Cummings Otolaryngology, Table 204.5
3. Hodgkin Lymphoma
Histopathology — The Hallmark Cell
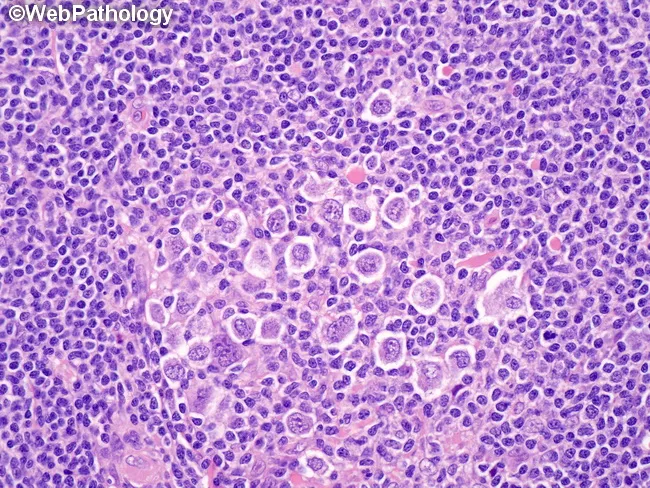
Reed-Sternberg (RS) cells are the diagnostic hallmark. They are:
- Large (up to ~100 µm), with abundant slightly basophilic cytoplasm
- Binucleated or multinucleated with prominent, large eosinophilic nucleoli ("owl-eye" appearance)
- Mononuclear Hodgkin cells (variants) with similar nuclear features
- RS cells are the minority of cells; the bulk of the lesion is a reactive inflammatory background infiltrate of lymphocytes, eosinophils, plasma cells, and histiocytes
Microdissection + single-cell PCR demonstrated monoclonal immunoglobulin gene rearrangements in RS cells, confirming B-cell lineage — the reason "Hodgkin lymphoma" replaced "Hodgkin disease."
Classic Hodgkin lymphoma: large RS cells with prominent "owl-eye" nucleoli amid a polymorphic inflammatory background
Immunophenotype
| Marker | Classic HL | NLPHL |
|---|---|---|
| CD30 | ✅ Positive | Negative |
| CD15 | ✅ Positive | Negative |
| PAX5 | Weak positive | Strong positive |
| CD20 | Usually negative | ✅ Positive |
| CD45 | Negative | Positive |
| EMA | Negative | Positive |
Classic HL Subtypes
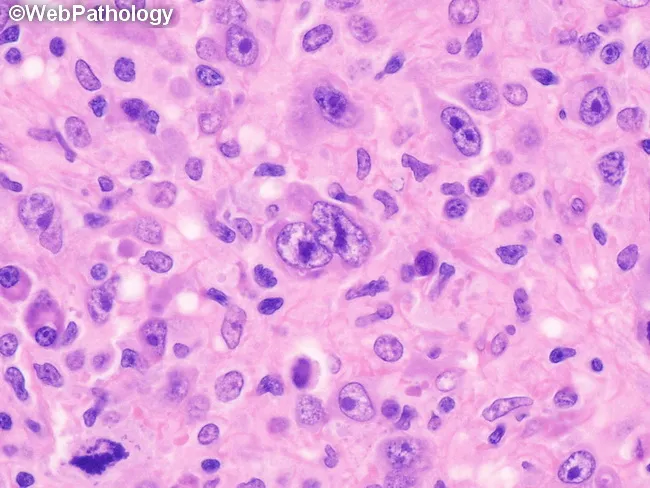
1. Nodular Sclerosis (most common — 70% adolescents)
- Broad collagen bands divide lymph node into cellular nodules
- RS cells in lacunar spaces ("lacunar cells" — artifact of formalin fixation)
- Predilection: lower cervical, supraclavicular, mediastinal nodes
Nodular sclerosis CHL: lacunar RS cells within fibrous bands, nodular architecture
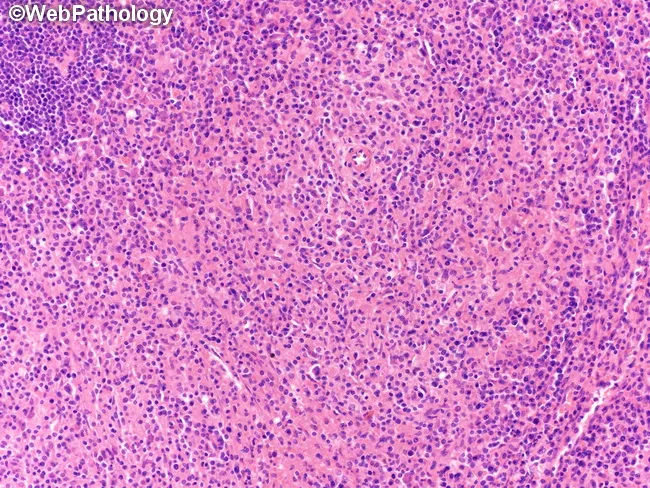
2. Mixed Cellularity
- No fibrotic bands; polymorphic background (histiocytes, eosinophils, plasma cells)
- Classic RS cells are more easily identified
- Often EBV-positive
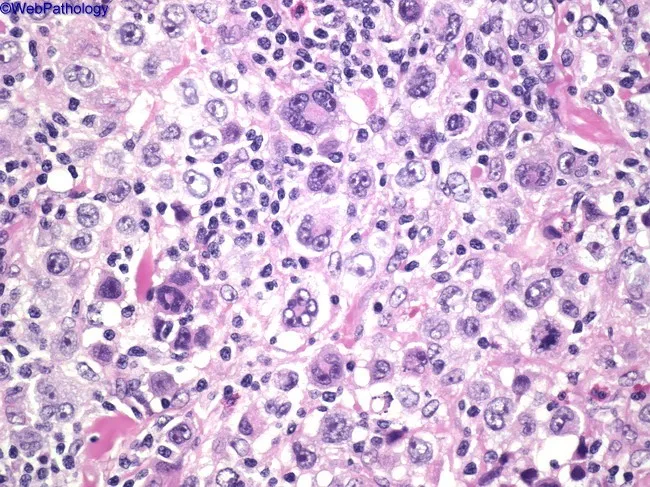
3. Lymphocyte-Depleted
- Marked lymphocyte depletion; sheets of RS and pleomorphic cells
- Sparse inflammatory background; may have fibrosis
- Most aggressive; often advanced stage at presentation
4. Lymphocyte-Rich
- Abundant small reactive lymphocytes; classic RS cells present but infrequent
Staging (Ann Arbor)
| Stage | Definition |
|---|---|
| I | Single lymph node region |
| II | ≥2 regions, same side of diaphragm |
| III | Regions both sides of diaphragm |
| IV | Diffuse/disseminated extranodal involvement |
"B" symptoms: fever >38°C, drenching night sweats, ≥10% weight loss in 6 months.
- Cummings Otolaryngology, p. 3850; K.J. Lee's Essential Otolaryngology, p. 1050
4. Non-Hodgkin Lymphoma — Key Entities
CLL / Small Lymphocytic Lymphoma
- Most common leukemia of adults (Western world); median age 60; 2:1 male predominance
- Pathogenesis: deletions 13q14.3 (loss of miR-15a/miR-16-1 → BCL2 overexpression), del 11q, del 17p, trisomy 12q; NOTCH1 mutations (10–18%, poor prognosis)
- Morphology: small, round, mature-appearing lymphocytes with clumped chromatin; "proliferation centers" (pale-staining aggregates with larger prolymphocytes)
- Immunophenotype: CD5+, CD19+, CD20 (dim), CD23+, surface Ig (dim); CD5 co-expression distinguishes CLL from other B-cell lymphomas
- Genetics: unmutated IgVH genes = more aggressive course
- Robbins, p. 555
Follicular Lymphoma
- Second most common NHL in the West; derived from germinal center B cells
- Hallmark translocation: t(14;18) → BCL2::IGH → BCL2 overexpression → impaired apoptosis in follicular center cells
- Morphology: nodular/follicular architecture; centrocytes (small cleaved) and centroblasts (large noncleaved) in neoplastic follicles
- Immunophenotype: CD10+, CD19+, CD20+, BCL2+, BCL6+, CD5−, cyclin D1−
- Indolent but typically incurable with conventional therapy; ~30–40% transform to DLBCL (Richter-like transformation)
Mantle Cell Lymphoma
- Hallmark translocation: t(11;14) → cyclin D1 (CCND1)::IGH → cyclin D1 overexpression → cell cycle dysregulation
- Morphology: monomorphic small-medium lymphocytes with irregular nuclear contours; diffuse or vaguely nodular pattern; no RS cells, no proliferation centers
- Immunophenotype: CD5+, CD19+, CD20+, cyclin D1+, CD23−, CD10−
- Aggressive; median survival ~3–5 years without modern therapy
MALT Lymphoma (Extranodal Marginal Zone)
- Low-grade B-cell lymphoma arising in mucosa-associated lymphoid tissue
- Driven by chronic antigenic stimulation: H. pylori (gastric MALT); Sjögren syndrome (salivary gland); Hashimoto thyroiditis (thyroid)
- H. pylori eradication alone can induce remission in early gastric MALT lymphoma
- Morphology: heterogeneous small lymphocytes, monocytoid B cells, plasma cells; lymphoepithelial lesions (tumor cells invading glandular epithelium)
- Key translocations: t(11;18), t(14;18) (BCL10::IGH), t(3;14)
Diffuse Large B-Cell Lymphoma (DLBCL)
- Most common NHL (~30–40% of all NHL)
- Morphology: large cells, vesicular nuclei, prominent nucleoli, diffuse growth pattern; no residual follicular architecture
- Molecular subgroups (cell of origin):
- Germinal Center B-cell (GCB): BCL2 rearrangement, EZH2 mutations; better prognosis
- Activated B-cell (ABC): NF-κB pathway activation, MYD88 mutations; worse prognosis
- Immunophenotype: CD19+, CD20+, CD79a+; BCL6 and/or MUM1 for subclassification
- Aggressive but potentially curable with R-CHOP
Burkitt Lymphoma
- Extremely aggressive; one of the fastest growing human tumors (Ki-67 ~100%)
- Three clinical forms: Endemic (jaw mass, African children; >90% EBV+), Sporadic (abdominal, young adults; 20% EBV+), Immunodeficiency-associated (HIV; ~40% EBV+)
- Hallmark translocation: t(8;14) → MYC::IGH (or t(2;8), t(8;22) variants) → MYC overexpression → dysregulated cell cycle, increased proliferation
- Morphology: medium-sized monomorphic cells, round nuclei, multiple small nucleoli, scant cytoplasm; brisk mitotic activity + apoptosis → phagocytosis by macrophages = "starry sky" pattern (pale macrophages against dark blue lymphoid background)
- Immunophenotype: CD20+, CD10+, BCL6+, BCL2−, surface IgM+; Ki-67 ~100%
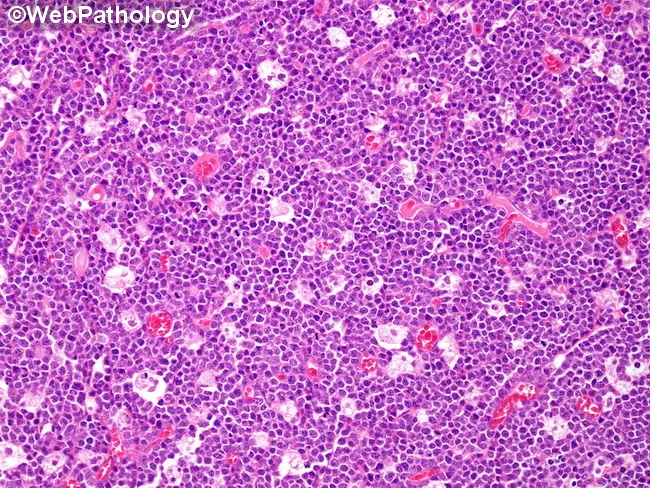
Burkitt lymphoma: dense sheets of medium B cells with tingible-body macrophages producing the pathognomonic "starry sky" pattern
Anaplastic Large Cell Lymphoma (ALCL)
- T-cell lymphoma; two forms: ALK+ (younger patients, better prognosis) and ALK− (older, worse)
- ALK+ driven by t(2;5) → NPM1::ALK fusion → constitutive ALK tyrosine kinase signaling
- Morphology: large pleomorphic cells with eccentric horseshoe-shaped nuclei ("hallmark cells"), sometimes sinusoidal spread
- Immunophenotype: CD30+++ (strong), ALK+ (in ALK+ form), CD3 often lost, EMA+
Adult T-Cell Leukemia/Lymphoma (ATLL)
- Caused by HTLV-1 retrovirus (endemic Japan, Caribbean)
- Morphology: "flower cells" — T cells with multilobated, clover-leaf nuclei
- Highly aggressive; often presents with hypercalcemia, lytic bone lesions, skin involvement
5. Key Immunohistochemistry Panel
| Marker | CD5 | CD10 | CD20 | CD23 | CD30 | CD15 | Cyclin D1 | BCL2 | BCL6 | Ki-67 |
|---|---|---|---|---|---|---|---|---|---|---|
| CLL/SLL | + | − | dim | + | − | − | − | + | − | Low |
| FL | − | + | + | +/− | − | − | − | + | + | Low–mod |
| MCL | + | − | + | − | − | − | + | + | − | Variable |
| DLBCL | − | +/− | + | − | − | − | − | +/− | +/− | High |
| Burkitt | − | + | + | − | − | − | − | − | + | ~100% |
| Classic HL | − | − | − | − | + | + | − | − | − | Variable |
| ALCL | − | − | − | − | +++ | − | − | − | − | High |
6. Clinical Features Shared Across Lymphomas
- Painless lymphadenopathy (most common presentation)
- B symptoms: fever, night sweats, ≥10% weight loss
- Mediastinal mass: particularly NS-CHL, T-lymphoblastic lymphoma, DLBCL
- Extranodal disease: stomach (MALT), CNS (DLBCL), skin (mycosis fungoides, ALCL), liver/spleen, bone marrow
- Staging: CT chest/abdomen/pelvis; whole-body PET-CT; bone marrow biopsy selectively
- Prognosis: HL has ~95% overall survival; DLBCL curable in ~60% with R-CHOP; Burkitt lymphoma >90% cure rates in pediatric stages I–III with intensive chemotherapy
Sources: Robbins & Cotran Pathologic Basis of Disease (9780443264528); Cummings Otolaryngology Head and Neck Surgery (9780323612173); K.J. Lee's Essential Otolaryngology (9781260122237)
This is a shared conversation. Sign in to Orris to start your own chat.