Explain in detail neuroplasticity
Reading File
search_medical_text
neuroplasticity mechanisms synaptic plasticity brain remodeling
search_medical_text
neuroplasticity clinical applications rehabilitation learning memory
GradeResponse
GradeResponse
The retrieved content provides a useful anchor from Harrison's. I'll now synthesize a comprehensive answer drawing on that and my own extensive knowledge of neuroscience.
Neuroplasticity
Definition
Neuroplasticity (also called brain plasticity or neural plasticity) refers to the brain's lifelong ability to reorganize its structure, function, and connections in response to experience, learning, injury, or disease. It encompasses changes at every level — from individual synapses and neurons to large-scale cortical maps and networks.
As Harrison's Principles of Internal Medicine (p. 13832) describes, this plasticity operates at the microscale (cellular/molecular), mesoscale (between cortical and subcortical areas), and macroscale (whole-brain networks), and the brain remains highly plastic regardless of age and even in the face of injury or degenerative processes.
Types of Neuroplasticity
1. Synaptic Plasticity
Changes in the strength or efficacy of existing synaptic connections. This is the most studied and well-understood form.
| Subtype | Description |
|---|---|
| Long-Term Potentiation (LTP) | Persistent strengthening of synapses following repeated, high-frequency stimulation. Molecular basis of learning and memory. |
| Long-Term Depression (LTD) | Persistent weakening of synapses following low-frequency stimulation. Allows for "forgetting" and fine-tuning of circuits. |
| Short-Term Plasticity | Transient changes (milliseconds to minutes) in synaptic strength — includes facilitation and synaptic depression. |
| Homeostatic Plasticity | Global scaling of synaptic strength up or down to keep neuronal activity within a functional range (synaptic scaling). |
2. Structural Plasticity
Physical changes in the architecture of neurons and networks.
- Dendritic remodeling — growth, retraction, or reshaping of dendritic spines and branches
- Axonal sprouting — formation of new axonal branches and connections
- Synaptogenesis — formation of entirely new synapses
- Synaptic pruning — elimination of weak or unused synapses (critical during development and adolescence)
- Changes in myelination — experience can alter the thickness and extent of myelin sheaths, affecting conduction velocity
3. Neurogenesis
The generation of new neurons. In adults, neurogenesis is largely restricted to two regions:
- Hippocampal dentate gyrus (important for memory and mood regulation)
- Olfactory bulb (via the subventricular zone)
Adult neurogenesis is modulated by exercise, stress, antidepressants, and enriched environments.
4. Cortical Remapping
Large-scale reorganization of cortical representations (maps).
- Use-dependent expansion: Regions controlling heavily-used body parts or skills physically expand (e.g., larger somatosensory representation of the fingers in musicians).
- Cross-modal plasticity: Sensory-deprived cortex is recruited by other modalities (e.g., the visual cortex processes touch and sound in the blind).
- Injury-induced remapping: After focal brain damage, adjacent or homologous areas in the opposite hemisphere can take over lost functions.
Cellular and Molecular Mechanisms
Hebbian Plasticity — "Cells that fire together, wire together"
Donald Hebb's 1949 postulate: when a presynaptic neuron repeatedly activates a postsynaptic neuron, the synaptic connection between them is strengthened. This is the conceptual foundation for LTP.
Molecular Cascade of LTP (NMDA-dependent)
- Glutamate binds AMPA receptors → depolarization of postsynaptic membrane
- Depolarization relieves Mg²⁺ block of NMDA receptors
- Ca²⁺ influx through NMDA receptors activates CaMKII (Ca²⁺/calmodulin-dependent protein kinase II)
- CaMKII phosphorylates existing AMPA receptors (increasing conductance) and triggers insertion of more AMPA receptors into the synapse
- For long-lasting LTP: activation of CREB (cAMP response element-binding protein) → transcription of plasticity genes → new protein synthesis → structural synaptic changes
Key Molecular Players
| Molecule | Role |
|---|---|
| NMDA receptors | "Coincidence detectors" — require both pre- and postsynaptic activity |
| AMPA receptors | Primary mediators of fast excitatory transmission; trafficked in/out during plasticity |
| CaMKII | Master kinase for LTP induction |
| BDNF (Brain-Derived Neurotrophic Factor) | Promotes synapse formation, LTP, and neuronal survival |
| CREB | Transcription factor essential for long-term memory consolidation |
| Arc/Arg3.1 | Immediate-early gene that regulates AMPA receptor trafficking |
| mTOR pathway | Translational control of local synaptic protein synthesis |
Developmental vs. Adult Plasticity
Critical Periods
During early development, the brain is especially sensitive to environmental input. These "critical periods" are windows during which specific neural circuits require proper stimulation to develop normally.
- Example: Binocular vision — if one eye is occluded in early childhood (amblyopia), the deprived eye's cortical representation is permanently diminished. This same occlusion in adulthood causes no lasting change.
- Critical periods are governed by the balance of excitatory/inhibitory (E/I) inputs and the maturation of parvalbumin-positive interneurons and perineuronal nets (extracellular matrix structures that stabilize circuits).
Adult Plasticity
While less dramatic than developmental plasticity, the adult brain retains robust capacity for change:
- Skill learning (motor, cognitive)
- Memory formation and consolidation
- Recovery from injury
- Adaptation to sensory loss
Plasticity in Learning and Memory
Memory consolidation involves two main processes:
- Synaptic consolidation — occurs within hours; depends on NMDA receptor activation, protein synthesis, and AMPA receptor insertion at synapses
- Systems consolidation — occurs over weeks to years; memories initially dependent on the hippocampus gradually become stored in neocortical networks
The hippocampus is essential for declarative memory (episodic and semantic) formation, while the amygdala is critical for emotional memory, the cerebellum for procedural and motor learning, and the basal ganglia for habit formation.
Plasticity After Brain Injury
Following stroke, traumatic brain injury (TBI), or other focal lesions, the brain undergoes several adaptive changes:
| Phase | Time Frame | Mechanism |
|---|---|---|
| Acute | Hours–days | Diaschisis resolution, unmasking of latent connections, ionic changes |
| Subacute | Days–weeks | Synaptic sprouting, LTP-like potentiation in perilesional cortex |
| Chronic | Weeks–months/years | Cortical remapping, compensatory recruitment of contralesional hemisphere, neurogenesis |
Harrison's (p. 13832) explicitly notes that "there is now growing evidence to support neurologic restorative efforts for both 'static' (e.g., stroke) and progressive neurologic disorders," underscoring the clinical relevance of harnessing plasticity.
Clinical and Therapeutic Applications
Rehabilitation Medicine
- Constraint-Induced Movement Therapy (CIMT): Restraining the unaffected limb forces use of the paretic limb, driving cortical remapping in stroke patients.
- Repetitive task practice and motor imagery both drive use-dependent plasticity.
- Robotic-assisted rehabilitation and virtual reality provide high-dose, repetitive training to engage plasticity mechanisms.
Neurostimulation
- Transcranial Magnetic Stimulation (TMS): Focal magnetic pulses can induce LTP- or LTD-like changes and modulate cortical excitability.
- Transcranial Direct Current Stimulation (tDCS): Weak electrical currents shift membrane potential, priming neurons for activity-dependent plasticity.
- Deep Brain Stimulation (DBS): Used in Parkinson's disease and depression; mechanisms may include plasticity-mediated circuit normalization.
Pharmacology
Several drugs modulate neuroplasticity:
- SSRIs/SNRIs: Increase BDNF expression and promote neurogenesis in the hippocampus (contributing to antidepressant effects)
- Ketamine (NMDA antagonist): Paradoxically produces rapid synaptic potentiation via AMPA receptor upregulation — the basis for its rapid antidepressant action
- D-cycloserine: NMDA partial agonist used to enhance fear extinction in PTSD treatment
- Ampakines: Positive modulators of AMPA receptors under investigation for cognitive enhancement
Sensory Substitution and Prosthetics
- Brain-computer interfaces (BCIs) leverage plasticity to allow the brain to learn to control external devices or interpret novel sensory streams.
- Cochlear implants and retinal prostheses work because the auditory/visual cortex remains plastic enough to decode artificial electrical signals.
Factors That Promote or Impair Neuroplasticity
Promoting Factors
| Factor | Effect |
|---|---|
| Aerobic exercise | Increases BDNF, promotes hippocampal neurogenesis, upregulates LTP |
| Sleep | Critical for memory consolidation; synaptic homeostasis hypothesis (Tononi) — synapses downscaled during sleep to preserve signal-to-noise |
| Enriched environments | Increase dendritic complexity, spine density, and synapse number |
| Novelty and challenge | Drive dopaminergic signaling, which gates plasticity |
| Social interaction | Modulates oxytocin and serotonin pathways that support plasticity |
| Meditation/mindfulness | Associated with structural changes in prefrontal cortex and insula |
Impairing Factors
| Factor | Effect |
|---|---|
| Chronic stress | Elevated cortisol causes dendritic atrophy in hippocampus, impairs LTP |
| Sleep deprivation | Disrupts consolidation and synaptic homeostasis |
| Aging | Reduced NMDA receptor function, decreased BDNF, impaired LTP induction |
| Alcohol/substance abuse | Dysregulates glutamate/GABA balance, impairs hippocampal neurogenesis |
| Neuroinflammation | Microglial activation and cytokines (TNF-α, IL-1β) suppress LTP |
Maladaptive Neuroplasticity
Not all plasticity is beneficial. The same mechanisms that enable learning and recovery can also encode pathological states:
- Chronic pain: Central sensitization in the dorsal horn and somatosensory cortex — LTP-like changes amplify pain signals (wind-up phenomenon)
- Addiction: Drug-induced dopamine surges drive pathologically strong LTP in the nucleus accumbens and prefrontal-limbic circuits, encoding compulsive drug-seeking
- PTSD: Hyperconsolidation of fear memories via amygdala LTP; impaired hippocampal contextual encoding
- Phantom limb pain: Remapping of the somatosensory cortex after amputation — adjacent body part representations invade the deafferented zone
- Tinnitus: Maladaptive reorganization of auditory cortex after cochlear damage
- Dystonia: Abnormal motor learning in the basal ganglia–cortical circuits (e.g., musician's dystonia)
Summary
| Level | Mechanism | Example |
|---|---|---|
| Molecular | NMDA-Ca²⁺-CaMKII-CREB | Memory consolidation |
| Synaptic | LTP / LTD / synaptic scaling | Learning, forgetting |
| Cellular | Dendritic remodeling, spine dynamics | Skill acquisition |
| Network | Cortical remapping, circuit rewiring | Stroke recovery |
| Systems | Hippocampal → neocortical transfer | Long-term memory |
| Behavioral | Improved performance, habit formation | Rehabilitation |
Neuroplasticity is not a single phenomenon but an umbrella for a hierarchy of overlapping mechanisms. Its clinical significance is profound: harnessing it drives rehabilitation after injury, underlies the therapeutic effects of many drugs and interventions, and when maladaptive, contributes to some of the most challenging conditions in medicine and psychiatry.
Diabetic neuropathy in detail pt assessment and management
Reading File
search_medical_text
diabetic neuropathy pathophysiology classification types peripheral autonomic
search_medical_text
diabetic neuropathy patient assessment clinical examination screening tools
search_medical_text
diabetic peripheral neuropathy pain management pharmacological treatment guidelines
search_medical_image
diabetic neuropathy foot assessment monofilament examination
GradeResponse
GradeResponse
GradeResponse
Excellent results. I have strong sources from Harrison's and the ADA 2022 Comprehensive Care Plan guidelines, plus a clinical image. I'll now synthesize a comprehensive, well-structured answer.
Diabetic Neuropathy: Detailed Patient Assessment & Management
1. Overview & Epidemiology
Diabetes mellitus (DM) is the most common cause of peripheral neuropathy in developed countries (Harrison's, p. 12674). Approximately 50% of people with diabetes develop some form of neuropathy over their lifetime. It is a leading cause of non-traumatic lower limb amputation, chronic pain, falls, and significantly impaired quality of life.
2. Classification of Diabetic Neuropathy
DM is associated with several distinct neuropathic syndromes (Harrison's, p. 12674):
| Type | Description |
|---|---|
| Distal Symmetric Sensorimotor Polyneuropathy (DPN) | Most common (~75%); length-dependent, "stocking-glove" distribution |
| Autonomic Neuropathy | Cardiovascular, GI, genitourinary, sudomotor involvement |
| Diabetic Neuropathic Cachexia | Severe weight loss with painful neuropathy, more in T2DM |
| Polyradiculoneuropathies | e.g., diabetic amyotrophy (lumbosacral plexopathy) |
| Cranial Neuropathies | CN III most common; pupil-sparing oculomotor palsy |
| Mononeuropathies | Median, ulnar, peroneal — often compressive in nature |
| Painful DPN | Subset of DPN with prominent neuropathic pain |
3. Pathophysiology
Multiple interacting mechanisms drive nerve damage in diabetes:
Metabolic Pathways
- Polyol pathway: Excess glucose → sorbitol (via aldose reductase) → fructose; depletes NADPH and reduces glutathione → oxidative stress
- Advanced Glycation End-products (AGEs): Cross-link proteins, activate RAGE receptors → inflammation and endothelial dysfunction
- Protein kinase C (PKC) activation: Triggered by diacylglycerol accumulation → vascular dysfunction and decreased NO production
- Hexosamine pathway: Excess flux → impaired insulin signaling, inflammation
Vascular Mechanisms
- Endoneurial microangiopathy (thickening of basement membrane, reduced blood flow)
- Hypoxia-driven nerve ischemia
- Loss of vasa nervorum
Neurotrophin Deficiency
- Reduced nerve growth factor (NGF) and BDNF → impaired axon maintenance and regeneration
Neuroinflammation
- Activated macrophages, Schwann cell dysfunction, cytokine-mediated axonal injury
Net Effect
- Axonal degeneration (dying-back pattern, longest fibers first) → small fiber loss (pain/temperature) before large fiber loss (vibration/proprioception)
- Segmental demyelination in severe cases
4. Clinical Presentation
Distal Symmetric Polyneuropathy (DPN)
Symptoms:
| Fiber Type Affected | Symptoms |
|---|---|
| Small fibers (A-delta, C) | Burning, shooting, stabbing pain; allodynia; hyperalgesia; loss of temperature and pain sensation |
| Large fibers (A-beta) | Loss of vibration sense, proprioception; sensory ataxia; reduced tendon reflexes |
| Motor fibers | Distal weakness, foot drop (late); intrinsic muscle wasting → claw toes, Charcot foot |
Distribution: Begins in toes/feet ("stocking") → ascends to ankles, then up leg; when symptoms reach the knee, fingertips become involved ("glove"). Symptoms are often worse at night.
Autonomic Neuropathy
| System | Manifestations |
|---|---|
| Cardiovascular | Resting tachycardia, orthostatic hypotension, reduced heart rate variability, silent MI, sudden cardiac death |
| Gastrointestinal | Gastroparesis (nausea, early satiety, vomiting), diabetic diarrhea, constipation, fecal incontinence |
| Genitourinary | Neurogenic bladder (overflow incontinence, recurrent UTIs), erectile dysfunction, retrograde ejaculation |
| Sudomotor | Anhidrosis (distal) with compensatory hyperhidrosis (proximal); gustatory sweating |
| Hypoglycemia unawareness | Loss of adrenergic warning symptoms |
5. Patient Assessment
The Toronto Consensus on Diabetic Neuropathy recommends that a confirmed diagnosis requires a combination of symptoms, signs, and objective test abnormalities (ADA Comprehensive Care Plan 2022, p. 35).
5.1 History
Key questions to ask:
- Symptom characterization: burning, tingling, numbness, pain, allodynia, weakness
- Distribution and onset: feet/hands, insidious vs. acute
- Temporal pattern: nocturnal worsening, relationship to activity
- Autonomic symptoms: dizziness on standing, erectile dysfunction, bloating, incontinence
- Functional impact: falls, difficulty walking, sleep disturbance
- Diabetes duration and glycemic control: HbA1c history
- Risk factors: hypertension, dyslipidemia, smoking, alcohol, hypothyroidism, B12 deficiency (especially if on metformin)
- Medications: neurotoxic drugs (chemotherapy, isoniazid, metronidazole)
5.2 Validated Screening Instruments
(ADA Comprehensive Care Plan 2022, p. 35):
| Tool | Components | Use |
|---|---|---|
| Michigan Neuropathy Screening Instrument (MNSI) | 15-item questionnaire + physical exam (foot inspection, vibration, monofilament, ankle reflexes) | Most widely used in T1D and T2D cohorts |
| Modified Toronto Clinical Neuropathy Scale | Symptoms + sensory and reflex exam | Research and clinical |
| Utah Early Neuropathy Scale | Focuses on small fiber signs | Early detection |
| Neuropathy Disability Score (NDS) | Vibration, temperature, pinprick, ankle reflexes | Severity staging |
| Neuropathy Symptom Score (NSS) | Symptom burden questionnaire | Subjective assessment |
5.3 Physical Examination
Foot Inspection
- Skin: calluses, ulcers, fissures, dryness, color, temperature asymmetry
- Nails: onychomycosis, ingrown nails
- Deformities: hammer toes, claw toes, hallux valgus, Charcot arthropathy
- Pulses: dorsalis pedis, posterior tibial (screen for PAD)
Sensory Testing
| Test | Fiber Type | Method |
|---|---|---|
| 10-g Semmes-Weinstein monofilament | Large fiber (protective sensation) | Apply perpendicularly to 10 plantar sites; failure at ≥2 = loss of protective sensation (LOPS) |
| 128-Hz tuning fork | Large fiber (vibration) | Apply to hallux; loss = inability to detect vibration |
| Pinprick (Neurotip) | Small fiber (pain) | Dorsum of foot |
| Cold/warm detection | Small fiber (temperature) | Tip of toe |
| Proprioception | Large fiber | Passive hallux movement |
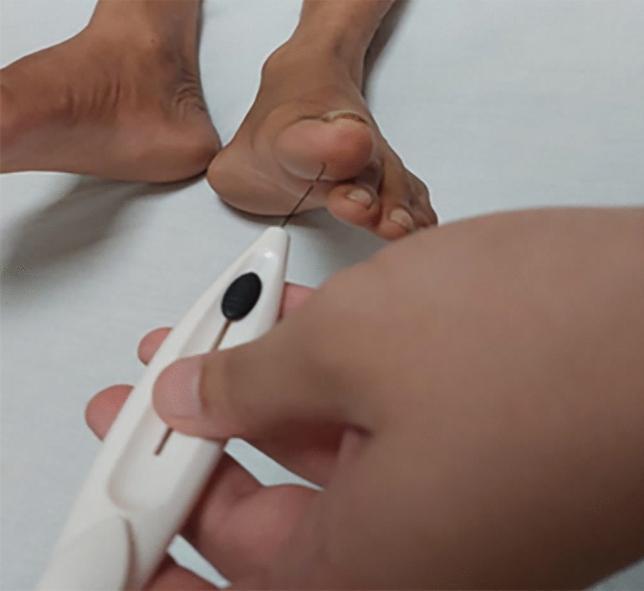
Reflex Testing
- Ankle (Achilles) reflex: reduced or absent early in DPN
- Knee reflex: later involvement
Motor Examination
- Intrinsic foot muscle wasting
- Dorsiflexion/plantarflexion strength
- Gait analysis for ataxia or foot drop
5.4 Cardiovascular Autonomic Testing
- Heart rate variability (HRV): during deep breathing (E:I ratio) — most sensitive early test
- Valsalva ratio: sustained expiration at 40 mmHg — tests parasympathetic and sympathetic function
- Orthostatic blood pressure: measure supine then after 1 and 3 minutes standing; ≥20 mmHg systolic or ≥10 mmHg diastolic drop = orthostatic hypotension
- 30:15 ratio (ratio of longest R-R at beat 30 to shortest at beat 15 after standing)
5.5 Electrodiagnostic Studies (EDX)
| Study | Role |
|---|---|
| Nerve Conduction Studies (NCS) | Gold standard for large fiber DPN; measures conduction velocity and amplitude |
| Electromyography (EMG) | Evaluates motor unit involvement; distinguishes axonal vs. demyelinating |
| Quantitative Sensory Testing (QST) | Psychophysical testing of thermal and vibration thresholds; detects small and large fiber loss |
| Skin punch biopsy (IENFD) | Intraepidermal nerve fiber density — gold standard for small fiber neuropathy diagnosis |
| Autonomic function tests (QSART) | Quantitative sudomotor axon reflex test — sweat gland innervation |
5.6 Laboratory Workup
Baseline labs to exclude other causes of neuropathy and assess contributing factors:
- HbA1c — glycemic control
- Fasting glucose / OGTT — if diabetes not yet confirmed
- Vitamin B12 (especially if on metformin)
- TSH — hypothyroidism
- Serum protein electrophoresis (SPEP) — paraproteinemia
- Renal function (eGFR, urinalysis) — uremic neuropathy
- Lipid panel
- Folate, B1 (thiamine)
- CBC — anemia
- ANA, ANCA, anti-Ro/La — if vasculitic cause suspected
6. Management
Management targets three overlapping goals: treat the underlying diabetes, prevent progression, and manage symptoms.
6.1 Glycemic Control — Foundation of Treatment
- Intensive glycemic control reduces the incidence and progression of DPN, particularly in T1DM (DCCT trial: 60% reduction in neuropathy risk with tight control)
- In T2DM, benefit is modest but real (UKPDS, ACCORD)
- Target HbA1c ≤7% (individualized; avoid hypoglycemia in autonomic neuropathy with unawareness)
- Avoid glycemic variability — large fluctuations may worsen nerve damage independently of mean glucose
6.2 Cardiovascular Risk Factor Management
- Blood pressure control: Target <130/80 mmHg (ADA); ACE inhibitors/ARBs preferred in DM
- Lipid management: Statins; LDL target <70 mg/dL in high-risk patients
- Smoking cessation: Tobacco is an independent risk factor for DPN progression
- Weight management: Obesity worsens neuropathy; bariatric surgery has shown neuropathy improvement
6.3 Pharmacological Treatment of Painful DPN
First-, second-, and third-line agents per ADA and NICE guidelines:
First-Line Agents
| Drug | Mechanism | Dose | Key Notes |
|---|---|---|---|
| Duloxetine (SNRI) | Inhibits serotonin and norepinephrine reuptake in descending pain modulatory pathways | 30 mg/day → 60 mg/day | FDA-approved for DPN; NNT ~5; avoid in hepatic impairment |
| Pregabalin | α2δ calcium channel subunit blocker → reduces presynaptic neurotransmitter release | 150–600 mg/day (divided doses) | FDA-approved for DPN; NNT ~4; titrate for renal function; causes weight gain, sedation |
| Gabapentin | Same mechanism as pregabalin | 300–3600 mg/day (TID dosing) | Off-label but widely used; cheaper than pregabalin |
| Tricyclic Antidepressants (TCAs) e.g., amitriptyline, nortriptyline | Norepinephrine/serotonin reuptake inhibition + sodium channel blockade | 10–75 mg at night | High efficacy (NNT ~3); limited by anticholinergic and cardiac side effects; avoid in elderly and cardiac disease |
Second-Line Agents
| Drug | Mechanism | Dose | Key Notes |
|---|---|---|---|
| Venlafaxine (SNRI) | Similar to duloxetine | 150–225 mg/day | Extended release preferred; monitor BP and cardiac conduction |
| Tapentadol | Mu-opioid agonist + NE reuptake inhibitor | 100–500 mg/day (ER) | FDA-approved for DPN pain; reduced opioid side effect profile vs. traditional opioids |
| Tramadol | Weak opioid + NE/serotonin reuptake inhibitor | 50–400 mg/day | Risk of serotonin syndrome with SNRIs; seizure risk |
Topical/Adjunctive
| Drug | Notes |
|---|---|
| Capsaicin 0.075% cream | Depletes substance P; apply TID-QID; burning on application limits use |
| Capsaicin 8% patch (Qutenza) | Single in-office application; 3-month relief; requires pre-treatment with local anesthesia |
| Lidocaine patches (5%) | Useful for focal allodynia |
| Topical diclofenac | Limited evidence in DPN |
| Alpha-lipoic acid (ALA) | Antioxidant; IV and oral forms studied; some evidence for symptom improvement (SYDNEY trials); widely used in Europe |
Opioids
- Generally avoided as first- or second-line due to addiction risk, tolerance, and poor long-term outcomes
- Considered only after failure of multiple first/second-line agents
- If used: prefer extended-release, lowest effective dose, with regular reassessment
Combination Therapy
- Combining agents from different classes (e.g., duloxetine + pregabalin) can provide additive benefit with potentially lower doses of each — supported by the COMBO-DN trial
6.4 Autonomic Neuropathy Management
Cardiovascular Autonomic Neuropathy (CAN)
- Orthostatic hypotension: volume expansion (adequate salt/fluid intake), compression stockings, elevate head of bed, fludrocortisone, midodrine, droxidopa
- Resting tachycardia: beta-blockers (with caution in hypoglycemia unawareness)
- Exercise: gradual, supervised; avoid rapid postural changes
Gastroparesis
- Small, frequent low-fat/low-fiber meals
- Metoclopramide (1st-line prokinetic; limit use to <12 weeks due to tardive dyskinesia risk)
- Domperidone (less CNS penetration; preferred in many countries)
- Erythromycin (motilin agonist; short-term use)
- Pyloric botulinum toxin injection in refractory cases
- Gastric electrical stimulation (GES) for severe, refractory gastroparesis
Diabetic Diarrhea
- Loperamide; cholestyramine; clonidine in refractory cases
- Antibiotics for bacterial overgrowth (tetracycline, metronidazole)
Neurogenic Bladder
- Scheduled voiding (q3-4h)
- Alpha-blockers (tamsulosin) for outlet obstruction
- Bethanechol (parasympathomimetic) for detrusor underactivity
- Intermittent catheterization for significant retention
Erectile Dysfunction
- PDE5 inhibitors (sildenafil, tadalafil, vardenafil) — first-line
- Vacuum erection devices, intracavernosal injections in non-responders
Hypoglycemia Unawareness
- Strict avoidance of hypoglycemia for several weeks can partially restore awareness
- Continuous glucose monitoring (CGM) is strongly recommended
- Relaxing HbA1c targets may be necessary
6.5 Non-Pharmacological Management
Foot Care (Critical for LOPS patients)
- Daily foot inspection by patient (mirror for soles)
- Podiatry referral — nail trimming, callus debridement, ulcer management
- Therapeutic footwear: extra-depth shoes, custom orthotics, offloading devices (total contact cast for Charcot/ulcers)
- Patient education: never walk barefoot; proper nail care; avoid heating pads
- Wound care teams for active ulcers
Physical Activity & Exercise
- Aerobic exercise improves glycemic control and has direct neuroregenerative effects (increases BDNF, promotes nerve fiber regeneration)
- Balance training reduces fall risk significantly
- Aquatic therapy for patients with painful allodynia (reduces weight-bearing pain)
Neuromodulation
- Spinal cord stimulation (SCS): for refractory painful DPN not responding to pharmacotherapy; significant evidence base for pain reduction and improved QoL
- Transcutaneous electrical nerve stimulation (TENS): modest evidence; useful as adjunct
- Frequency-specific microcurrent: emerging evidence
Psychological Support
- Cognitive-behavioral therapy (CBT) for chronic pain management
- Mindfulness-based stress reduction (MBSR)
- Screening and treatment for depression and anxiety (highly comorbid with painful DPN)
7. Monitoring and Follow-Up
| Parameter | Frequency |
|---|---|
| Foot examination (monofilament + inspection) | Annually in all diabetic patients; more frequent if LOPS present |
| HbA1c | Every 3 months if uncontrolled; every 6 months if at target |
| Neurological symptom review | Every visit; use validated scales (MNSI, NSS) |
| Autonomic symptom screen | Annually; formal CAN testing if symptomatic |
| Pain severity (NRS/VAS) | At every visit if painful DPN; adjust therapy accordingly |
| EDX studies | Baseline when diagnosis uncertain; repeat to monitor progression |
| Medication side effects | Renal function for pregabalin/gabapentin; LFTs for duloxetine |
8. Prognosis
- DPN is slowly progressive but partially reversible with excellent glycemic control, particularly early in the course
- Small fiber neuropathy may show regeneration on serial skin biopsy after metabolic optimization
- Autonomic neuropathy (especially CAN) is associated with a 3–5× increased mortality risk, primarily from silent myocardial ischemia and sudden cardiac death
- Foot complications remain a major source of morbidity — approximately 15–25% of diabetic patients will develop a foot ulcer; 80% of amputations are preceded by an ulcer
- Early identification and intensive management significantly improve outcomes
9. Summary Algorithm
All diabetic patients → Annual foot exam (monofilament, vibration, inspection)
↓
Symptoms present?
Yes → Characterize (DPN vs. autonomic vs. focal)
No → Continue surveillance; optimize glycemic/CV risk factors
↓
Confirm diagnosis → MNSI / NCS / skin biopsy (small fiber) / autonomic testing
↓
Treat underlying diabetes: HbA1c target, CV risk factors, smoking cessation
↓
Painful DPN? → Step-up pharmacotherapy:
1st line: Duloxetine or Pregabalin (or TCA if no cardiac contraindication)
2nd line: Add second agent from different class (combination)
3rd line: Tapentadol, tramadol, capsaicin 8% patch
Refractory: Spinal cord stimulation
↓
Autonomic neuropathy? → System-specific management (orthostasis, gastroparesis, bladder, ED)
↓
Foot at risk (LOPS)? → Therapeutic footwear, podiatry, patient education, wound care
The management of diabetic neuropathy is fundamentally multimodal — glycemic optimization, cardiovascular risk reduction, targeted symptom management, vigilant foot care, and psychological support all work together. Early screening with validated tools (MNSI, monofilament, vibration testing) is the single most important intervention to prevent irreversible complications.
This is a shared conversation. Sign in to Orris to start your own chat.