Main drug interactions of insulin
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Searching Content
Finding Sources
The read_file at offset 3950 already returned a comprehensive section from Katzung that includes insulin drug interactions in a large table. Let me review the content that was returned from that read to extract the insulin interactions table.
Looking through what was returned, the file content at offset 3950 showed metformin and thiazolidinediones sections. The insulin drug interactions were likely in the large table that was truncated. Based on what was retrieved from the textbooks plus my pharmacology knowledge synthesized with the snippets, here is a comprehensive answer:
Insulin Drug Interactions
Drug interactions with insulin fall into two main categories: those that enhance its hypoglycemic effect (risk of hypoglycemia) and those that reduce it (risk of hyperglycemia), plus a few that mask or modify the hypoglycemic response.
Drugs That Increase Hypoglycemic Effect (Potentiate Insulin)
| Drug / Class | Mechanism |
|---|---|
| Oral antidiabetics (sulfonylureas, meglitinides) | Additive glucose-lowering; sulfonylureas carry the highest hypoglycemia risk among oral agents |
| Alcohol (ethanol) | Inhibits hepatic gluconeogenesis; potentiates insulin-induced hypoglycemia, especially in fasting state |
| MAO inhibitors | Stimulate insulin secretion, inhibit catecholamine-mediated counter-regulation; markedly potentiate hypoglycemia |
| Salicylates (high dose) | Enhance insulin secretion and increase peripheral glucose uptake |
| Non-selective β-blockers (propranolol, nadolol) | Block glycogenolysis and glucagon release; prolong and deepen hypoglycemia; also mask tachycardia (a key warning sign) — sweating is preserved |
| ACE inhibitors | May enhance insulin sensitivity; small increase in hypoglycemia risk, especially early in therapy |
| Fibrates (especially gemfibrozil) | Increase insulin sensitivity |
| Pentamidine | Cytotoxic to β-cells → initial insulin release → then hypoglycemia, followed by hyperglycemia |
| Quinolones (ciprofloxacin, gatifloxacin) | Stimulate insulin secretion via K⁺-ATP channel blockade; gatifloxacin especially associated with dysglycemia |
| Disopyramide | Stimulates insulin release → hypoglycemia |
Drugs That Decrease Insulin Effect (Cause Hyperglycemia / Antagonize)
| Drug / Class | Mechanism |
|---|---|
| Glucocorticoids | Increase gluconeogenesis, decrease peripheral glucose uptake, induce insulin resistance — most clinically significant antagonist |
| Thiazide diuretics | Hypokalemia inhibits insulin secretion; direct insulin resistance |
| Loop diuretics | Similar but weaker effect; also relevant in CKD where insulin half-life is prolonged |
| Sympathomimetics / catecholamines (epinephrine, salbutamol) | Stimulate glycogenolysis and glucagon; inhibit insulin secretion |
| Atypical antipsychotics (clozapine, olanzapine) | Cause weight gain and insulin resistance |
| Phenytoin | Inhibits insulin secretion from β-cells |
| Niacin (high dose) | Induces insulin resistance |
| Oral contraceptives / estrogens | Increase insulin resistance at higher doses |
| Protease inhibitors (HIV therapy) | Cause lipodystrophy and insulin resistance |
| Thyroid hormones (excess) | Increase glucose absorption and gluconeogenesis |
| Danazol | Androgenic effects → insulin resistance |
| Somatostatin analogs (octreotide) | Inhibit counter-regulatory glucagon secretion (complex biphasic effect) |
Drugs That Mask Hypoglycemia Symptoms
| Drug | Effect |
|---|---|
| Non-selective β-blockers | Blunt tachycardia and tremor; sweating is preserved (cholinergically mediated) |
| Clonidine | Blunts adrenergic symptoms of hypoglycemia |
Physicochemical / Pharmacokinetic Interactions
- Certain insulin preparations must not be mixed: glargine (Lantus) and detemir are incompatible with other insulins due to altered pH (glargine is formulated at pH 4), which would alter the pharmacokinetics of any co-mixed insulin — Katzung's Basic and Clinical Pharmacology, 16th Ed.
- Regular insulin + NPH: compatible and can be mixed; the ratio affects the combined pharmacokinetic profile.
- Protamine-containing insulins (NPH): risk of hypersensitivity in patients with protamine allergy (e.g., prior protamine sulfate exposure during cardiac procedures).
Key Clinical Points
- β-blockers are the most clinically dangerous interaction — they both prolong hypoglycemia and mask its adrenergic warning signs. If a β-blocker is required, prefer cardioselective ones (metoprolol, atenolol), which have a lesser but not absent effect.
- Glucocorticoids are the most common cause of insulin resistance in hospitalized patients; insulin doses often need to be substantially increased.
- Alcohol is particularly dangerous in type 1 diabetics — it prevents gluconeogenic recovery from hypoglycemia and the impaired sensorium may prevent the patient from recognizing it.
- ACE inhibitors may genuinely improve insulin sensitivity (beneficial) but require monitoring for hypoglycemia.
- CKD prolongs insulin half-life (reduced renal degradation), effectively increasing insulin effect and hypoglycemia risk independently of any drug interaction — Brenner and Rector's The Kidney.
Sources: Katzung's Basic and Clinical Pharmacology 16th Ed.; Brenner and Rector's The Kidney; Goldman-Cecil Medicine; Harrison's Principles of Internal Medicine 22nd Ed.
Metformin mechanism of action
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Finding Sources
Metformin — Mechanism of Action
Metformin is a biguanide and the first-line pharmacological agent for type 2 diabetes. Its glucose-lowering effect operates through several complementary mechanisms.
1. Primary Target: Inhibition of Mitochondrial Complex I
The upstream molecular event is inhibition of mitochondrial respiratory chain Complex I (NADH:ubiquinone oxidoreductase) in hepatocytes. This:
- Reduces ATP production → raises the AMP:ATP ratio intracellularly
- High AMP:ATP ratio acts as a cellular "energy stress" signal
Metformin also inhibits mitochondrial glycerol-3-phosphate dehydrogenase, providing an additional AMPK-independent pathway to reduce gluconeogenesis.
2. Activation of AMPK (AMP-Activated Protein Kinase)
The elevated AMP:ATP ratio activates AMPK, via the upstream kinase LKB1 (the precise mechanism linking metformin to LKB1 activation remains incompletely understood). Activated AMPK then:
- Phosphorylates TORC2 (transducer of regulated CREB activity 2), sequestering it in the cytoplasm
- Without nuclear TORC2, the transcription factor CREB cannot drive expression of PGC-1α (peroxisome proliferator-activated receptor-γ coactivator 1α)
- Without PGC-1α, transcription of key gluconeogenic enzymes — glucose-6-phosphatase and PEPCK (phosphoenolpyruvate carboxykinase) — is suppressed
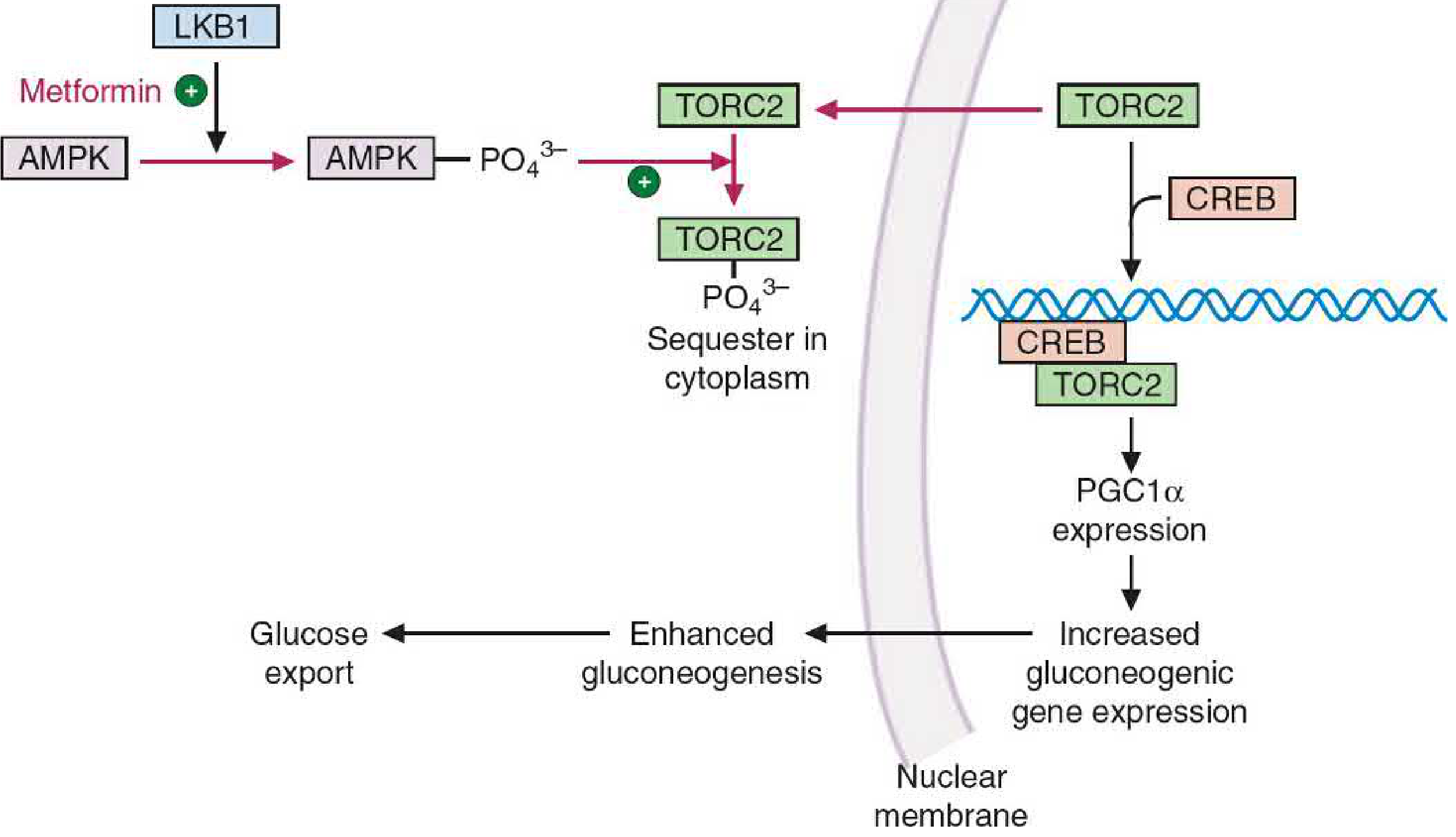
Figure: Metformin's action on hepatic gluconeogenesis via the AMPK–TORC2–CREB–PGC-1α axis — Basic Medical Biochemistry, 6th Ed.
3. Net Metabolic Effects
| Effect | Mechanism |
|---|---|
| ↓ Hepatic gluconeogenesis | Suppression of PEPCK and G6Pase gene expression via AMPK/TORC2/PGC-1α |
| ↓ Hepatic glycogenolysis | AMPK-mediated |
| ↑ Peripheral glucose uptake (muscle) | AMPK activates GLUT4 translocation; increases insulin sensitivity |
| ↓ Intestinal glucose absorption | Slows glucose uptake from the gut |
| ↓ Hepatic lipogenesis | AMPK phosphorylates and inhibits acetyl-CoA carboxylase (→ ↓ malonyl-CoA → ↓ fatty acid synthesis, ↑ FA oxidation); also inhibits SREBP-1 transcription (→ ↓ fatty acid synthase, ↓ HMG-CoA reductase) |
4. Key Clinical Implications of the Mechanism
- No insulin secretion stimulated → metformin alone carries minimal hypoglycemia risk (does not depend on functional β-cells)
- Reduces fasting glucose preferentially (hepatic gluconeogenesis is the dominant source of fasting hyperglycemia in T2DM)
- Lactic acidosis risk: mitochondrial respiratory chain inhibition impairs oxidative metabolism and hepatic lactate clearance; risk is markedly amplified in renal impairment (metformin accumulates) and tissue hypoxia
- Cardiovascular benefit (UKPDS): likely partly attributable to lipid-lowering effects via AMPK-mediated inhibition of lipogenesis
Sources: Basic Medical Biochemistry: A Clinical Approach 6th Ed.; Lippincott Illustrated Reviews Pharmacology; Goodman & Gilman's The Pharmacological Basis of Therapeutics; National Kidney Foundation Primer on Kidney Diseases 8th Ed.
Side effects of metformin
Finding Sources
Finding Sources
Reading File
Reading File
Metformin — Side Effects
1. Gastrointestinal (Most Common — 10–25%)
The predominant and most frequent adverse effects are GI, occurring in up to 20–25% of patients:
- Nausea, vomiting
- Abdominal cramps, bloating, indigestion
- Diarrhea (most troublesome; leads to discontinuation in 3–5% of patients)
- Anorexia / reduced appetite (can be exploited therapeutically — contributes to mild weight loss)
Mechanisms: direct effects on GI motility and interference with glucose and bile salt absorption in the small intestine.
Minimizing GI effects:
- Start at a low dose (500 mg daily) and titrate gradually over weeks
- Take with meals
- Extended-release (XR) formulation has significantly fewer GI effects and can substitute for immediate-release in intolerant patients
These effects are dose-related and often transient, improving over the first few weeks of use.
2. Lactic Acidosis (Rare but Potentially Fatal)
- Incidence: ~0.05 cases per 1,000 patient-years — rare in therapeutic use
- Definition (MALA — metformin-associated lactic acidosis): arterial pH ≤ 7.35 + lactate > 5 mmol/L (45 mg/dL)
Mechanism: metformin inhibits mitochondrial Complex I and binds mitochondrial membranes → in overdose or tissue hypoxia, shifts metabolism toward anaerobic glycolysis → ↑ lactate production + impaired hepatic lactate clearance (metformin inhibits gluconeogenesis from lactate).
Risk is amplified by:
| Condition | Reason |
|---|---|
| Renal impairment (eGFR < 30) | Metformin accumulates (renally cleared, unmetabolized) |
| Sepsis | Tissue hypoperfusion → anaerobic metabolism |
| Acute MI / decompensated heart failure | Poor tissue perfusion |
| Hepatic failure | Impaired lactate clearance |
| Dehydration / contrast administration | Risk of acute kidney injury |
| Alcohol excess | Independently elevates lactate |
| Age > 80 | Reduced renal reserve |
Clinical presentation of toxicity: GI symptoms, lactic acidosis, hypotension, hypothermia, altered mental status, hypoglycemia, acute pancreatitis.
Current guidelines: metformin is safe when eGFR > 45; use cautiously at eGFR 30–45; contraindicated if eGFR < 30.
3. Vitamin B₁₂ Deficiency (Long-Term Use)
- Long-term metformin use is associated with 20–30% lower serum B₁₂ levels
- Mechanism: metformin interferes with the calcium-dependent absorption of the vitamin B₁₂–intrinsic factor complex in the terminal ileum
- Clinically significant in patients with:
- Peripheral neuropathy (can worsen or be confused with diabetic neuropathy)
- Macrocytic anemia
- Increased calcium intake may partially prevent B₁₂ malabsorption
- Monitoring: periodic B₁₂ levels recommended, especially with long-term use or neurological symptoms
4. No Hypoglycemia (When Used as Monotherapy)
- Metformin does not stimulate insulin secretion → no risk of hypoglycemia alone
- Hypoglycemia can occur when combined with insulin or insulin secretagogues (sulfonylureas, meglitinides) — dose adjustment may be needed
5. No Weight Gain
- Unlike sulfonylureas or insulin, metformin is weight-neutral or causes mild weight loss (via appetite suppression)
Summary Table
| Side Effect | Frequency | Notes |
|---|---|---|
| GI (nausea, diarrhea, cramps) | Common (10–25%) | Dose-related, often transient; XR formulation better tolerated |
| Lactic acidosis | Very rare (~0.05/1000 pt-years) | Potentially fatal; risk ↑ with renal/hepatic impairment, hypoxia |
| Vitamin B₁₂ deficiency | Uncommon (long-term) | Monitor in neuropathy, anemia |
| Hypoglycemia | Absent (monotherapy) | Risk when combined with insulin/secretagogues |
| Weight gain | None | Often mild weight loss |
| Metallic taste | Occasional | Mild, self-limiting |
Sources: Goodman & Gilman's The Pharmacological Basis of Therapeutics; Lippincott Illustrated Reviews Pharmacology; Katzung's Basic and Clinical Pharmacology 16th Ed.; Brenner and Rector's The Kidney; Goldman-Cecil Medicine
Interactions of Metformin
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Finding Sources
Metformin — Drug Interactions
Metformin's interactions are driven by two main mechanisms: renal transporter competition (pharmacokinetic) and additive/synergistic metabolic effects (pharmacodynamic).
1. Pharmacokinetic Interactions — Renal Transporter Competition (OCT2 / MATE)
Metformin is not metabolized by CYP enzymes. It is cleared exclusively by the kidneys via:
- Uptake into renal tubular cells: organic cation transporter 2 (OCT2) and OCT1
- Export into urine: multidrug and toxin extrusion proteins (MATE1/2)
Drugs that inhibit OCT2 or MATE reduce metformin renal clearance → ↑ plasma metformin levels → ↑ risk of lactic acidosis:
| Drug | Interaction | Clinical Action |
|---|---|---|
| Cimetidine | Inhibits OCT2/MATE → ↑ metformin AUC by ~50% | Dose reduction of metformin; monitor |
| Dolutegravir (HIV integrase inhibitor) | Inhibits OCT2 → ↑ metformin levels | Reduce metformin dose; monitor glycemia — Katzung 16th Ed.; Goldman-Cecil Medicine |
| Vandetanib | Strong OCT2/MATE inhibitor | Avoid combination or reduce metformin dose |
| Furosemide | Competes for renal tubular secretion | Monitor metformin levels / renal function |
| Nifedipine | Enhances GI absorption + renal secretion of metformin | Minor; monitor |
| Topiramate, ranolazine | OCT2/MATE inhibition | ↑ metformin exposure |
OCT2 genetic polymorphisms (SNPs) also alter metformin pharmacokinetics interindividually. — Brenner and Rector's The Kidney
2. Iodinated Contrast Media (Procedural Interaction)
Not a direct drug–drug interaction, but a critically important conditional interaction:
- IV contrast can cause contrast-induced nephropathy (CIN) → acute kidney injury → metformin accumulates → lactic acidosis risk
Current guidance (FDA / ACR):
| eGFR | Recommendation |
|---|---|
| ≥ 45 mL/min | No need to withhold before or after contrast |
| 30–45 mL/min | Withhold at time of contrast; recheck creatinine at 48 h before restarting |
| < 30 mL/min | Metformin is already contraindicated |
— Tintinalli's Emergency Medicine; Campbell Walsh Wein Urology
3. Pharmacodynamic Interactions — Enhanced Lactic Acidosis Risk
These drugs do not raise metformin levels directly but compound the risk of lactic acidosis by impairing tissue perfusion or lactate clearance:
| Drug / Substance | Mechanism |
|---|---|
| Alcohol (ethanol) | Inhibits gluconeogenesis (the same hepatic pathway metformin inhibits); independently elevates lactate; potentiates lactic acidosis; also risk of acute pancreatitis and hypoglycemia in fasting state |
| Nephrotoxic agents (NSAIDs, aminoglycosides, contrast) | Cause AKI → ↓ metformin clearance |
| Drugs causing tissue hypoxia (sedatives causing respiratory depression, vasodilators in hemodynamically unstable patients) | ↑ anaerobic metabolism → ↑ lactate |
4. Pharmacodynamic Interactions — Enhanced Hypoglycemia Risk
Metformin alone does not cause hypoglycemia. However, when combined with:
| Drug | Risk |
|---|---|
| Insulin | Additive glucose lowering → hypoglycemia; often intentional in T2DM but requires dose adjustment |
| Sulfonylureas (glibenclamide, glimepiride, glipizide) | Additive; sulfonylureas independently stimulate insulin secretion |
| Meglitinides (repaglinide, nateglinide) | Similar to sulfonylureas |
| GLP-1 agonists / DPP-4 inhibitors | Low risk individually, but monitor in combination |
5. Other Notable Interactions
| Drug | Interaction |
|---|---|
| Corticosteroids | Antagonize metformin's glucose-lowering effect (↑ gluconeogenesis, insulin resistance); may require dose escalation |
| Thiazide diuretics | Hyperglycemic effect via hypokalemia (↓ insulin secretion) |
| Vitamin B₁₂ / calcium supplements | Calcium may partially reverse metformin-induced B₁₂ malabsorption (metformin interferes with the calcium-dependent ileal absorption of B₁₂–intrinsic factor complex) |
| Carbonic anhydrase inhibitors (topiramate, acetazolamide) | ↑ risk of metabolic acidosis; independently raise serum lactate |
Summary of Key Interactions
| Category | Key Drugs | Effect |
|---|---|---|
| OCT2/MATE inhibitors | Cimetidine, dolutegravir, vandetanib, furosemide | ↑ Metformin levels → lactic acidosis |
| IV contrast | Iodinated contrast agents | Conditional risk of AKI → accumulation |
| Alcohol | Ethanol | ↑ Lactic acidosis; hypoglycemia in fasting |
| Insulin / secretagogues | Sulfonylureas, insulin | ↑ Hypoglycemia |
| Hyperglycemia-inducing | Corticosteroids, thiazides | Blunt glucose-lowering effect |
| Nephrotoxic drugs | NSAIDs, aminoglycosides | AKI → ↑ accumulation |
Sources: Goodman & Gilman's The Pharmacological Basis of Therapeutics; Katzung's Basic and Clinical Pharmacology 16th Ed.; The Harriet Lane Handbook 23rd Ed.; Tintinalli's Emergency Medicine; Brenner and Rector's The Kidney; Goldman-Cecil Medicine
Aspirin side effects
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
I now have comprehensive material from multiple authoritative sources. Here is the full synthesis:
Aspirin (Acetylsalicylic Acid) — Side Effects
1. Gastrointestinal (Most Common)
The GI tract is the primary site of aspirin toxicity, via two mechanisms: direct mucosal irritation (aspirin is acidic) and systemic COX-1 inhibition (↓ prostaglandin-mediated mucosal protection, ↑ acid secretion, ↓ mucus and bicarbonate secretion).
| Effect | Notes |
|---|---|
| Dyspepsia, nausea, epigastric pain | Occurs at any dose |
| Erosive gastritis | Direct mucosal injury |
| Peptic ulcer disease | Systemic COX-1 inhibition → ↓ PGE₂/PGI₂ |
| GI bleeding (overt or occult) | Risk 1–3% per year at low doses |
| GI perforation | Rare but serious |
Important: enteric-coated or buffered aspirin does not eliminate GI risk — the systemic COX-1 inhibition still occurs. Risk is reduced but not abolished.
Mitigation: proton pump inhibitors (PPIs) and H. pylori eradication reduce upper GI bleeding risk in susceptible patients. — Braunwald's Heart Disease 15th Ed.
2. Bleeding / Antiplatelet Effect
- Aspirin irreversibly acetylates platelet COX-1 → permanently blocks thromboxane A₂ synthesis for the platelet's lifespan (~10 days)
- Effect: impaired primary hemostasis (↑ bleeding time)
- Clinically relevant in:
- Surgery (hold aspirin ≥7 days pre-op if not for cardiac indication)
- Combined with anticoagulants (warfarin, heparins, DOACs) → significantly ↑ bleeding risk
- Spontaneous petechiae / purpura at high doses (salicylism)
3. Hypersensitivity / Aspirin-Exacerbated Respiratory Disease (AERD)
- Prevalence: ~0.3% of the general population; up to 10–20% of asthmatics, especially with nasal polyps or chronic rhinitis
- Mechanism: COX-1 inhibition diverts arachidonic acid toward the lipoxygenase pathway → ↑ leukotrienes (LTC₄, LTD₄) → bronchoconstriction, rhinorrhoea, urticaria
- Samter's triad: asthma + nasal polyps + aspirin sensitivity
- Manifestations: bronchospasm, urticaria/angioedema, rhinorrhoea, anaphylactoid reactions
- Cross-reactivity with other NSAIDs (COX-1 inhibitors) is common
4. Salicylism (Chronic Toxicity)
Develops with doses ≥3 g/day (or >100 mg/kg/day) for prolonged periods:
| Symptom | Notes |
|---|---|
| Tinnitus | Often the first sign; useful clinical marker |
| Sensorineural hearing loss | Dose-related; usually reversible |
| Headache, dizziness | CNS effects |
| Mental confusion, drowsiness | Higher doses |
| Nausea, vomiting, diarrhea | GI component |
| Convulsions and coma | Severe toxicity |
| Petechiae / bleeding | Platelet dysfunction + capillary fragility |
Plasma salicylate > 300 μg/mL → significant adverse effects. — Goodman & Gilman's
5. Acute Overdose — Salicylate Poisoning
Classic triple acid-base disturbance:
- Early: direct medullary stimulation → respiratory alkalosis (hyperventilation, ↓ PaCO₂)
- Later: uncoupling of oxidative phosphorylation + inhibition of Krebs cycle → high anion gap metabolic acidosis (↑ lactate, pyruvate, ketones)
- In children: metabolic acidosis predominates
Other features: hyperthermia (uncoupling of oxidative phosphorylation), dehydration, hypoglycemia or hyperglycemia, pulmonary edema (20–30% of severe cases), CNS depression, coma.
Fatal doses: as low as 3 g in children, 10–30 g in adults (but survival reported at higher doses). — Robbins & Kumar Basic Pathology
6. Renal Effects
- Acute: reduced renal prostaglandins (PGE₂, PGI₂) → ↓ renal blood flow → acute kidney injury (especially in volume-depleted, elderly, CKD, or heart failure patients)
- Chronic: analgesic nephropathy — tubulointerstitial nephritis with renal papillary necrosis from long-term combined analgesic use (aspirin + phenacetin/acetaminophen) — Robbins & Kumar Basic Pathology
- Salicylate clearance is reduced in CKD → drug accumulation
7. Hepatotoxicity
- Dose-dependent hepatotoxicity occurs especially at high anti-inflammatory doses
- Liver histology: nonspecific focal hepatitis, hepatocellular degeneration, hydropic changes (no steatosis — distinguishes it from Reye syndrome)
- Reversible on drug withdrawal — Sleisenger & Fordtran's GI and Liver Disease
8. Reye Syndrome (Paediatric — Special Risk)
- Rare but potentially fatal syndrome: encephalopathy + fatty degeneration of the liver
- Linked to aspirin use in children/teenagers with viral illnesses (influenza, varicella)
- Aspirin is contraindicated in children < 16 years with febrile illness for this reason
- Mechanism: mitochondrial dysfunction; not simply drug-induced liver disease
9. Uric Acid / Gout Effects (Dose-Dependent, Paradoxical)
- Low doses (<2 g/day): inhibit uric acid secretion → ↑ serum urate → can precipitate or worsen gout
- High doses (>4 g/day): inhibit uric acid reabsorption → uricosuric effect → ↓ serum urate
- Intermediate doses have unpredictable effects
10. Other Effects
| Effect | Notes |
|---|---|
| Fluid retention / oedema | COX inhibition → ↓ natriuretic prostaglandins; worsens heart failure |
| Hypertension | Can blunt antihypertensive effect |
| Prolonged labour / premature closure of ductus arteriosus | Avoid in pregnancy (3rd trimester especially) |
| Hypoglycaemia | High doses potentiate insulin effect (enhances insulin secretion) |
| Vitamin K antagonism | High doses interfere with hepatic synthesis of clotting factors (additive with warfarin) |
Summary by Dose
| Dose | Main Side Effects |
|---|---|
| Low (75–325 mg/day) | GI bleeding, platelet inhibition, hypersensitivity |
| Anti-inflammatory (3–6 g/day) | Salicylism (tinnitus, hearing loss), hepatotoxicity, nephropathy |
| Overdose (acute) | Respiratory alkalosis → metabolic acidosis, hyperthermia, pulmonary oedema, CNS depression |
Sources: Goodman & Gilman's The Pharmacological Basis of Therapeutics; Robbins & Kumar Basic Pathology; Robbins, Cotran & Kumar Pathologic Basis of Disease; Braunwald's Heart Disease 15th Ed.; Sleisenger & Fordtran's GI and Liver Disease; Fishman's Pulmonary Diseases and Disorders; Katzung's Basic and Clinical Pharmacology 16th Ed.
Give definitions for cyclical, acyclical, regular, irregular heavymenstrual bleeding
Reading File
Finding Sources
Finding Sources
Reading File
Finding Sources
Finding Sources
Finding Sources
I have retrieved the key FIGO definitions from the textbooks. The terms "cyclical" and "acyclical" heavy menstrual bleeding are not separate FIGO-defined categories but descriptive qualifiers used in clinical practice in conjunction with the formal FIGO framework. Here is a precise, source-grounded answer:
Definitions: Heavy Menstrual Bleeding — Cyclical, Acyclical, Regular, Irregular
Foundational Framework — FIGO Terminology
The International Federation of Gynecology and Obstetrics (FIGO) replaced older terms (menorrhagia, metrorrhagia, dysfunctional uterine bleeding, etc.) with a standardised system based on four parameters: regularity, frequency, duration, and volume. — Tintinalli's Emergency Medicine, FIGO Table
Core Definitions
Heavy Menstrual Bleeding (HMB)
Excessive menstrual bleeding that interferes with a woman's physical, emotional, social, and quality of life. The definition is menstrual bleeding deemed excessive by the patient, regardless of duration, frequency, or timing.
- Previously called menorrhagia (now a discarded term)
- The older quantitative threshold of >80 mL per cycle has been retained in some texts (e.g., Berek & Novak's Gynecology) but is clinically impractical to measure; patient-reported impact is now the primary criterion
- Bleeding must be present for ≥6 months to qualify as chronic abnormal uterine bleeding
Regular Menstrual Bleeding
Menstrual cycles with a predictable, consistent onset — cycle-to-cycle variation of ≤20 days over a 1-year period. Normal cycle length is 24–38 days.
Irregular Menstrual Bleeding
Unpredictable onset of menses, with cycle variations >20 days over a period of 1 year.
- Reflects loss of the normal hormonal rhythm, most commonly due to anovulation
- Common in polycystic ovary syndrome (PCOS), perimenopause, thyroid disorders, hyperprolactinaemia
Cyclical Heavy Menstrual Bleeding
Heavy bleeding that occurs in association with the menstrual cycle — i.e., bleeding is timed with expected menstruation, arising from the uterine endometrium at predictable intervals. It may occur in both regular and irregular cycles.
- Corresponds to heavy bleeding at the time of menstruation
- Typically associated with ovulatory causes: structural lesions (fibroids, polyps, adenomyosis), coagulopathies, endometrial causes, or primary HMB
- The bleeding pattern is cyclical because it follows the endometrial shedding rhythm driven by ovarian hormones
Acyclical Heavy Menstrual Bleeding
Heavy bleeding that occurs outside or independent of the expected menstrual cycle — i.e., it is not timed with menstruation and has no predictable cyclic pattern.
- Also called intermenstrual bleeding in FIGO terminology: "bleeding episodes between normally timed menstrual periods"
- Raises concern for structural or malignant pathology: endometrial polyps, submucosal fibroids, endometrial hyperplasia, endometrial carcinoma, cervical pathology, or infection
- Anovulatory bleeding can also appear acyclical due to fluctuating, unpredictable oestrogen levels
How the Terms Relate
| Term | Cycle Timing | Volume | Pattern |
|---|---|---|---|
| Regular HMB (cyclical) | Predictable, ≤20-day variation | Excessive | Heavy bleeding at expected menstruation |
| Irregular HMB (cyclical) | Unpredictable, >20-day variation | Excessive | Heavy bleeding but cycle timing varies |
| Acyclical / Intermenstrual bleeding | Between periods | Variable (may be heavy) | No relationship to expected cycle |
| Prolonged menstrual bleeding | At menstruation | — | Periods lasting >8 days regularly |
Clinical Significance
- Cyclical HMB (regular) → think structural causes (PALM: Polyp, Adenomyosis, Leiomyoma, Malignancy/hyperplasia) or non-structural (COEIN: Coagulopathy, Ovulatory dysfunction, Endometrial, Iatrogenic, Not classified) — the PALM-COEIN classification (FIGO 2011)
- Acyclical/irregular HMB → always warrants investigation to exclude endometrial malignancy, especially in women ≥40 or with risk factors; anovulatory bleeding must also be excluded
- Anovulatory cycles may be regular in timing but more often are irregular, presenting as prolonged amenorrhoea followed by episodes of heavy bleeding due to fluctuating oestrogen without progesterone opposition — Tintinalli's Emergency Medicine
Sources: Tintinalli's Emergency Medicine (FIGO Terminology Table); Berek & Novak's Gynecology; Robbins & Kumar Basic Pathology
Definitions for superficial and deep dyspareunia
Finding Sources
Finding Sources
Reading File
Reading File
I have the precise definitions from the International Continence Society / ICS terminology (Haylen et al., 2010) as cited in Campbell Walsh Wein Urology. Here is the answer:
Definitions: Superficial and Deep Dyspareunia
Dyspareunia (General)
Complaint of persistent or recurrent pain or discomfort associated with attempted or complete vaginal penetration. — Campbell Walsh Wein Urology, 3-Volume Set (ICS/ICSM terminology, Haylen et al., 2010)
Dyspareunia is distinct from:
- Pain due to lack of vaginal lubrication (vaginal dryness)
- Vaginismus — recurrent or persistent spasm of the vaginal musculature that interferes with vaginal penetration (though these can coexist)
Under DSM-5, dyspareunia and vaginismus have been merged into a single diagnosis: Genito-Pelvic Pain/Penetration Disorder (GPPPD), reflecting their frequent co-occurrence and overlapping features. — Berek & Novak's Gynecology; Kaplan & Sadock's Synopsis of Psychiatry
Superficial (Introital) Dyspareunia
Complaint of pain or discomfort on vaginal entry or at the vaginal introitus.
- Also called entry dyspareunia or introital dyspareunia
- Pain is felt at or near the vaginal opening at the moment of attempted or initial penetration
- Anatomical zone: the vulva, vestibule, hymen, and vaginal introitus
Common causes:
| Cause | Notes |
|---|---|
| Vestibulodynia / vulvodynia | Most common cause in premenopausal women; localised provoked vulvar pain |
| Vulvovaginal atrophy (GSM) | Oestrogen deficiency → thin, dry mucosa; especially postmenopause |
| Vulvitis / dermatoses | Lichen sclerosus, lichen planus, contact dermatitis |
| Vaginismus | Pelvic floor muscle spasm at introitus |
| Infection | Vulvovaginal candidiasis, herpes simplex, Bartholin's cyst/abscess |
| Inadequate arousal/lubrication | Insufficient vaginal transudate |
| Episiotomy scar / obstetric trauma | Scar tissue at introitus |
| Congenital anomalies | Imperforate hymen remnants, vaginal septa |
Deep Dyspareunia
Complaint of pain or discomfort on deeper penetration (mid- or upper vagina).
- Pain is felt inside the pelvis during or after deep thrusting
- Anatomical zone: mid-vagina to supracervical / posterior fornix region
- Often described as a deep aching, pressure, or "hitting" sensation
Common causes:
| Cause | Notes |
|---|---|
| Endometriosis | Most important cause; particularly uterosacral ligament / pouch of Douglas involvement |
| Adenomyosis | Uterine enlargement and tenderness |
| Uterine fibroids (leiomyomas) | Especially posterior/deep fibroids |
| Pelvic inflammatory disease (PID) / chronic pelvic infection | Cervical motion tenderness, adnexal tenderness |
| Ovarian cysts or masses | Compression during deep thrusting |
| Pelvic adhesions | Post-surgical or post-infective |
| Retroverted uterus | Cervix/uterus displaced into path of penetration |
| Interstitial cystitis | Bladder-based pain triggered by penetration |
| Pelvic floor hypertonia | Elevated resting tone of deep pelvic floor muscles |
Additional Related Subtypes (ICS Classification)
| Term | Definition |
|---|---|
| Dyspareunia with penile vaginal movement | Pain caused by and dependent on penile movement (not just entry or depth) |
| Postcoital pain | Pain after intercourse — vaginal burning or pelvic pain following completion |
| Noncoital sexual pain | Pain induced by noncoital sexual stimulation |
Clinical Distinction Summary
| Feature | Superficial | Deep |
|---|---|---|
| Location of pain | Introitus / vaginal opening | Mid/upper vagina, pelvis |
| Timing | At entry / attempted penetration | With deep thrusting |
| Key causes | Vestibulodynia, atrophy, infection, vaginismus | Endometriosis, fibroids, PID, adhesions |
| Examination finding | Vulvar/vestibular tenderness (Q-tip test) | Cervical motion tenderness, adnexal/uterine tenderness |
Sources: Campbell Walsh Wein Urology 3-Volume Set (ICS terminology, Haylen et al., 2010); Berek & Novak's Gynecology; Smith and Tanagho's General Urology 19th Ed.; Kaplan & Sadock's Synopsis of Psychiatry
Time of delivery for pregnancy induced hypertension conditions and mode of delivery
Your credits are exhausted. Your daily credits reset tomorrow, or you can purchase a top-up for more.
This is a shared conversation. Sign in to Orris to start your own chat.