GOUT
Reading File
Finding Sources
Searching PubMed
"gout"[MeSH Terms] AND "management"[MeSH Terms]
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
Reading File
Searching Images
gout urate crystals joint tophi pathology
I now have comprehensive information. Let me compile a complete gout overview.
GOUT
Definition & Epidemiology
Gout is a crystal-induced arthropathy caused by the deposition of monosodium urate (MSU) crystals in joints and periarticular tissues, secondary to persistent hyperuricemia. It affects approximately 4% of adults in the United States (~5% of males, ~3% of females), with prevalence increasing with age. It is the most common form of inflammatory arthritis in adults.
Risk factors: obesity, hypertension, diabetes mellitus, thiazide diuretics, cyclosporin use, chronic kidney disease, purine-rich diet (red meats, shellfish, anchovies, beer, legumes).
Pathophysiology
Uric Acid Metabolism
Uric acid is the end product of purine catabolism. Purines are synthesized via:
- De novo pathway — synthesis from nonpurine precursors
- Salvage pathway — recycling of free purine bases (diet or DNA/RNA degradation)
Hyperuricemia (plasma urate > 6.8 mg/dL) is necessary but not sufficient for gout — only ~10% of hyperuricemic individuals develop clinical disease.
Causes of hyperuricemia:
| Mechanism | Examples |
|---|---|
| Reduced excretion (most common, ~90%) | Idiopathic, CKD, diuretics, cyclosporin |
| Overproduction | HGPRT deficiency (Lesch-Nyhan syndrome), tumor lysis syndrome, myeloproliferative disorders |
Lesch-Nyhan syndrome: Complete absence of hypoxanthine-guanine phosphoribosyltransferase (HGPRT) → inability to salvage purines → severe hyperuricemia + neurological manifestations (self-mutilation, intellectual disability, spasticity).
Crystal-Induced Inflammation
- MSU crystals precipitate in supersaturated extracellular fluid (especially in joints)
- Resident macrophages phagocytose crystals → activate NLRP3 inflammasome
- Inflammasome activates caspase-1 → IL-1β and TNF-α release
- IL-1β drives neutrophil recruitment to the synovium
- Neutrophils release cytokines, free radicals, and proteases; lysosomal enzyme leakage destroys cartilage
- Neutrophils lower local pH → further urate precipitation (self-amplifying cycle)
Clinical Stages
| Stage | Features |
|---|---|
| Asymptomatic hyperuricemia | Elevated urate, no symptoms; may last 20–30 years |
| Acute gouty arthritis | Sudden-onset monoarthritis; peak 1–2 days; self-limiting within 1–2 weeks |
| Intercritical (interval) gout | Asymptomatic periods between attacks; attacks become more frequent over time |
| Chronic tophaceous gout | Tophi form; permanent joint damage; polyarticular |
Acute Attack Features
- Podagra — first metatarsophalangeal (MTP) joint is the classic site (most common)
- Also: knee, ankle, tarsal joints, wrist, fingers
- Up to 20% have polyarticular involvement
- Severe pain (at rest and with movement), erythema, swelling, warmth — can mimic cellulitis
- Systemic fever possible → must rule out septic arthritis
Clinical Images
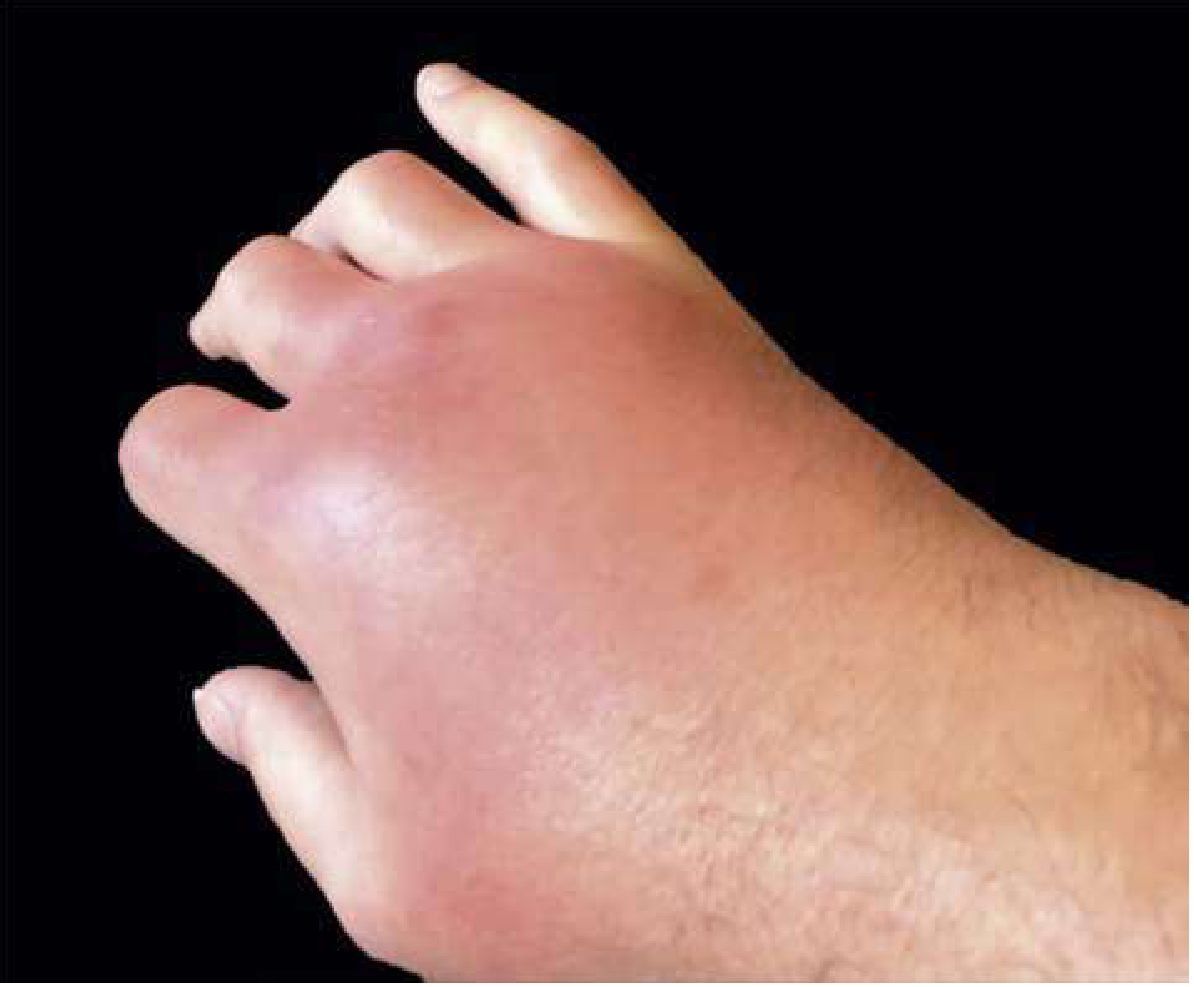
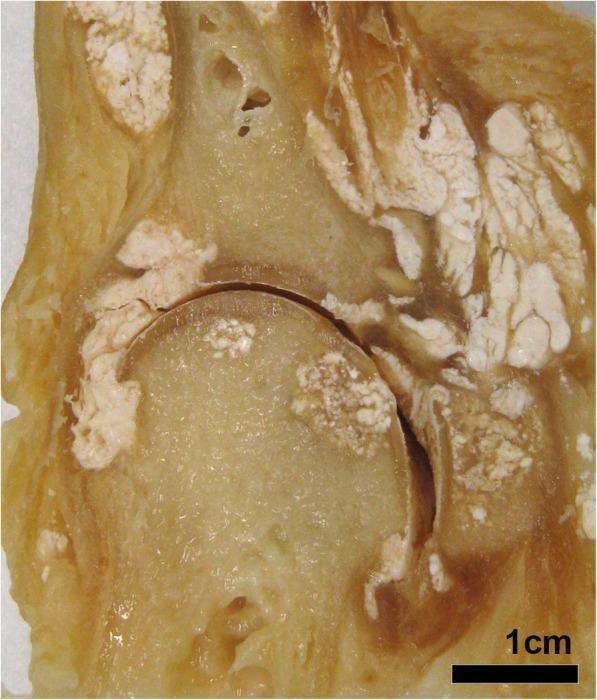
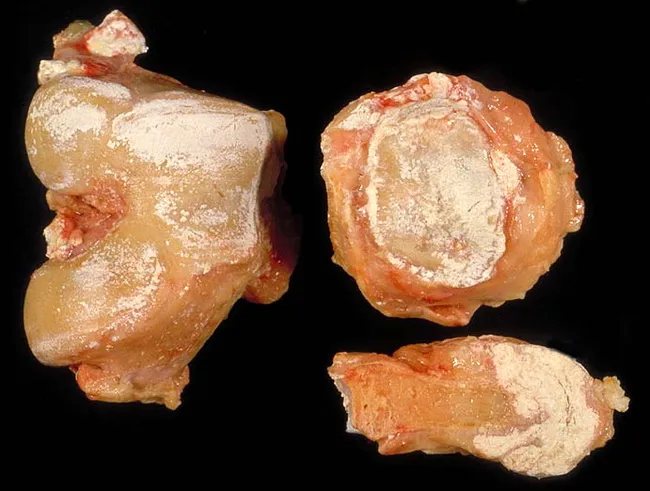
Tophi
Aggregates of monosodium urate crystals + inflammatory tissue forming in:
- Synovial membranes, periarticular tissues
- Bursae, joint space, subcutaneous tissue (especially helix of ear, Achilles tendon, olecranon bursa)
- Generally painless but cause bony erosion and joint deformity
- May ulcerate, discharging chalky-white material
Diagnosis
Synovial Fluid Analysis (Gold Standard)
- Negatively birefringent, needle-shaped crystals under polarized light microscopy
- Leukocytosis in synovial fluid (neutrophil predominance)
- Arthrocentesis mandatory for first attack; can treat empirically if established gout + not systemically unwell
Serum Uric Acid
- Unhelpful during acute attacks — can be normal during a flare and elevated without symptoms
- Useful for monitoring urate-lowering therapy (target < 6 mg/dL)
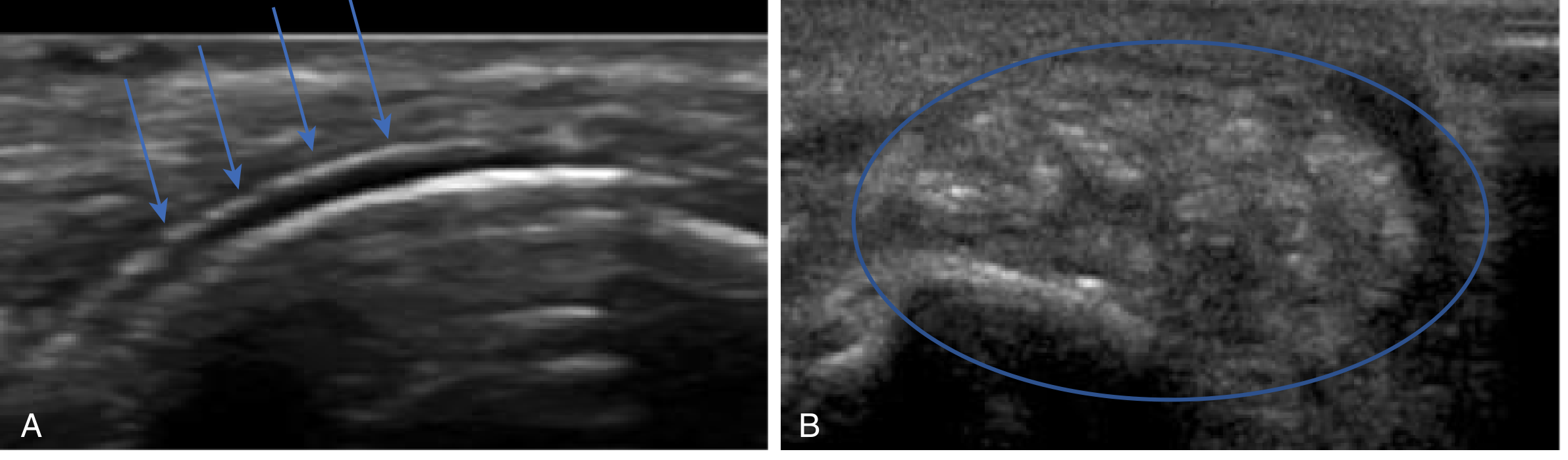
Imaging
- Plain X-ray: soft tissue swelling (acute); asymmetric sclerotic "punched-out" erosions outside the joint capsule with overhanging edges (chronic)
- Ultrasound: "double contour sign" (urate coating cartilage surface); "lump of sugar" appearance of tophi
- Dual-energy CT (DECT): color-coded urate deposits; highly specific
Renal Function
Always check — gout is associated with renal insufficiency, and most treatments are nephrotoxic.
Management
Acute Attack
| Drug | Notes |
|---|---|
| NSAIDs (first-line) | Indomethacin, naproxen, ibuprofen; start promptly, continue 24 hrs after resolution; avoid in PUD, GI bleeding, renal insufficiency |
| Colchicine (second-line) | Must be started within 36 hours of attack onset; inhibits NLRP3 inflammasome, tubulin polymerization, neutrophil migration; narrow therapeutic window; avoid in renal/hepatic insufficiency; GI side effects common |
| Corticosteroids | Oral (prednisone 40 mg/day × 5–7 days) or intra-articular; preferred when NSAIDs/colchicine contraindicated; do NOT combine oral steroids + NSAIDs |
| Combination therapy | Intra-articular steroid + colchicine or NSAID for severe/polyarticular disease |
| Non-pharmacologic | Ice, elevation, oral hydration, rest, avoid triggers |
Long-standing urate-lowering drugs (allopurinol, febuxostat, probenecid) should NOT be initiated during an acute attack but may be continued if already prescribed.
Urate-Lowering Therapy (ULT) — Chronic Gout
Indications:
- ≥ 2 attacks/year
- Chronic kidney disease
- Nephrolithiasis
- Presence of tophi
Target serum urate: < 6 mg/dL (symptomatic patients)
| Drug Class | Drug | Mechanism | Notes |
|---|---|---|---|
| Xanthine oxidase inhibitors (first-line) | Allopurinol | Inhibits xanthine oxidase → ↓ uric acid synthesis | Start at 100–200 mg/day; titrate slowly; adjust for renal function; risk of severe hypersensitivity (allopurinol hypersensitivity syndrome, especially in HLA-B*5801 carriers) |
| Febuxostat | Non-purine xanthine oxidase inhibitor | 40–80 mg/day; no renal dose adjustment; higher CV mortality than allopurinol in high-CV-risk patients (CARES trial) — use with caution | |
| Uricosuric agents | Probenecid | Blocks proximal tubule urate reabsorption | Second-line; avoid in nephrolithiasis |
| Uricase | Pegloticase | Converts urate → allantoin (more soluble) | IV; refractory tophaceous gout; FDA-approved 2010; anti-drug antibodies cause loss of efficacy + infusion reactions |
Key drug interaction: Allopurinol and febuxostat inhibit xanthine oxidase → block azathioprine metabolism → risk of severe myelosuppression. If combination required, reduce azathioprine dose by ~25% and monitor CBC closely. Alternatively, switch to mycophenolate.
Prophylaxis When Starting ULT
Initiation of ULT can precipitate an acute flare due to rapid urate level changes. Co-prescribe:
- Low-dose colchicine, or
- Low-dose NSAIDs, or
- Low-dose corticosteroids
Continue for at least 6 months after starting ULT.
Secondary Gout — Special Populations
Post-Transplant Gout
- Risk factors: pre-transplant hyperuricemia, CKD, diuretics, cyclosporin (> tacrolimus)
- Acute management: ice, rest + colchicine / increased corticosteroids / NSAIDs (NSAIDs used cautiously)
- ULT: allopurinol starting at 100–200 mg/day; dose-titrate every 4 weeks
Tumor Lysis Syndrome
- Rapid cell lysis after chemotherapy → massive purine release → secondary gout/urate nephropathy
- Prevented/treated with rasburicase (recombinant uricase) or allopurinol prophylaxis
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Septic arthritis | Fever, very high synovial WBC (>50,000), positive culture |
| Pseudogout (CPPD) | Calcium pyrophosphate crystals — positively birefringent, rhomboid-shaped; chondrocalcinosis on X-ray; associated with hemochromatosis, hypothyroidism, hyperparathyroidism |
| Rheumatoid arthritis | Symmetric, RF/anti-CCP positive, morning stiffness |
| Cellulitis | No crystals, responds to antibiotics, no joint effusion |
Key Drug: Colchicine — Mechanism Summary
Colchicine binds tubulin → depolymerizes microtubules → impairs:
- Neutrophil chemotaxis and adhesion
- NLRP3 inflammasome activation → ↓ IL-1β, IL-18
- Mast cell degranulation
Adverse effects: nausea, vomiting, diarrhea (dose-limiting); chronic use → myopathy, neutropenia, aplastic anemia, alopecia. Metabolized by CYP3A4 and P-glycoprotein — significant drug interactions (clarithromycin, itraconazole, verapamil, amiodarone increase colchicine toxicity).
Sources: Robbins & Kumar Basic Pathology; Rosen's Emergency Medicine; Goodman & Gilman's Pharmacological Basis of Therapeutics; Lippincott Illustrated Reviews: Pharmacology; Comprehensive Clinical Nephrology 7e
This is a shared conversation. Sign in to Orris to start your own chat.