First generation anti allergic drugs moa ,side effects, contraindications
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
First-Generation Antihistamines (H1-Receptor Blockers)
Examples
Diphenhydramine, chlorpheniramine, promethazine, hydroxyzine, cyproheptadine, meclizine, dimenhydrinate, doxylamine, brompheniramine
Mechanism of Action
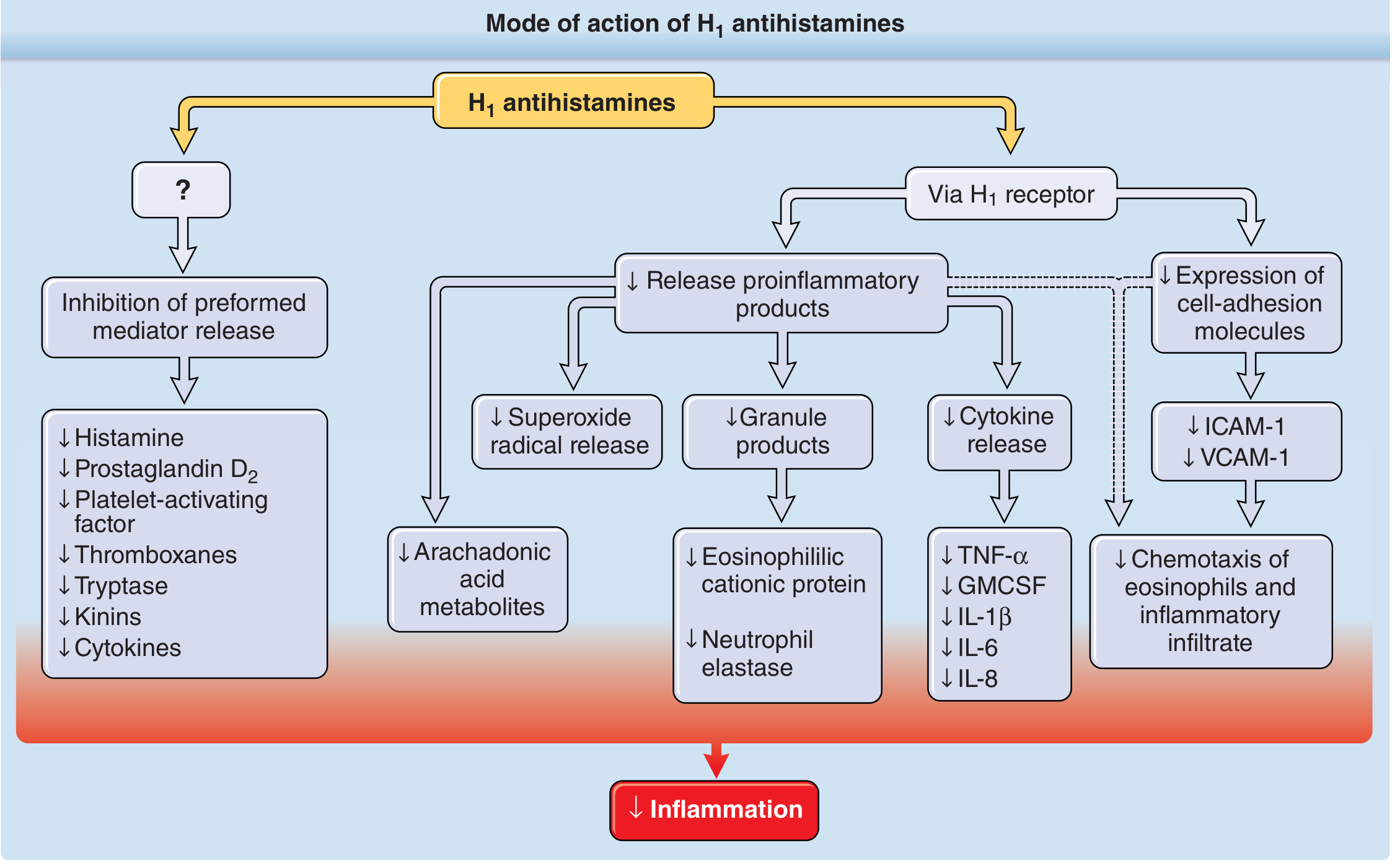
First-generation H1 antihistamines act as competitive, reversible antagonists at H1 receptors. They do not prevent histamine formation or release; instead, they block receptor-mediated responses in target tissues. They are far more effective at preventing allergic symptoms than reversing them once established.
Key MOA features (via H1 receptor):
- Inhibit histamine-induced vascular permeability, smooth muscle contraction, and pruritus
- Decrease proinflammatory cytokine release (TNF-α, IL-1β, IL-6, IL-8, GMCSF)
- Reduce expression of cell-adhesion molecules (ICAM-1, VCAM-1)
- Inhibit eosinophil chemotaxis
- Suppress release of preformed mediators (histamine, prostaglandin D₂, platelet-activating factor, tryptase, kinins)
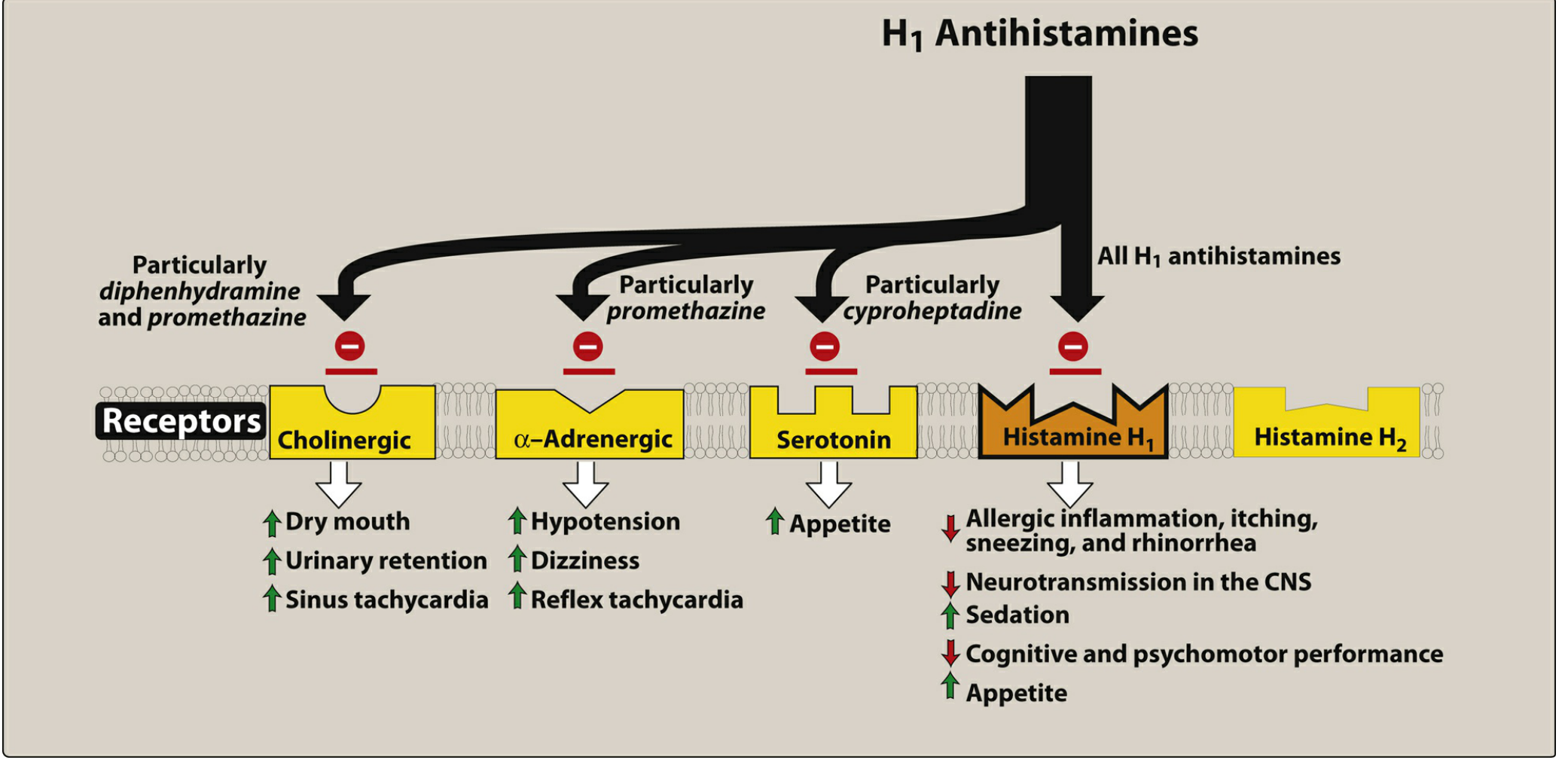
Beyond H1 blockade, first-generation agents (unlike second-generation) also block:
- Muscarinic/cholinergic receptors (especially diphenhydramine, promethazine) → anticholinergic effects
- α-Adrenergic receptors (especially promethazine) → hypotension
- Serotonin receptors (especially cyproheptadine) → appetite stimulation
CNS penetration: These drugs readily cross the blood-brain barrier (they are lipophilic), blocking central H1 receptors — this causes the characteristic sedation and cognitive impairment.
Pharmacokinetics
- Well absorbed orally; peak serum levels at 1–2 hours
- Duration of action: 4–6 hours (requires dosing q4–8h)
- Half-lives of brompheniramine, chlorpheniramine, and hydroxyzine exceed 20 hours in adults
- Distributed to all tissues including CNS
- Metabolized by hepatic CYP3A4 (glucuronide conjugates, excreted in urine)
- Wheal-and-flare suppression may persist up to 7 days after discontinuation
Therapeutic Uses
| Use | Notes |
|---|---|
| Allergic rhinitis, urticaria, angioedema | First-line for histamine-driven symptoms |
| Motion sickness / nausea | Diphenhydramine, dimenhydrinate, meclizine, promethazine — act on chemoreceptor trigger zone and vestibular pathways via central H1 and M1 blockade |
| Vertigo | Meclizine |
| Insomnia | Diphenhydramine, doxylamine (exploiting sedative side effect) |
| Pruritus (atopic dermatitis, chronic urticaria) | Effective in ~50% of chronic idiopathic urticaria |
| Anaphylaxis (adjunct) | Note: epinephrine remains the drug of choice |
| Serotonin syndrome (cyproheptadine) | Due to serotonin-antagonist properties |
Side Effects
CNS (most common and clinically significant)
- Sedation — the most prominent effect; results from central H1 blockade (CNS neurotransmission reduced)
- Cognitive and psychomotor impairment
- Dizziness, tinnitus, blurred vision
- Irritability, nervousness, insomnia (paradoxical, especially in children)
- Tremor
- Acute overdose: hallucinations, excitement, ataxia, convulsions → coma → cardiorespiratory collapse
Anticholinergic effects (due to muscarinic receptor blockade)
- Dry mouth, dry nasal mucosa
- Urinary retention and hesitancy
- Constipation
- Blurred vision
- Postural hypotension, dizziness
- Sinus tachycardia / reflex tachycardia
- QT prolongation, ventricular arrhythmias, torsades de pointes
- Erectile dysfunction
GI (uncommon)
- Anorexia, nausea, vomiting, epigastric distress, diarrhea/constipation
- (Taking with food reduces these)
Other
- Topical formulations → contact dermatitis (local hypersensitivity)
Drug Interactions
| Interaction | Result |
|---|---|
| CNS depressants (alcohol, benzodiazepines, opioids) | Potentiated sedation |
| MAO inhibitors (e.g., phenelzine) | MAOIs exacerbate both sedative AND anticholinergic effects — combination should be avoided |
| Cholinesterase inhibitors (donepezil, rivastigmine, galantamine) | First-generation antihistamines reduce effectiveness in Alzheimer's treatment |
Contraindications
| Contraindication | Reason |
|---|---|
| Narrow-angle glaucoma | Anticholinergic effects increase intraocular pressure |
| Concurrent MAOI use | Dangerously potentiated sedative/anticholinergic effects |
Risk Factors Requiring Caution (Not Absolute Contraindications)
- Prostatic hypertrophy — urinary retention risk (requires close monitoring)
- First trimester of pregnancy — potential teratogenicity risk
- Cardiac arrhythmias (particularly ventricular arrhythmias) — QT prolongation risk
- Occupations requiring wakefulness (drivers, pilots, heavy machinery operators)
- Children — paradoxical CNS stimulation; overdose particularly dangerous
— Lippincott Illustrated Reviews: Pharmacology, pp. 1309–1318
— Fitzpatrick's Dermatology, pp. 3482–3485
This is a shared conversation. Sign in to Orris to start your own chat.