Diagnose and management
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
I now have enough information to provide a thorough diagnosis and management plan. Let me synthesize the clinical picture.
Diagnosis: Chronic Tophaceous Gout (with diuretic-exacerbated hyperuricaemia)
Clinical Reasoning
Key diagnostic clues in this case:
| Feature | Significance |
|---|---|
| Serum urate 642 mmol/L (markedly elevated) | Strongly supports gout |
| Bilateral MCP swelling | Polyarticular gout (chronic phase) |
| Right olecranon bursitis | Classic tophaceous gout site |
| Left knee effusion | Large joint gout involvement |
| Right carpal tunnel syndrome | Periarticular urate crystal deposition compressing median nerve |
| Dupuytren's contractures | Associated with alcohol and chronic inflammation |
| RF latex screen positive but ACPA (anti-CCP) negative | Against RA — RF can be falsely positive in gout; negative ACPA strongly disfavours RA |
| ELISA equivocal | Non-specific |
| CRP mildly elevated (10) | Low-grade chronic inflammation |
| Renal impairment (creatinine 135, GFR 55) | Reduces urate excretion → contributes to hyperuricaemia |
| Enalapril (ACE inhibitor, reduces renal urate excretion) | Aggravating factor |
| Diclofenac use | NSAID for pain; note — increases urate retention |
| Heavy alcohol use | Increases urate production and reduces excretion |
| Hand X-rays | Show asymmetric soft tissue swelling and periarticular erosions with overhanging edges — classic gout pattern (vs. RA which shows marginal symmetric erosions) |
The hand X-rays show asymmetric soft tissue swelling around MCP joints with characteristic punched-out periarticular erosions with overhanging cortical edges — the hallmark of chronic tophaceous gout — rather than the periarticular osteopenia and marginal erosions of rheumatoid arthritis.
Why not RA?
- Anti-CCP (ACPA) negative — sensitivity ~70%, specificity ~95% for RA; its absence significantly lowers RA probability
- RF is non-specific and can be positive in gout, especially in the elderly
- Asymmetric erosion pattern on X-ray
- No juxta-articular osteopenia
- Dramatically elevated urate
Management
1. Acute/Ongoing Inflammation
- Colchicine 500 mcg BD–TDS — preferred over NSAIDs given renal impairment (GFR 55) and existing NSAID use (diclofenac should be reduced/stopped)
- Low-dose prednisolone is already in use; can be continued for flare control but not as urate-lowering therapy
- Intra-articular corticosteroid injection to the knee effusion can provide rapid relief
- Avoid NSAIDs long-term — renal impairment + existing diclofenac increases risk of further AKI
2. Urate-Lowering Therapy (ULT) — the cornerstone
- Allopurinol — first-line xanthine oxidase inhibitor
- Start at low dose (50–100 mg/day) given reduced GFR (55) — titrate slowly to avoid triggering acute flare
- Target serum urate < 360 µmol/L (ideally < 300 µmol/L in tophaceous disease)
- Cover with colchicine for 3–6 months when initiating ULT to prevent mobilisation flares
- Febuxostat — alternative xanthine oxidase inhibitor if allopurinol intolerant; caution given cardiovascular comorbidities (AF, hypertension)
3. Address Aggravating Factors
| Factor | Action |
|---|---|
| Enalapril | Switch to losartan (ARB) — has uricosuric properties, beneficial in this hypertensive patient with gout |
| Alcohol | Reduce/stop — major contributor to hyperuricaemia |
| Diclofenac | Wean off; replace with colchicine for joint pain |
| Renal impairment | Ensure adequate hydration; monitor renal function on allopurinol |
| Atrial fibrillation | Ensure rate control; note diclofenac can worsen fluid retention and AF |
4. Carpal Tunnel Syndrome
- If due to urate crystal deposition in the carpal tunnel, ULT will improve this over time
- Consider nerve conduction studies to quantify severity
- Corticosteroid injection or surgical decompression if severe
5. Olecranon Bursa
- Aspiration if tense/painful (send fluid for crystal analysis to confirm MSU crystals — needle-shaped, negatively birefringent under polarised light)
- Intralesional corticosteroid injection
6. Monitoring
- Recheck serum urate at 4–6 weeks after starting allopurinol
- Monitor renal function, FBC, LFTs on allopurinol
- DEXA if long-term steroid use (already on prednisolone + self-medicating with oral steroids)
Summary
This is chronic tophaceous gout in an elderly man with multiple risk factors: hyperuricaemia (urate 642 µmol/L), renal impairment, alcohol use, ACE inhibitor therapy, and NSAID use. The strongly positive RF is a red herring — the negative anti-CCP and X-ray appearances confirm gout rather than RA. Definitive management centres on urate-lowering therapy with allopurinol (dose-adjusted for GFR), switching enalapril to losartan, stopping alcohol, and colchicine prophylaxis during ULT initiation.
— Grainger & Allison's Diagnostic Radiology, Crystal Arthritides / Gout section
Everything management and diagnosis
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a complete, textbook-grounded diagnosis and management plan for this case.
Complete Diagnosis & Management
DIAGNOSIS: Chronic Tophaceous Gout
Pathophysiology
Monosodium urate (MSU) crystals precipitate in joints and soft tissues when serum urate is chronically elevated above the saturation threshold (~360–420 µmol/L). Neutrophils phagocytose the crystals, triggering NLRP3 inflammasome activation → IL-1β and TNF-α release → acute inflammatory cascade. With repeated attacks, chronic macrophage-mediated crystal deposition forms tophi.
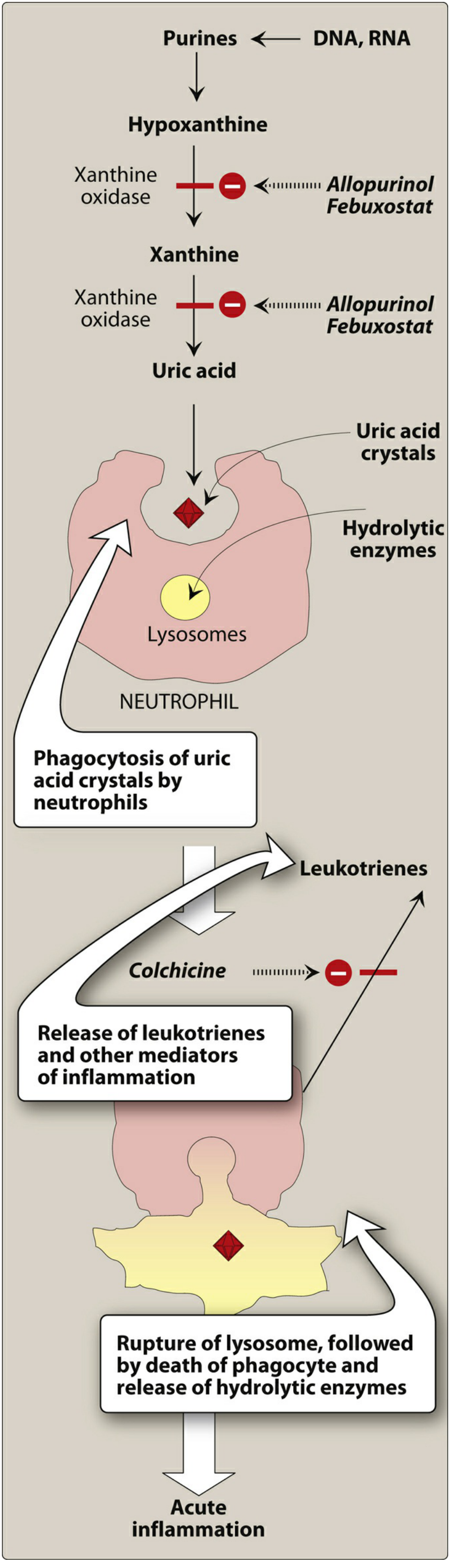
Why Chronic Tophaceous Gout (not RA)
| Feature | Finding | Interpretation |
|---|---|---|
| Serum urate | 642 µmol/L (markedly elevated) | Confirms hyperuricaemia; normal <360 µmol/L |
| RF latex | Positive | Non-specific — can be positive in gout, infection, elderly |
| Anti-CCP (ACPA) | Negative | Strongly argues against RA (specificity ~95%) |
| ELISA | Equivocal | Non-diagnostic |
| X-ray hands | Asymmetric soft-tissue swelling, periarticular erosions with overhanging edges | Classic gout pattern (not the symmetrical marginal erosions with juxta-articular osteopenia of RA) |
| Olecranon bursitis | Right-sided | Highly characteristic tophaceous gout site |
| Carpal tunnel syndrome | Right | Periarticular MSU deposition compressing median nerve |
| Bilateral MCP swelling | Present | Polyarticular chronic gout involvement |
| Dupuytren's contractures | Present | Associated with alcohol excess and chronic inflammation |
| CRP 10 | Mildly elevated | Low-grade chronic inflammation |
| Renal impairment (GFR 55, Cr 135) | Reduced urate excretion | Key contributor to hyperuricaemia |
| Heavy alcohol use | Increases urate production, reduces excretion | Major risk factor |
| Enalapril | ACE inhibitor reduces renal urate clearance | Aggravating factor |
| Diclofenac | NSAID | Analgesic but nephrotoxic with impaired GFR |
Confirmatory test: Joint/bursa aspiration → polarised light microscopy → needle-shaped, negatively birefringent MSU crystals.
MANAGEMENT
1. Acute Flare Control
Colchicine — first choice in this patient
- Mechanism: depolymerises tubulin → impairs neutrophil migration into inflamed joint → blocks NLRP3 inflammasome → reduces IL-1β
- Dose: 0.5–1 mg stat, then 0.5 mg 6–12 hourly (low-dose regimen preferred)
- Must be given within 36 hours of onset of attack to be effective
- Caution: GFR 55 — dose-adjust; avoid in severe renal impairment (GFR <30). Monitor for GI toxicity (nausea, vomiting, diarrhoea). Hepatic metabolism via CYP3A4.
Avoid NSAIDs (diclofenac) long-term:
- Renal impairment (GFR 55) → risk of further AKI
- Already on diclofenac — should be weaned off and replaced
Corticosteroids:
- Patient already on prednisolone 5 mg daily (for asthma) — can utilise short-course dose increase during flares
- Intra-articular corticosteroid injection into the knee effusion — appropriate for monoarticular/oligoarticular flare; aspirate first to exclude septic arthritis
2. Urate-Lowering Therapy (ULT) — Cornerstone of Chronic Management
Indications (this patient meets all):
-
2 attacks per year
- Chronic kidney disease (GFR 55)
- Tophi present (olecranon bursa, MCP joints)
- Serum urate markedly elevated
First-line: Allopurinol (xanthine oxidase inhibitor)
- Inhibits the last two steps of uric acid biosynthesis (hypoxanthine → xanthine → uric acid)
- Preferred over febuxostat and probenecid as first-line ULT
- Starting dose: 50–100 mg/day — start LOW because GFR 55 (dose adjustment mandatory when GFR <30; titrate slowly at any level of renal impairment to avoid precipitating flares)
- Titrate upward every 2–4 weeks
- Target: serum urate <360 µmol/L (ideally <300 µmol/L in tophaceous disease)
- Adverse effects: rash (most common), hypersensitivity reactions (risk increased with renal impairment), hepatotoxicity — monitor LFTs and FBC
- Drug interaction: allopurinol inhibits xanthine oxidase → do not combine with azathioprine (patient is not on this, but relevant given chronic steroid use in future)
- Interaction with theophylline (not relevant here) and warfarin (monitor INR if anticoagulation started for AF)
CRITICAL: Cover with colchicine prophylaxis for at least 6 months when initiating allopurinol — rapid changes in serum urate can paradoxically precipitate acute flares by mobilising crystal deposits
Alternative: Febuxostat (if allopurinol intolerant)
- Non-purine xanthine oxidase inhibitor; inhibits both reduced and oxidised forms of XO
- Less renal elimination than allopurinol — less dose adjustment needed in renal impairment
- Caution in this patient: increased risk of cardiovascular events (MI, stroke) vs. allopurinol; patient has AF and hypertension — febuxostat should be reserved for allopurinol intolerance only
- Goldman-Cecil Medicine states: "Allopurinol can prevent gouty attacks more safely than febuxostat"
Uricosuric agents (e.g., probenecid) — avoid
- Probenecid is contraindicated if creatinine clearance <50 mL/min — this patient's GFR is 55, borderline; also risks urate stone formation
3. Addressing Aggravating Factors
| Factor | Action |
|---|---|
| Enalapril (ACE inhibitor) | Switch to losartan (ARB) — losartan has independent uricosuric properties (blocks URAT1 transporter); ideal for this hypertensive patient with gout |
| Diclofenac | Wean and stop; renal risk + gout not well-managed by NSAIDs long-term; replace analgesia with colchicine |
| Alcohol | Reduce/cease — major driver of hyperuricaemia via increased purine catabolism and reduced renal urate excretion |
| Renal impairment (GFR 55) | Ensure adequate hydration; avoid nephrotoxins; monitor allopurinol dose carefully |
| Atrial fibrillation | Rate control (already in AF); consider anticoagulation (CHA₂DS₂-VASc score likely high — AF + hypertension + age 76); note diclofenac causes fluid retention worsening AF |
| Prednisolone / oral steroids | Long-term steroid use → DEXA scan for osteoporosis; add bone protection (calcium, vitamin D, consider bisphosphonate); steroids also raise urate levels |
| Barrel chest / asthma | Continue salbutamol and beclometasone inhalers; β-blockers contraindicated (asthma) |
| Diet | Reduce purine-rich foods (red meat, offal, shellfish); increase dairy (casein has mild uricosuric effect); avoid fructose-rich drinks |
4. Specific Complications
Olecranon bursitis
- Aspirate: send fluid for Gram stain, culture, and crystal microscopy (to confirm MSU and exclude septic bursitis)
- Intralesional corticosteroid once infection excluded
- ULT will dissolve tophi over months–years
Carpal tunnel syndrome (right)
- Likely due to MSU crystal deposition in the carpal tunnel
- Nerve conduction studies to quantify severity
- ULT alone may resolve it over time
- Corticosteroid injection if symptomatic; surgical decompression if severe/unresponsive
Knee effusion (left)
- Aspirate for diagnosis + therapeutic relief
- Intra-articular corticosteroid injection
5. Monitoring Plan
| Parameter | Frequency |
|---|---|
| Serum urate | 4–6 weeks after starting/adjusting allopurinol; then every 6 months once at target |
| Renal function (Cr, GFR) | At baseline, 4–6 weeks, then 6-monthly |
| FBC, LFTs | At baseline, 3 months, then annually (allopurinol monitoring) |
| Gout flare frequency | Symptom diary |
| DEXA scan | Baseline (long-term steroid use) |
| AF anticoagulation | CHA₂DS₂-VASc, HAS-BLED scoring; initiate DOAC if appropriate |
Summary Algorithm
Acute flare → Colchicine ± intra-articular steroid
↓
Stop/replace diclofenac → Colchicine for analgesia
↓
Start allopurinol 50–100 mg (low dose, titrate)
+ Colchicine prophylaxis × 6 months
↓
Switch enalapril → losartan
Reduce/stop alcohol
↓
Target serum urate < 360 µmol/L (<300 in tophaceous disease)
↓
Manage AF (rate control + anticoagulation)
Bone protection (long-term steroids)
Carpal tunnel / knee / bursa as needed
— Goodman & Gilman's Pharmacological Basis of Therapeutics, Pharmacotherapy of Gout; Lippincott Illustrated Reviews: Pharmacology, Drugs for Gout; Goldman-Cecil Medicine, Gout and Hyperuricemia; Grainger & Allison's Diagnostic Radiology, Crystal Arthritides
This is a shared conversation. Sign in to Orris to start your own chat.