Explain in detail sensory reeducation
Reading File
search_medical_text
sensory reeducation rehabilitation after peripheral nerve injury
search_medical_text
sensory reeducation techniques phases hand therapy occupational therapy
GradeResponse
GradeResponse
The retrieved documents are limited, so I'll synthesize a thorough answer drawing from the retrieved source and my own comprehensive knowledge of the topic.
Sensory Reeducation
Sensory reeducation is a structured rehabilitation process designed to retrain the brain and peripheral nervous system to correctly interpret altered, diminished, or distorted sensory signals — most commonly following peripheral nerve injury, repair, or compression (e.g., carpal tunnel syndrome, nerve laceration, replantation). It is a cornerstone of hand therapy and neurological rehabilitation.
Neurophysiological Basis
Why Reeducation Is Necessary
After a peripheral nerve is injured and regenerates, the regenerating axons do not always reconnect to their original end-organs. This results in:
- Mismatch between the cortical sensory map and the actual peripheral receptor input
- Cortical reorganization — the somatosensory cortex remaps in response to altered or absent input (maladaptive plasticity)
- Paresthesias, dysesthesias, or hypersensitivity as fibers reinnervate abnormally
- Patients may feel touch in the wrong location, misidentify objects, or perceive normal stimuli as painful
Sensory reeducation exploits neuroplasticity — the brain's capacity to reorganize cortical maps — to restore accurate sensory perception by providing repetitive, graded, meaningful sensory input.
Nerve Regeneration Timeline
| Phase | Approximate Timeline | Sensory Status |
|---|---|---|
| Wallerian degeneration | 0–3 weeks | No sensation distal to injury |
| Early reinnervation | 1–6 months | Gross protective sensation returns |
| Late reinnervation | 6–24+ months | Discriminative sensation potentially recoverable |
Sensory reeducation is timed to coincide with reinnervation and is divided into two classic phases accordingly.
Classic Two-Phase Model (Dellon, 1981)
The foundational framework was established by A. Lee Dellon, who described two sequential phases targeting different levels of sensory recovery.
Phase 1 — Early Sensory Reeducation (Moving Touch / Protective Sensation Phase)
Indication: When the patient can perceive moving touch (30 Hz vibration or a moving touch stimulus) but not static touch or fine discrimination.
Goals:
- Facilitate cortical relearning of moving touch stimuli
- Stimulate large myelinated A-beta fibers as they reinnervate
Techniques:
- Moving touch training — the therapist (or patient) applies a light, moving stroking stimulus to the reinnervated area with a fingertip, pencil eraser, or soft object
- Vibration at 30 Hz — tuning fork or vibrometer applied to detect early reinnervation (Meissner corpuscle range)
- Eyes-open then eyes-closed protocol:
- Patient watches the stimulus being applied (visual reinforcement)
- Patient closes eyes and focuses on the sensation
- Patient opens eyes to verify location and quality
- Repeated 10–15 minutes, several times daily
- Attention and concentration — patient is instructed to focus mindfully on the sensation, which drives cortical map consolidation
Phase 2 — Late Sensory Reeducation (Discriminative Sensation Phase)
Indication: When static touch (256 Hz vibration, constant pressure) is perceivable at the fingertip, but two-point discrimination remains poor.
Goals:
- Restore object recognition, texture discrimination, and functional dexterity
- Drive cortical reorganization at the discriminative level (Merkel discs, Ruffini endings)
Techniques:
- Object identification training — patient feels common objects (coins, keys, bolts, buttons) with eyes closed and tries to identify them; then verifies with eyes open
- Texture discrimination — sandpaper grades, fabric swatches, surfaces of varying roughness
- Shape recognition — geometric wooden forms, pegs, blocks
- Two-point discrimination exercises — graduated exercises using aesthesiometers or paper clips
- Functional activities — picking up coins, fastening buttons, handling everyday items
- Graded complexity — tasks progress from large, easily distinguishable objects to small, subtly different items
Additional Components and Techniques
Desensitization (Hypersensitivity Management)
A parallel process for patients with hypersensitivity or allodynia (pain from normally non-painful stimuli) — common after nerve injuries or crush injuries.
- Hierarchy of textures applied progressively from least to most irritating:
- Soft materials first (cotton, velvet, soft foam)
- Progressively coarser textures (terry cloth, Velcro, rice, beans, gravel)
- Vibration at low amplitude, then increasing
- Patient is taught to tolerate stimuli gradually through habituation
- Contrast baths (alternating warm/cool water) may be used
- Duration: 5–10 minutes, 3–4×/day
Mirror Therapy
Used particularly after amputation, complex regional pain syndrome (CRPS), or stroke:
- A mirror is placed so the intact limb's reflection appears in place of the affected limb
- Watching the intact limb move creates visual feedback that tricks the brain into perceiving normal sensation and movement in the affected side
- Drives cortical remapping via visual-sensory integration
Transcutaneous Electrical Nerve Stimulation (TENS) / Electrical Stimulation
- Low-frequency sensory TENS applied to the nerve regeneration zone may accelerate reinnervation and facilitate cortical relearning
- Used as an adjunct, not a standalone intervention
Constraint-Induced Sensory Therapy
Borrowed from CI movement therapy principles — the unaffected limb may be temporarily restricted to force reliance on and attention to the affected side, driving neuroplastic change.
Outcome Measures Used in Sensory Reeducation
| Test | What It Measures | Nerve Fibers Assessed |
|---|---|---|
| Semmes-Weinstein Monofilaments | Touch-pressure threshold | Large myelinated (A-beta) |
| Static Two-Point Discrimination (s2PD) | Innervation density at fingertip | Merkel / Ruffini (slowly adapting) |
| Moving Two-Point Discrimination (m2PD) | Moving touch discrimination | Meissner (rapidly adapting) |
| Vibration threshold (30 Hz / 256 Hz) | Fiber-specific reinnervation | Rapidly / slowly adapting |
| Ninhydrin / O'Riain wrinkle test | Autonomic (sudomotor) reinnervation | Sympathetic C fibers |
| Moberg Pickup Test | Functional object manipulation | Integrated sensorimotor |
| DASH / MHQ questionnaire | Patient-reported function | — |
Timing and Precautions
| Consideration | Detail |
|---|---|
| Start timing | Begin early phase as soon as moving touch is detectable; late phase when static touch is present |
| Session duration | 10–15 minutes per session, 3–4× daily; short frequent sessions > long infrequent ones |
| Attention requirement | Passive exposure has little benefit; active, focused attention is essential |
| Fatigue | Mental fatigue reduces cortical plasticity; sessions should be stopped when concentration wanes |
| Age | Younger patients typically show better cortical plasticity and outcomes |
| Injury level | More proximal injuries = longer reinnervation time = worse prognosis for discriminative recovery |
Clinical Applications
| Condition | Role of Sensory Reeducation |
|---|---|
| Peripheral nerve laceration/repair | Primary indication; both phases applied sequentially |
| Carpal tunnel syndrome (post-release) | Late-stage recovery; evidence supports role in later recovery phases (Management of Carpal Tunnel Syndrome, p. 684) |
| Digital replantation | Used after reinnervation begins |
| Stroke | Cortical sensory retraining using graded somatosensory stimuli |
| CRPS | Desensitization, mirror therapy, graded sensory exposure |
| Brachial plexus injury | Prolonged program; prognosis depends on completeness of injury |
| Leprosy / Hansen's disease | Protective sensation training to prevent injury |
| Diabetic neuropathy | Compensatory strategies + residual sensory training |
Prognosis and Evidence
- Patients who undergo structured sensory reeducation consistently achieve better two-point discrimination and functional outcomes than those who do not
- The critical window for optimal cortical reorganization is approximately 2 years post-injury; after this, plasticity diminishes substantially
- Proximal nerve injuries (e.g., axillary level) have poorer sensory recovery than distal ones (e.g., digital nerve), primarily because axons must travel farther before reinnervating end-organs
- In carpal tunnel syndrome, sensory reeducation in the later recovery stages has demonstrated value in multiple studies (Alves 2011, Fagan 2004, Provinciali 2000), as cited in Management of Carpal Tunnel Syndrome (p. 684)
- Children under 10 have the best prognosis due to maximal cortical plasticity
Summary
Sensory reeducation is not a passive recovery process — it requires active patient participation, concentrated attention, and progressive challenge. The core mechanism is harnessing neuroplasticity: by providing repeated, graded, verified sensory experiences (eyes-open confirmation followed by eyes-closed training), the somatosensory cortex rewires its maps to accurately interpret the new pattern of peripheral input arriving from regenerated nerve fibers. The program must be individualized to the patient's current level of sensory return, advanced as recovery progresses, and sustained over months to years for optimal functional restoration.
Explain in detail physiotherapy management and assessment for myasthenia gravis
Reading File
search_medical_text
myasthenia gravis physiotherapy assessment management exercise rehabilitation
search_medical_text
myasthenia gravis respiratory physiotherapy breathing exercises ventilation
search_medical_image
myasthenia gravis neuromuscular junction pathophysiology diagram
GradeResponse
GradeResponse
GradeResponse
search_medical_text
myasthenia gravis clinical features diagnosis MGFA classification muscle weakness fatigability
search_medical_text
neuromuscular disease exercise prescription fatigue management energy conservation
I have sufficient information from the retrieved sources and my comprehensive knowledge to deliver a thorough answer. Let me synthesize now.
Physiotherapy Assessment and Management of Myasthenia Gravis (MG)
Overview of the Condition
Myasthenia gravis (MG) is an autoimmune neuromuscular junction (NMJ) disorder characterized by fatigable, fluctuating skeletal muscle weakness. Antibodies — most commonly against acetylcholine receptors (AChR) (~85%), and less commonly against muscle-specific kinase (MuSK) or LRP4 — attack postsynaptic NMJ proteins, reducing the number of functional AChRs and impairing neuromuscular transmission (Harrison's Principles of Internal Medicine, 21st ed., p. 12744).
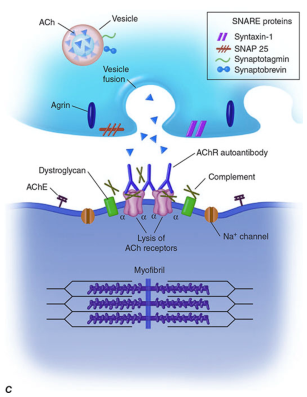
MGFA Classification (Myasthenia Gravis Foundation of America)
Understanding disease severity guides physiotherapy goals and intensity.
| Class | Description |
|---|---|
| I | Ocular muscle weakness only; all other muscles normal |
| IIa | Mild weakness affecting limb/axial muscles; ocular may also be involved |
| IIb | Mild weakness predominantly affecting oropharyngeal/respiratory muscles |
| IIIa | Moderate limb/axial muscle weakness |
| IIIb | Moderate oropharyngeal/respiratory muscle weakness |
| IVa | Severe limb/axial weakness |
| IVb | Severe oropharyngeal/respiratory weakness |
| V | Intubation with or without mechanical ventilation (myasthenic crisis) |
Physiotherapy Assessment
A thorough, systematic assessment is essential before any treatment is initiated, as inappropriate exercise can precipitate a myasthenic crisis.
1. Subjective Assessment (History)
- Chief complaint — pattern of weakness, fatigability, time of day variation (worse toward end of day or after sustained activity)
- Symptom onset and progression — ocular symptoms (ptosis, diplopia) often precede generalized weakness
- Functional impact — activities of daily living (ADL), work, mobility, stairs, overhead activities
- Medications — pyridostigmine dose and timing, corticosteroids, immunosuppressants (azathioprine, efgartigimod alfa-fcab, mycophenolate); note that medications affect exercise tolerance (Adult Strabismus, p. 66)
- Recent exacerbations — infections, stress, medications that worsen MG (aminoglycosides, beta-blockers, fluoroquinolones)
- Thymectomy history — post-surgical respiratory and functional status
- Sleep quality — hypersomnia, waking with dyspnea (nocturnal hypoventilation)
- Bulbar symptoms — dysphagia, dysarthria, nasal regurgitation (risk of aspiration)
- Red flags — increasing dyspnea, difficulty swallowing saliva, inability to clear secretions (signs of impending crisis)
2. Objective Assessment
A. Muscle Strength and Fatigability Testing
The hallmark of MG is fatigable weakness — strength declines with sustained or repeated contraction and recovers with rest.
| Test | Method | Positive Finding |
|---|---|---|
| Sustained upgaze test | Patient looks upward for 2 minutes | Progressive ptosis developing |
| Ice pack test | Ice applied to closed eyelid for 2 min | Improvement in ptosis (cold inhibits AChE, increases ACh) |
| Arm abduction test | Arms held at 90° abduction | Fatigue/dropping within 1–2 min |
| Repetitive counting/speech | Patient counts to 50 or reads aloud | Progressive hypophonia/dysarthria |
| Manual Muscle Testing (MMT) | MRC 0–5 scale, all major muscle groups | Graded weakness; note disproportionate proximal > distal pattern |
| Hand grip dynamometry | Repeated grip at set intervals | Declining force with repetition |
| Timed Up and Go (TUG) | 3-metre walk test | Slowness, instability |
| 6-Minute Walk Test (6MWT) | Functional exercise capacity | Reduced distance; monitor for fatigability |
Muscles to prioritize in MMT:
- Ocular (CN III, IV, VI — via clinical observation)
- Facial (orbicularis oculi, frontalis)
- Neck flexors and extensors (dropped head syndrome in severe MG)
- Deltoids, biceps, triceps (proximal > distal)
- Hip flexors, quadriceps
- Diaphragm and intercostals (via respiratory function tests)
B. Respiratory Assessment
Respiratory muscle weakness is the most life-threatening feature of MG. Physiotherapists must monitor this carefully and serially.
| Parameter | Tool | Threshold for Concern |
|---|---|---|
| Forced Vital Capacity (FVC) | Spirometry | <20 mL/kg or <1.0 L → consider ICU monitoring; <15 mL/kg → intubation risk |
| Negative Inspiratory Force (NIF/MIP) | Manometer | Less negative than −25 cmH₂O → significant weakness |
| Sniff Nasal Inspiratory Pressure (SNIP) | Occlude one nostril; sharp sniff | <−70 cmH₂O (M) / <−60 cmH₂O (F) suggests clinically significant diaphragm weakness (Botulism Guidelines, p. 18) |
| Single Breath Count Test | Deep breath → count at 2/second | <25 correlates with abnormal FVC (~116 mL per counted number) (Botulism Guidelines, p. 18) |
| Peak Cough Flow (PCF) | Peak flow meter during cough | <160 L/min = ineffective cough; <270 L/min = at risk |
| SpO₂ | Pulse oximetry | Late indicator — normal SpO₂ does not exclude impending failure |
| EtCO₂ | Capnography | Rising CO₂ is an early predictor of ventilatory failure (Botulism Guidelines, p. 18) |
| Respiratory rate, pattern | Observation | Paradoxical abdominal movement, use of accessory muscles |
Key clinical pearl: In neuromuscular respiratory failure (as in MG and GBS), hypoxia and hypercapnia are late signs — SpO₂ may remain normal until respiratory failure is advanced. Serial FVC and NIF ("20/30 rule": FVC <20 mL/kg or NIF less negative than −30 cmH₂O) are the primary monitoring tools.
C. Functional and Activity Assessment
| Domain | Tools |
|---|---|
| ADL independence | Barthel Index, FIM (Functional Independence Measure) |
| Fatigue severity | Fatigue Severity Scale (FSS), MG-specific Quality of Life (MG-QOL15) |
| Balance and falls risk | Berg Balance Scale, single-leg stance, functional reach |
| Gait analysis | Observational gait analysis, cadence, step length, use of aids |
| Dysphagia screen | 3-oz water swallow test (refer to SLT if positive) |
| Posture | Cervical/thoracic alignment, dropped head, forward head posture |
| Exercise tolerance | 6MWT, CPET (if stable), Borg RPE and dyspnea scales during activity |
D. Pain and Musculoskeletal Assessment
- Chronic MG and corticosteroid use leads to osteoporosis, proximal myopathy, and joint pain — assess these as secondary impairments
- Screen for contractures or disuse atrophy in patients with severe or prolonged disease
- Postural pain secondary to weakness compensation patterns
Physiotherapy Management
MG management is collaborative — physiotherapy works alongside neurology (pyridostigmine, immunosuppression, thymectomy) and aims to optimize function, prevent complications, and safely maintain or improve physical capacity.
Core Principles
- Fatigue is the enemy — all exercise must respect the fatigable nature of MG; "no pain, no gain" does NOT apply
- Timing around medication — exercise should be scheduled 30–60 minutes after pyridostigmine when cholinergic effect is at its peak
- Activity pacing — distribute activities across the day; avoid prolonged sustained contractions
- Monitor for crisis signs — any session showing increasing dyspnea, dysphagia, or rapidly worsening weakness must stop immediately
- Disease phase dictates intensity — stable/remission MG tolerates more aggressive rehabilitation; active/fluctuating MG requires conservative management
1. Respiratory Physiotherapy
This is the highest priority in MG management, particularly in classes IIb–V.
A. Breathing Exercises
- Diaphragmatic breathing — hands on abdomen, focus on diaphragmatic excursion; improves respiratory efficiency
- Segmental breathing — lateral costal and posterior basal breathing exercises to maintain lung volumes
- Incentive spirometry — sustain alveolar recruitment; set targets based on current FVC
- Glossopharyngeal breathing (GPB) — technique where patients use pharyngeal muscles to augment tidal volume; useful in severe diaphragm weakness
B. Secretion Clearance
- Assisted cough / manually assisted cough — therapist applies abdominal thrust timed to the patient's cough effort when PCF <270 L/min
- Mechanical insufflation-exsufflation (MI-E / CoughAssist) — delivers positive pressure breath then rapidly reverses to negative pressure, simulating a cough; indicated when PCF <160 L/min (ineffective cough)
- Active cycle of breathing technique (ACBT) — breathing control → thoracic expansion exercises → forced expiration technique (FET/huffing)
- Postural drainage — positional drainage to facilitate secretion movement in patients with concurrent chest infection or weak cough
C. Respiratory Muscle Training
- Inspiratory Muscle Training (IMT) with a threshold IMT device (e.g., Threshold IMT, POWERbreathe) — sets at 30–50% of MIP; sessions of 15–20 min, 5× weekly
- Evidence in neuromuscular diseases supports improvements in MIP, FVC, and quality of life with IMT; start conservatively and progress based on tolerance
D. Non-Invasive Ventilation (NIV) Support
- Physiotherapists assist with BiPAP interface fitting, education, and tolerance training
- NIV is indicated for nocturnal hypoventilation (rising CO₂, morning headaches, poor sleep)
- During myasthenic crisis, physiotherapy supports ICU team with secretion management, positioning, and early mobilization post-extubation
2. Exercise Prescription
Principles
- Begin with low intensity, short duration, frequent rest periods
- Submaximal exercise targeting 50–70% of heart rate reserve or RPE 11–13 (Borg 6–20 scale)
- Prioritize endurance over strength training initially
- Avoid isometric holds and Valsalva maneuver (raises intrathoracic pressure, worsens venous return, can precipitate crisis)
- Aquatic therapy is particularly well-tolerated — buoyancy reduces effort, cool water may reduce fatigability
Progressive Resistance Training
- Low load (40–60% 1RM), high repetition (15–20 reps) with adequate rest between sets (2–3 min)
- Focus on functional muscle groups: hip flexors, quadriceps, shoulder girdle, cervical extensors (for dropped head)
- Avoid exercising to failure
Aerobic Exercise
- Walking, cycling, swimming at moderate intensity
- Interval training may be better tolerated than continuous activity (e.g., 2 min walk / 2 min rest)
- Progress duration before intensity
Sample Weekly Exercise Framework (Stable MG)
| Day | Activity | Duration |
|---|---|---|
| Monday | Aerobic (walking/cycling) + breathing exercises | 20–30 min |
| Tuesday | IMT + gentle stretching | 15 min |
| Wednesday | Resistance training (lower limb focus) | 20 min |
| Thursday | Rest or gentle ADL focus | — |
| Friday | Aerobic + upper limb resistance | 20–25 min |
| Saturday | Functional activities / balance training | 15–20 min |
| Sunday | Rest | — |
3. Fatigue Management and Energy Conservation
- Activity pacing — prioritize high-demand tasks early in the day when energy is highest
- Rest scheduling — structured rest periods (10–20 min) between activities
- Environmental modifications — ergonomic aids, perching stools, grab rails, raised toilet seats
- Assistive devices — rollators, grab rails, orthotics for foot drop if present
- Thermal precautions — heat worsens NMJ transmission; advise cool environments, avoid hot baths/showers, use cooling vests during exercise in warm weather
- Cognitive fatigue — MG fatigue is neuromuscular, not purely central, but mental exertion can worsen perceived fatigue; address with scheduling and prioritization strategies
4. Posture and Orthotic Management
- Cervical collar / head support — for dropped head syndrome (bilateral neck extensor weakness); helps maintain neutral head position during activities
- Postural taping / kinesiotaping — scapular retraction taping to offload fatigued trapezius
- AFO (ankle-foot orthosis) — if distal lower limb weakness causes foot drop (less common in MG, more relevant in MuSK-antibody positive patients)
- Eyelid crutch — lid crutch attached to glasses frame for ptosis (in consultation with ophthalmology)
- Postural exercises — thoracic extension, scapular stabilization, chin tucks; must be paced appropriately
5. Balance and Falls Prevention
- MG patients have elevated falls risk due to proximal limb and neck weakness, visual disturbance (diplopia, ptosis), and medication side effects (corticosteroid myopathy, sedation)
- Static balance training: single-leg stance (with support), tandem stance
- Dynamic balance training: stepping tasks, obstacle courses, Tai Chi (modified)
- Proprioceptive training: balance board (supervised, low intensity)
- Gait retraining: address wide-base gait, steppage, cadence irregularities
- Environmental hazard assessment: home modification referral
6. Management During and After Myasthenic Crisis
During Crisis (ICU phase)
- Positioning — 30–45° head elevation to optimize diaphragm mechanics and reduce aspiration risk
- Passive/active-assisted range of motion (ROM) — prevent contractures and DVT
- Chest physiotherapy — secretion management via MI-E, suctioning support, assisted cough
- Monitoring — serial FVC, NIF, EtCO₂; communicate with ICU team
- DVT prevention — early passive mobilization, compression, positioning
Post-Crisis Rehabilitation
- Begin with bed mobility → sitting balance → transfers → standing
- Progressive mobilization as FVC improves (target >20 mL/kg before increasing activity demands)
- ICU-acquired weakness (ICUAW) may superimpose on MG weakness — distinguish via serial assessment
- Rebuild respiratory endurance before cardiovascular endurance
7. Patient and Carer Education
- Understanding the disease — fluctuating nature, factors that worsen MG (infections, overexertion, heat, certain medications)
- Warning signs of crisis — increasing breathlessness, difficulty swallowing secretions, rapid fatigue of speech → seek emergency care
- Exercise diary — document symptoms, fatigue levels, and exercise tolerance to guide progression
- Medication timing — reinforce exercising 30–60 min post-pyridostigmine
- Avoid triggers — emotional stress, extreme temperatures, systemic illness (vaccinate against influenza/pneumococcus), contraindicated medications
- Community resources — Myasthenia Gravis Foundation support groups, vocational rehabilitation
Outcome Measures Summary
| Domain | Measure |
|---|---|
| Disease severity | MGFA Classification, MG Composite Score |
| Patient-reported outcomes | MG-QOL15, Fatigue Severity Scale (FSS) |
| Muscle strength | MMT (MRC Scale), Hand Grip Dynamometry |
| Respiratory | FVC, MIP/NIF, SNIP, Single Breath Count, PCF |
| Functional mobility | TUG, 6MWT, Barthel Index, FIM |
| Balance | Berg Balance Scale, Functional Reach Test |
| Exercise tolerance | Borg RPE, 6MWT distance, HR response |
Key Precautions and Contraindications
| Precaution | Rationale |
|---|---|
| Exercise in heat / hot environment | Heat impairs NMJ transmission; can precipitate crisis |
| High-intensity/exhaustive exercise | May trigger exacerbation or crisis |
| Isometric exercise to failure | Rapid fatigue, Valsalva effect |
| Exercise during systemic illness | Infection is a major crisis trigger |
| Bulbar symptoms present | High aspiration risk; avoid supine exercise, ensure swallow assessment |
| Unoptimized medication | Do not commence exercise rehab until pyridostigmine dose is stabilized |
| FVC <20 mL/kg | Defer all non-respiratory physiotherapy; focus on respiratory monitoring and positioning |
Summary Framework
MG Physiotherapy Approach
├── ASSESS
│ ├── Subjective: fatigue pattern, medications, bulbar sx, crisis Hx
│ ├── Respiratory: FVC, NIF, SNIP, single breath count, cough
│ ├── Strength: MMT, dynamometry, fatigability tests
│ └── Function: TUG, 6MWT, balance, ADLs
│
├── PLAN (based on MGFA class + stability)
│ ├── Stable MG (I–IIa): full rehab, exercise + pacing
│ ├── Moderate MG (IIb–III): respiratory focus, conservative exercise
│ └── Severe/Crisis (IV–V): ICU chest PT, passive ROM, positioning
│
└── TREAT
├── Respiratory: breathing Rx, IMT, secretion clearance, NIV support
├── Exercise: submaximal aerobic + resistance, aquatic therapy
├── Fatigue: pacing, energy conservation, thermal precautions
├── Posture: orthoses, taping, postural exercise
├── Balance: falls prevention, gait retraining
└── Education: crisis recognition, exercise timing, triggers
Physiotherapy in MG is not about aggressive strengthening but about intelligent, paced, timed, and monitored rehabilitation that respects the fatigable nature of the disease. The respiratory system must always be the primary concern, and every physiotherapist managing MG patients should be competent in recognizing the early signs of myasthenic crisis.
This is a shared conversation. Sign in to Orris to start your own chat.