Malaria
Reading File
Asking for Preferences
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Here is a comprehensive overview of malaria — its epidemiology, causative agents, life cycle, and pathophysiology.
Malaria — Overview & Pathophysiology
Epidemiology
Malaria is one of the most serious infectious diseases in the world. The WHO estimated over 200 million cases and 600,000+ deaths annually (2020 data), with the burden concentrated in sub-Saharan Africa and Asia. More than 41% of the world's population lives in endemic areas. Thanks to global air travel, cases are now seen worldwide in non-endemic countries, particularly among returning travelers who failed to take prophylaxis. — Robbins & Kumar Basic Pathology; Rosen's Emergency Medicine
Causative Agents
Five Plasmodium species infect humans:
| Species | Disease | Severity |
|---|---|---|
| P. falciparum | Falciparum (tertian) malaria | Most dangerous — high fatality |
| P. vivax | Benign tertian malaria | Mild; can relapse (hypnozoites) |
| P. ovale | Ovale malaria | Mild; can relapse (hypnozoites) |
| P. malariae | Quartan malaria | Mild; chronic low-grade infection |
| P. knowlesi | Knowlesi malaria | Mild in most cases |
Vector: Bite of the female Anopheles mosquito. Humans are the only natural reservoir. — Robbins & Kumar Basic Pathology
Other (rare) routes of transmission include blood transfusion, contaminated needles, organ transplantation, perinatal transmission, and "airport malaria" (infected mosquitoes transported by aircraft).
Life Cycle of P. falciparum
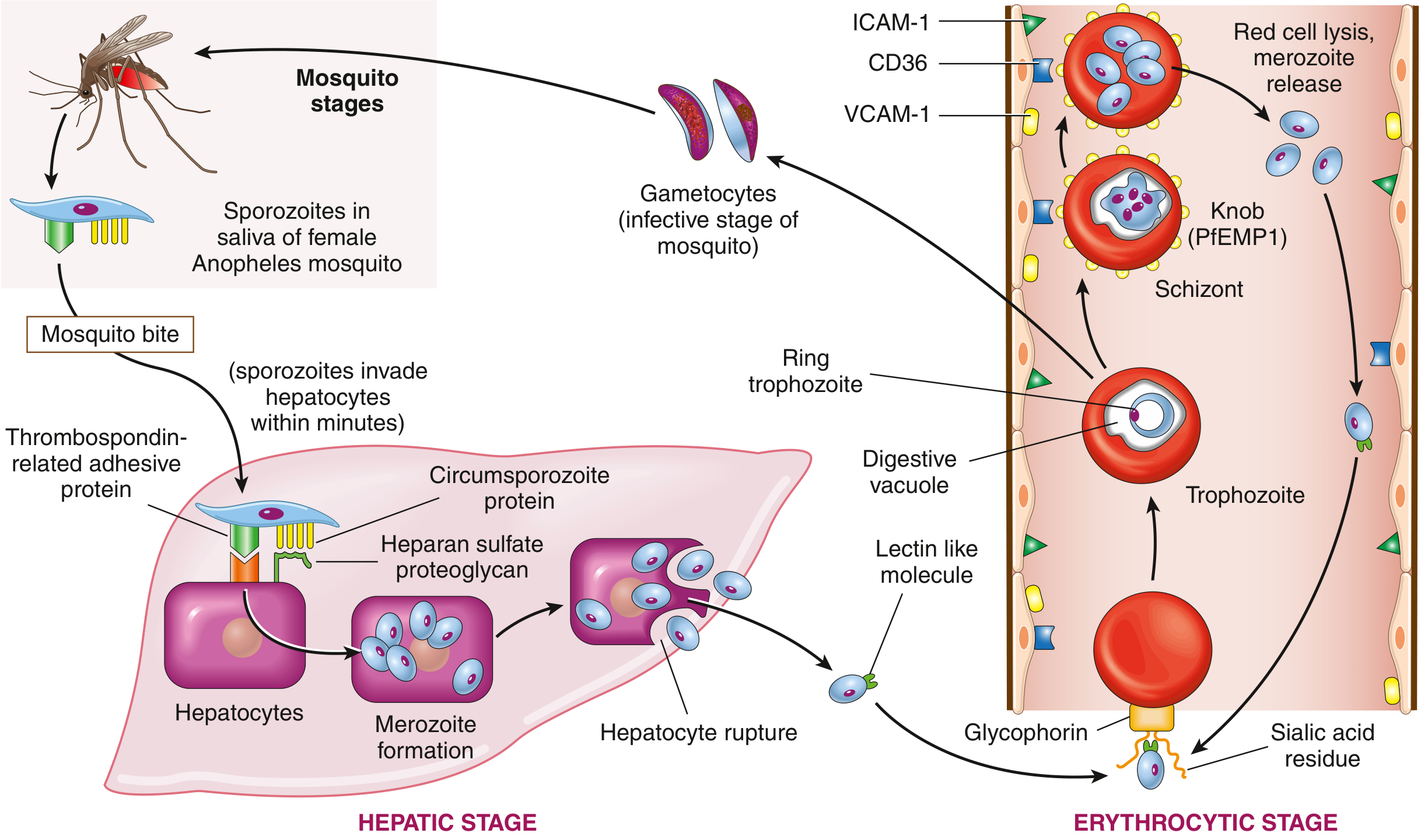
FIG. 10.8 — Life cycle of P. falciparum. — Robbins & Kumar Basic Pathology
1. Hepatic Stage (Pre-erythrocytic)
- A feeding Anopheles mosquito injects sporozoites into the bloodstream.
- Sporozoites travel to the liver within minutes. Two surface proteins — thrombospondin-related adhesive protein and circumsporozoite protein — bind to heparan sulfate proteoglycans on hepatocytes.
- Inside hepatocytes, sporozoites differentiate into merozoites over 1–4 weeks.
- Infected hepatocytes rupture and release merozoites into the blood.
- P. vivax and P. ovale can form dormant hypnozoites in the liver, causing relapses weeks to months later.
2. Erythrocytic Stage
- A lectin-like molecule on the merozoite surface binds to sialylated glycophorin (a red cell transmembrane protein), allowing the merozoite to invaginate into the RBC within a "digestive vacuole."
- The intra-erythrocytic organism matures through: ring trophozoite → mature trophozoite → schizont → merozoites.
- Upon RBC lysis, merozoites are released and infect fresh red cells — perpetuating the cycle.
- The periodicity of this cycle (every 48 hours for P. falciparum/vivax/ovale, every 72 hours for P. malariae) drives the characteristic cyclical fevers.
3. Sexual Stage (Return to Mosquito)
- Some trophozoites differentiate into gametocytes rather than schizonts.
- When ingested by a feeding mosquito, gametocytes undergo sexual reproduction in the midgut → sporozoites migrate to salivary glands → cycle restarts.
Key Pathophysiologic Mechanisms
Cytoadherence & Sequestration (P. falciparum)
- Maturing schizonts express PfEMP1 (Plasmodium falciparum erythrocyte membrane protein 1), which forms knob-like protrusions on the RBC surface.
- PfEMP1 binds endothelial adhesion molecules — ICAM-1, VCAM-1, and CD36 — causing parasitized RBCs to adhere to capillary walls and sequester in microvascular beds.
- This cytoadherence is the primary reason P. falciparum is far more lethal than other species: it causes microvascular obstruction, tissue hypoxia, and organ dysfunction (brain → cerebral malaria; kidney → acute kidney injury; placenta → pregnancy complications).
Hemolytic Anemia
- Repeated cycles of RBC rupture cause hemolytic anemia, which worsens with high-grade parasitemia.
- Splenic clearance of both parasitized and innocent bystander RBCs amplifies the anemia.
- Massive intravascular hemolysis in severe P. falciparum can cause blackwater fever (hemoglobinuria → dark urine → renal failure).
Immune-Mediated Injury
- Rupture of schizonts releases merozoite antigens, hemozoin (malaria pigment), and GPI anchors → triggers TNF-α, IL-1, and IL-6 release from macrophages.
- This cytokine surge is responsible for the fever, rigors, headache, and systemic symptoms.
- TNF-α also contributes to cerebral malaria by upregulating ICAM-1 on brain endothelium, enhancing sequestration.
Splenomegaly
- Chronic antigen stimulation and removal of parasitized RBCs lead to progressive splenomegaly, which can be massive in endemic populations ("tropical splenomegaly").
Summary Table
| Feature | P. falciparum | P. vivax / ovale |
|---|---|---|
| Fever periodicity | 48 h (tertian) | 48 h (tertian) |
| RBC preference | All ages | Reticulocytes |
| Cytoadherence (PfEMP1) | Yes | No |
| Hypnozoites / relapse | No | Yes |
| Severity | Severe/fatal | Mild |
| Cerebral malaria | Yes | Rare |
Sources: Robbins & Kumar Basic Pathology (9780323790185); Rosen's Emergency Medicine (9780323757898)
This is a shared conversation. Sign in to Orris to start your own chat.