Hepatites b in detail
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Reading File
Finding Sources
Reading File
Reading File
Now I have comprehensive information to provide a detailed answer on Hepatitis B. Let me compile everything.
Hepatitis B — Comprehensive Overview
1. Virology
Hepatitis B virus (HBV) belongs to the Hepadnaviridae family — DNA viruses that selectively infect hepatocytes. The viral particle (Dane particle) has:
- Partially double-stranded, circular DNA — a shorter (+) strand and a longer (−) strand
- Four genes:
- C gene — encodes the core protein (HBcAg)
- S gene — encodes surface antigen (HBsAg); has three open reading frames producing large, middle, and small surface proteins
- P gene — encodes DNA polymerase / reverse transcriptase
- X gene — protein product has unclear but likely oncogenic roles
- Four serotypes: adr, adw, ayr, ayw
- Eight genotypes (A–H) with different geographic distributions, virulences, and treatment responses
Replication is unique: viral mRNA is longer than the coding DNA, and replication requires reverse transcriptase (similar to retroviruses). This makes HBV susceptible to reverse transcriptase inhibitors.
Viral particles enter hepatocytes via receptor-mediated endocytosis and are transported to the nucleus by chaperone proteins. Up to 10¹⁰ particles/mL may circulate during active viremia.
2. Epidemiology
- >350 million chronic carriers worldwide; ~1 million in the United States
- ~600,000 deaths/year from HBV-related liver disease and hepatocellular carcinoma (HCC)
- 25% of carriers develop chronic active hepatitis
- 36% prevalence in HIV-infected persons (2008, US data)
- No seasonal trend; no age-group predilection — but definite high-risk groups exist
High-risk groups:
- Parenteral drug users
- Hemodialysis patients and staff (up to 50% become chronic carriers if infected)
- Health care workers (surgeons, pathologists, lab staff, blood bank personnel)
- Sexually promiscuous individuals / men who have sex with men
- Multiply transfused and organ transplant patients
- Newborns of HBsAg-positive mothers
- Institutionalized persons
- Household and sexual contacts of carriers
3. Transmission
- Primary routes: blood/body fluids, sexual contact, perinatal (vertical) transmission
- HBsAg detectable in: serum, saliva, semen, vaginal secretions, menstrual fluid, nasopharyngeal secretions
- Fecal–oral route is NOT documented
- Perinatal: infected mother → newborn during delivery; also household infant contacts
- Healthcare: improperly sterilized needles/syringes, tattooing, ear piercing
- Virus is resistant to drying — all body fluids from infected patients should be assumed infectious
- Mandatory donor screening (HBsAg, anti-HBc, HBV DNA) has substantially reduced transfusion-associated hepatitis
4. Serological Markers & Disease Course
The timeline below shows the sequence of viral antigens and antibodies:
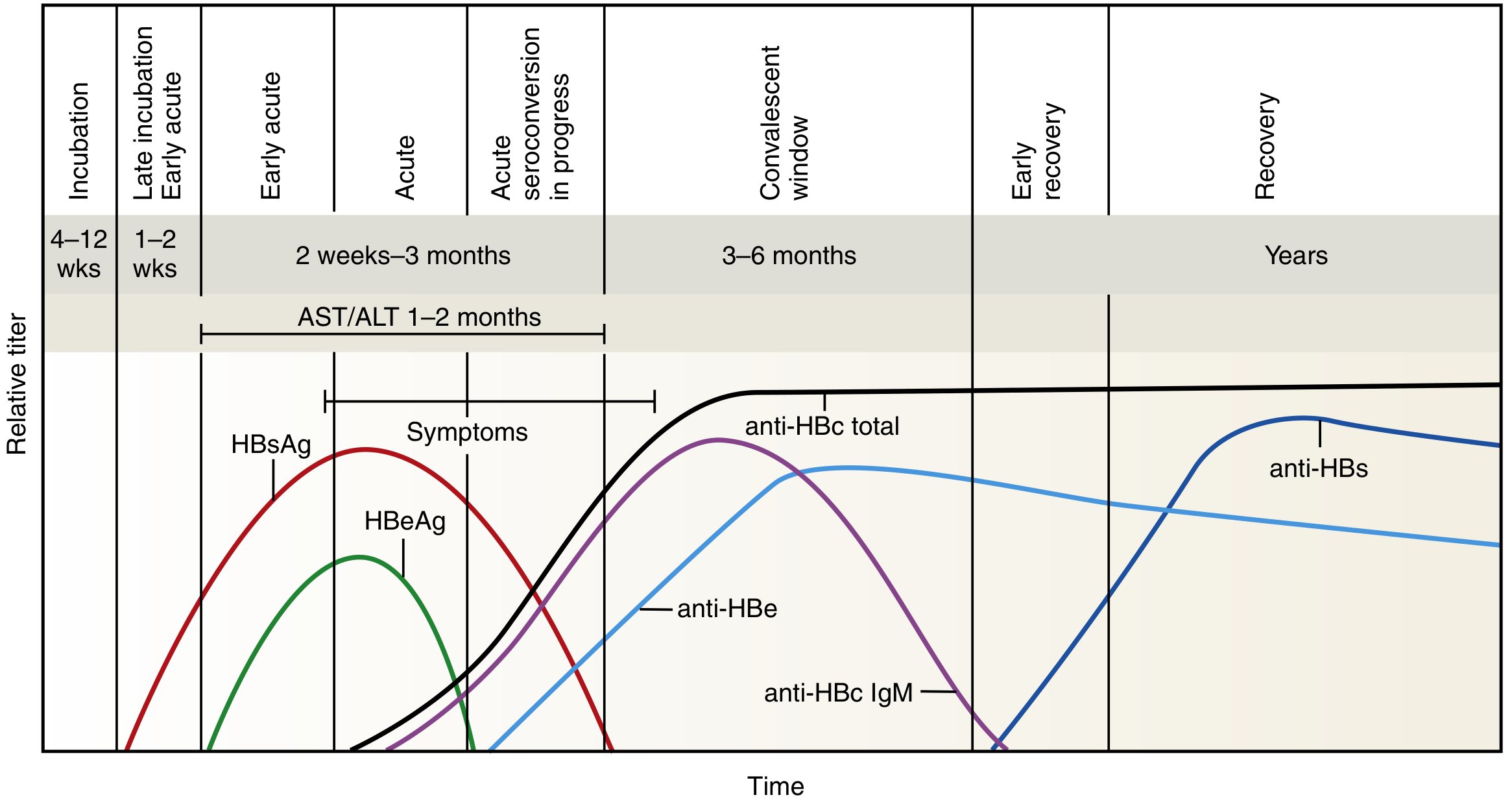
Key Markers:
| Marker | Significance |
|---|---|
| HBsAg | First detectable marker (2–6 weeks before symptoms); persists throughout active disease; >6 months = chronic |
| HBeAg | Marker of active viral replication and high infectivity |
| HBV DNA / DNA polymerase | Earliest markers of viremia; appear during incubation |
| IgM anti-HBc | Indicates acute infection and active viral replication; high titer = current/recent infection |
| IgG anti-HBc | Persists for life; present in both past infection and chronic infection |
| Anti-HBe | Replaces HBeAg; signals start of resolution |
| Anti-HBs | Last marker to appear; indicates recovery and immunity; also conferred by vaccination |
Core Window Period:
A gap (~3–6 months) exists where HBsAg has disappeared but anti-HBs has not yet risen. During this period, IgM anti-HBc and anti-HBe are the only markers of infection.
Serologic Interpretation:
| Pattern | Interpretation |
|---|---|
| HBsAg+ , IgM anti-HBc+ | Acute HBV infection |
| HBsAg+ >6 months, IgG anti-HBc+ | Chronic HBV infection |
| Anti-HBs+ , anti-HBc+ | Past infection with recovery |
| Anti-HBs+ only (no anti-HBc) | Vaccination |
| IgG anti-HBc+ only (isolated) | Core window, or past infection with waned anti-HBs |
5. Incubation & Clinical Features
- Incubation period: 50–180 days (mean 60–90 days)
- Prolonged with low-dose or non-percutaneous exposure
- Clinical spectrum:
- Subclinical/asymptomatic (most common — major hazard to healthcare workers)
- Acute symptomatic hepatitis: jaundice, fatigue, nausea, RUQ discomfort, elevated AST/ALT
- Fulminant hepatic failure (rare but life-threatening; worsened by HDV superinfection)
- Chronic hepatitis → cirrhosis → HCC
6. Chronic HBV Infection
- Defined as HBsAg persisting >6 months with HBeAg or anti-HBe
- Risk of chronicity varies by age at infection:
- Neonates: ~80%
- Immunocompromised patients: 5–10%
- Normal adults: 1–2%
- Pre-core mutants: Stop codon mutation at nucleotide 1896 → absent HBeAg production despite ongoing viral replication → HBeAg-negative chronic hepatitis (harder to monitor)
- Hemodialysis patients have a 50% chronic carrier rate vs. 2% in staff, illustrating immune response differences
- Occult HBV infection can occur: HBsAg negative but HBV DNA detectable; can still transmit virus
7. Complications
- Chronic active hepatitis (25% of carriers)
- Cirrhosis
- Hepatocellular carcinoma (HCC): HBV is a major risk factor, particularly in endemic regions; linked via HBx protein and chromosomal integration
- Extrahepatic manifestations: polyarteritis nodosa, membranous glomerulonephritis, arthritis, urticarial rash (due to immune complex deposition)
8. Diagnosis & Monitoring
For acute HBV: HBsAg + IgM anti-HBc
For chronic HBV: HBsAg + IgG anti-HBc + IgG anti-HBs + HBeAg + anti-HBe status
Monitoring chronic infection: HBsAg, HBeAg, quantitative HBV DNA PCR (detects as low as 200 copies/mL), ALT/AST
- Quantitative real-time PCR is the gold standard for monitoring treatment response
- HBV DNA >2,000 IU/mL (HBeAg-negative) or >20,000 IU/mL (HBeAg-positive) are thresholds for treatment consideration
9. Treatment
Acute HBV:
Generally does not require antiviral therapy — supportive management.
Chronic HBV — Indications for Treatment:
- HBsAg+ >6 months
- HBV DNA: >2,000 IU/mL (HBeAg-negative), >20,000 IU/mL (HBeAg-positive), or >200,000 IU/mL (pregnant)
- ALT >2× upper limit of normal
- Clinically decompensated disease (always treat)
Approved Agents:
Nucleic acid analogues (oral — preferred for most):
| Drug | Dose | Notes |
|---|---|---|
| Tenofovir disoproxil fumarate (TDF) | 300 mg/day PO | First-line; also active against lamivudine-resistant HBV |
| Tenofovir alafenamide (TAF) | 25 mg/day PO | Lower renal/bone toxicity than TDF |
| Entecavir | 0.5 mg/day (naïve); 1 mg/day (lamivudine-resistant) | First-line |
| Adefovir | Older; now rarely used | |
| Lamivudine | High resistance rates; rarely used now | |
| Telbivudine | Rarely used |
Immune modulators:
- Interferon-alfa-2b (standard IFN)
- Pegylated interferon-alfa-2a (PEG-IFN) — preferred immune modulator; finite duration treatment
Goals of treatment: Suppress HBV replication → prevent progression to cirrhosis and HCC
Duration:
- HBeAg-positive: until HBeAg seroconversion + undetectable HBV DNA, then ≥6 additional months
- HBeAg-negative: at least 1 year
- Decompensated cirrhosis / post-liver transplant: lifelong treatment
10. Prevention
Passive immunization:
- Hepatitis B immune globulin (HBIG): used post-exposure (needlestick, sexual assault, neonates of HBsAg+ mothers)
Active immunization (vaccine):
- Highly effective recombinant HBsAg vaccine
- Standard schedule: 3-dose series (0, 1, 6 months)
- Neonates born to HBsAg+ mothers receive HBIG + vaccine within 12 hours of birth
- Poor vaccine response in hemodialysis patients → vaccinate as early as possible in CKD progression
- Mandatory universal infant vaccination has dramatically reduced HBV incidence globally
- Vaccination also reduces HCC risk (effectively an anti-cancer vaccine)
Sources: Jawetz Melnick & Adelberg's Medical Microbiology 28e | Henry's Clinical Diagnosis and Management by Laboratory Methods | Goldman-Cecil Medicine | Brenner and Rector's The Kidney
This is a shared conversation. Sign in to Orris to start your own chat.