Elaborate essay under 1000 words on Conservative Laryngeal Surgeries
Conservative Laryngeal Surgeries
Introduction
Historical Background
Classification of Procedures
1. Vertical Partial Laryngectomy (VPL)
- Cordectomy (via laryngofissure): Removal of a single true vocal cord through a midline thyrotomy. Classically indicated for T1a lesions confined to the midcord, it offers local control exceeding 90% in appropriately selected patients.
- Frontolateral partial laryngectomy: Extends the resection to include the anterior commissure and contralateral vocal fold margin, addressing T1b lesions with anterior commissure involvement.
- Vertical hemilaryngectomy and extended hemilaryngectomy: Resects the ipsilateral true and false vocal cords and the adjacent thyroid cartilage perichondrium. Suitable for T1 and selected T2 glottic cancers. Local recurrence rates for T1 lesions range from 4% to 11% across major series, with most achieving local control above 90%. Extended forms address subglottic extension, though anterior commissure involvement carries a higher local failure risk — one series documenting a 25% failure rate in this subgroup. T2 lesions present a greater challenge, with multiple series reporting local failure rates exceeding 20%, underscoring the need for careful patient selection. — Cummings Otolaryngology Head and Neck Surgery
2. Horizontal Partial Laryngectomy
- Supraglottic partial laryngectomy (SGL): Removes the epiglottis, pre-epiglottic space, both false vocal cords, and the upper portion of the thyroid cartilage — preserving the true vocal cords and thus voice quality. It is indicated for T1 and T2 supraglottic cancers without true cord involvement. The procedure was historically performed as an open operation, but has been largely supplanted by endoscopic (transoral laser) approaches offering equivalent oncologic results with reduced morbidity.
- Supracricoid partial laryngectomy (SCPL): The most significant advance in conservation laryngeal surgery in the modern era. SCPL removes the thyroid cartilage, both true and false vocal cords, the paraglottic spaces, and — depending on the variant — the epiglottis. The cricoid cartilage, at least one arytenoid, and the hyoid bone are preserved. Two reconstruction variants exist: cricohyoidopexy (CHP), which approximates the cricoid directly to the hyoid (used after supraglottic extension), and cricohyoidoepiglottopexy (CHEP), which preserves the epiglottis and is used for pure glottic lesions. SCPL provides consistent oncologic and functional outcomes for selected T2 and T3 glottic and supraglottic carcinomas, and is particularly valuable for patients with small recurrences following prior radiation. The procedure avoids permanent tracheostomy; with rehabilitation, patients recover swallowing through a neoglottis formed by arytenoid-to-epiglottis or arytenoid-to-hyoid contact. The main long-term functional sequela is dysphonia, which is predictable and generally acceptable to patients. — Cummings Otolaryngology Head and Neck Surgery
3. Endoscopic (Transoral) Approaches
Patient Selection and Preoperative Assessment
Oncologic and Functional Outcomes
Conclusion
In Headings of
- "In headings of [book name]..."
- "In headings of conservative laryngeal surgeries..."
1. TLM/TELR
From Block 24 (Conservation Laryngeal Surgery chapter):
- Complications of Transoral Laser Microresection
- Contraindications to Transoral Laser Microresection
- Results of Transoral Laser Microresection
From Block 23 (Early/Advanced Glottic Cancer chapter):
- Laser Surgery and Transoral Laser Microsurgery in the Treatment of Locally Early, Intermediate, and Advanced Cancers
- Transoral Laser Microsurgery Compared with Open Conservation Surgery
- Instrumentation and Techniques of Transoral Laser Microsurgery
- Transoral Laser Microsurgery in Relation to Each of the Five Clinical Categories of Laryngeal Cancer Stratified by Local Extent
From Block 20 (general ENT procedures):
- Transoral Laser Microsurgery (appears twice — likely main heading and a sub-reference)
1. TELR/TLM. 2. SCPL + CHEP. 3. VPL. 4.Supraglottic Laryngectomy. All 4 types with diagrams, indications, contraindications, Surgical Steps & Complications
transoral laser microsurgery larynx TLM cordectomy endoscopic
supracricoid partial laryngectomy SCPL CHEP cricohyoidoepiglottopexy surgical diagram
supraglottic laryngectomy surgical steps epiglottis hyoid resection vertical hemilaryngectomy VPL diagram
Conservative Laryngeal Surgeries — Comprehensive Guide
Overview of the Four Procedures
| Procedure | Approach | Structures Resected | Permanent Tracheostomy |
|---|---|---|---|
| TLM/TELR | Endoscopic (transoral) | Variable (mucosa → cord → paraglottic) | No |
| VPL | Open, vertical thyrotomy | Ipsilateral cord(s) ± cartilage | No |
| SCPL + CHEP | Open, horizontal | Thyroid cartilage, both cords, epiglottis preserved | No |
| Supraglottic Laryngectomy | Open, horizontal thyrotomy | Epiglottis, false cords, pre-epiglottic space | No |
1. Transoral Endoscopic Laser Resection (TELR) / Transoral Laser Microsurgery (TLM)
Concept
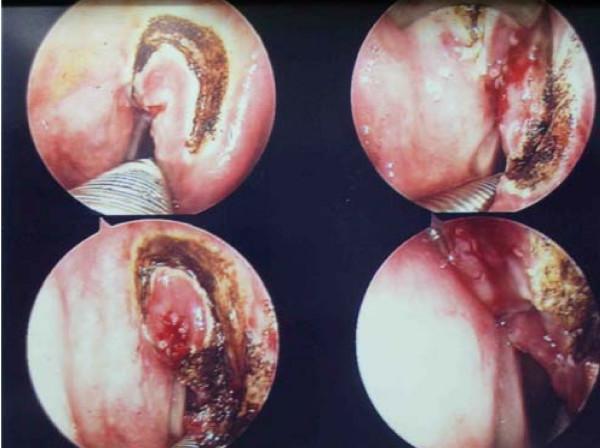
Indications
- T1a, T1b glottic carcinoma (primary treatment of choice in many centres)
- T2 glottic and supraglottic carcinoma (selected cases)
- T3 lesions in selected patients (locally intermediate/advanced)
- Early supraglottic carcinomas (replacing open supraglottic laryngectomy)
- Recurrent cancers after prior radiotherapy (salvage)
- Benign laryngeal lesions (papillomatosis, subglottic stenosis, Reinke's edema)
Contraindications
- Poor endoscopic exposure — macroglossia, retrognathia, trismus, capped/fragile teeth
- Uncontrolled coagulopathy or recent antiplatelet therapy (risk of catastrophic hemorrhage)
- Ossified thyroid cartilage (limits margin assessment)
- Tumors too bulky to extract transorally
- Named artery involvement without ability to safely clip/ligate
- Patients unable to tolerate general anesthesia — Cummings Otolaryngology Head and Neck Surgery
Surgical Steps
- Setup & Positioning: Patient supine with neck extended. Suspension laryngoscopy via Kleinsasser or similar laryngoscope is paramount — this is the crux of exposure, not the laser.
- Airway: Small double-cuffed laser endotracheal tube; FiO₂ kept below 30% with saline in each cuff to prevent fire risk.
- Protection: Face and eyes covered with wet toweling; OR personnel wear protective eyewear; angled suction plume protectors placed over cords.
- Laser settings:
- Pulsed mode, 1–3 W → delicate glottic mucosa (minimum thermal injury, best texture definition at cut surface)
- Continuous mode, ~6 W → standard laryngeal incisions with adequate hemostasis
- Defocused, 15–20 W → vaporization of friable, bulky necrotic tumour centres
- Resection principle (Steiner): Divide tumor in situ into manageable subunits. Begin at tumor periphery, confirm depth at each step — "follow the tumor." Avoid vaporization of specimen margins — excise cleanly so the pathologist can read margins.
- Hemostasis: Arterioles → electrocautery. Named vessels (e.g., superior laryngeal artery) → clip, never trust cautery alone.
- Reconstruction: Generally not required. Healing is by secondary intention.
- Tracheotomy decision: Most TLM cases require no tracheotomy. Consider it for: prolonged tongue base edema (lengthy case), large supraglottic resections with aspiration risk, significant hemorrhage risk, or when the endotracheal tube impedes subglottic access. — Cummings Otolaryngology Head and Neck Surgery
Complications
| Complication | Notes |
|---|---|
| Stray laser burns — mucosa, skin, eyes | Use wet toweling, protective eyewear |
| Airway fire | Keep FiO₂ < 30%, saline-filled laser tube cuffs |
| Delayed hemorrhage | Most dangerous without a tracheotomy; clip named arteries |
| Lingual contusion/swelling | Prolonged suspension causes tongue pressure |
| Dysphagia/dysesthesia | Post-suspension tongue injury |
| Dental injury | Use thermosetting Aquaplast over upper incisors |
| Retained sponge/foreign body | Vigilance with every laryngoscope repositioning |
| Airway edema | Managed expectantly or with repeat laser laryngoscopy |
| Tracheotomy wound issues | When indicated |
2. Supracricoid Partial Laryngectomy (SCPL) + CHEP
Concept
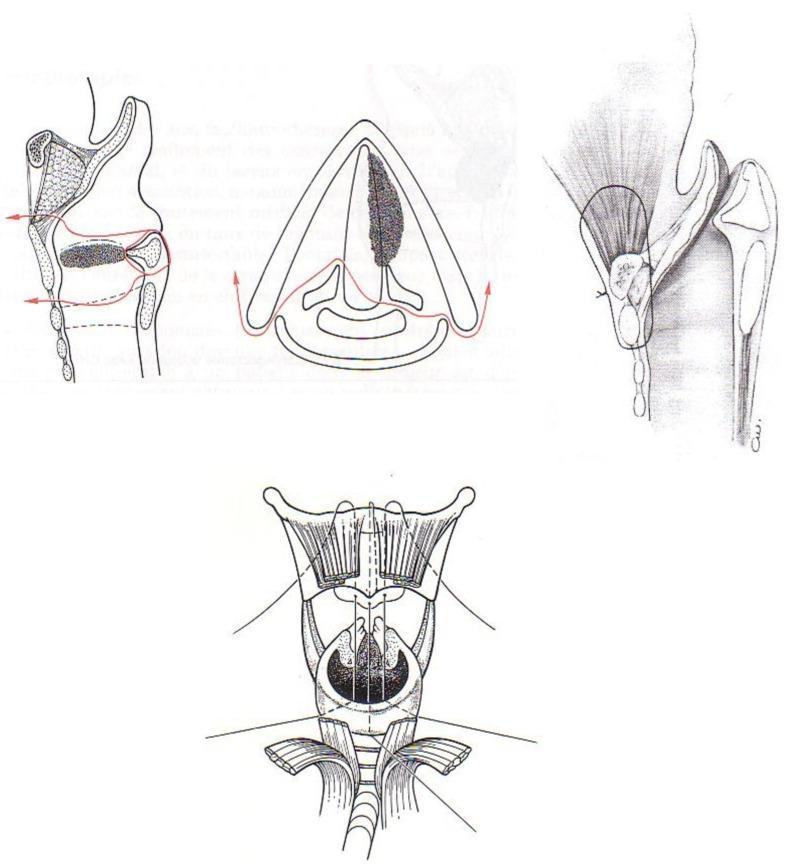
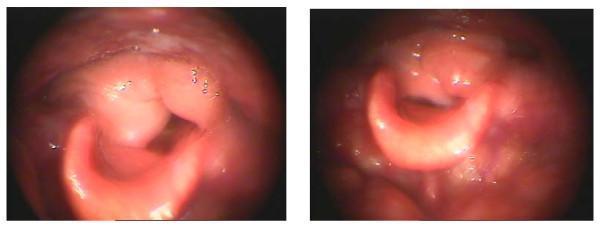
Indications
- T2 glottic carcinoma with impaired cord mobility (not amenable to VPL)
- T3 glottic carcinoma with limited pre-epiglottic and subglottic invasion
- T2 supraglottic carcinoma extending to the glottis (CHEP variant — epiglottis retained)
- Small recurrent glottic/supraglottic cancers post-radiotherapy
- Patients requiring organ preservation where endoscopic resection is inadequate
- Transglottic cancers (T3) not invading the cricoid or requiring total laryngectomy
Contraindications
- Cricoid cartilage invasion (absolute — cricoid is the keystone of reconstruction)
- Bilateral arytenoid fixation (need at least one mobile arytenoid for neoglottic competence)
- Massive pre-epiglottic space involvement (for CHEP; requires switching to CHP)
- Subglottic extension >10 mm anteriorly or >5 mm posteriorly
- Poor pulmonary reserve — mandatory to tolerate aspiration during rehabilitation
- Significant swallowing dysfunction pre-operatively
Surgical Steps (CHEP)
- Tracheotomy is performed first (temporary — can be decannulated post-rehabilitation).
- Apron (collar) incision; bilateral modified neck dissection if N+ disease.
- Strap muscle division at level of superior thyroid cartilage; sternohyoid and thyrohyoid muscles separated.
- Superior laryngeal nerve identified and preserved bilaterally.
- Thyroid cartilage skeletonization: External perichondrium elevated; thyroid cartilage exposed entirely.
- Horizontal cuts: Inferior cut above the cricoid arch (preserving the cricoid); superior cuts above the false cords (preserving the epiglottis in CHEP).
- Paraglottic space resection: Both paraglottic spaces, thyroid cartilage, both true and false cords removed en bloc.
- Arytenoid assessment: At least one fully mobile arytenoid preserved; the contralateral may be resected if required for oncologic clearance.
- CHEP reconstruction: The cricoid is pulled superiorly to meet the hyoid + base of epiglottis. Three heavy non-absorbable sutures (cricoid-to-hyoid) provide the pexy, creating a neoglottis formed by arytenoid contact against the retained epiglottis.
- Wound closure in layers; nasogastric tube placed for early postoperative feeding. — Cummings Otolaryngology Head and Neck Surgery
Complications
| Complication | Notes |
|---|---|
| Aspiration (universal early) | Expected; resolves with rehabilitation in weeks–months |
| Aspiration pneumonia | Most significant early morbidity |
| Deglutition failure | Rare but may require conversion to total laryngectomy |
| Wound hematoma/seroma | Standard post-neck dissection risk |
| Fistula | Uncommon |
| Delayed decannulation | Especially in elderly or poor lung reserve |
| Dysphonia | Predictable; main long-term sequela — voice is hoarse but functional |
| Pexy dehiscence | Disruption of cricohyoid suture line — rare |
| Local recurrence | ~10–15% for T2–T3; managed by salvage total laryngectomy |
3. Vertical Partial Laryngectomy (VPL)
Concept
Variants
| Procedure | Resection | Indication |
|---|---|---|
| Cordectomy (via laryngofissure) | Single true cord | T1a mid-cord |
| Frontolateral laryngectomy | Cord + anterior commissure + contralateral margin | T1b |
| Vertical hemilaryngectomy | Ipsilateral true + false cord + cartilage | T1, selected T2 |
| Extended hemilaryngectomy | + subglottis / arytenoid | T2 with subglottic extension |
| Epiglottic laryngoplasty | Bilateral cords ± arytenoid + epiglottic advancement | T1b, T2, selected T3 bilateral lesions |
Indications
- T1a glottic carcinoma — cordectomy / hemilaryngectomy
- T1b with anterior commissure — frontolateral partial laryngectomy
- T2 glottic with limited subglottic extension (selected)
- Lesions confined to the glottis without thyroid cartilage invasion
- Recurrent T1 after radiotherapy in selected patients
Contraindications
- Thyroid cartilage invasion (precludes adequate margin without destroying framework)
- Anterior commissure involvement with VHL — high subglottic recurrence risk (14–25%)
- Subglottic extension >5 mm posteriorly (cricoid not resected in standard VPL)
- Supraglottic extension through the ventricle (risk of cartilage invasion)
- Bilateral arytenoid involvement
- T3–T4 disease with paraglottic/extralaryngeal spread
Surgical Steps (Vertical Hemilaryngectomy)
- Tracheotomy performed first.
- Collar incision; strap muscles separated in midline.
- Thyroid perichondrium incised in midline; elevated with overlying strap musculature.
- Vertical thyrotomy #1: 3–4 mm anterior to the posterior border of the thyroid cartilage on the involved side.
- Vertical thyrotomy #2: Midline (or more anteriorly on the less-involved side for bilateral lesions).
- Transverse cricothyrotomy performed.
- Paraglottic space transected on the less-involved side with right-angled scissors.
- Involved side resection: Ipsilateral true + false cord, with or without arytenoid.
- Key surgical points:
- Tack the epiglottis petiole back with 3-0 Vicryl to prevent posterior prolapse blocking glottic view post-op.
- Suture the anterior commissure of the non-involved side to the external perichondrium to maintain cord position and vocal tendon tension (4-0 Vicryl).
- External perichondrial flap + strap muscles closed across midline. — Cummings Otolaryngology Head and Neck Surgery
Complications
| Complication | Notes |
|---|---|
| Local recurrence | T1: 4–11%; T2: >20% in multiple series |
| Subglottic recurrence | Especially with anterior commissure involvement (14–25%) |
| Wound seroma/hematoma | Uncommon |
| Fistula | Uncommon in standard procedures |
| Delayed decannulation | Extended VPL → more edema |
| Stenosis | Extended procedures |
| Long-term dysphagia | Extended hemilaryngectomy |
| Persistent airway edema | Managed with laser laryngoscopy |
| Dysphonia | Expected; varies with extent of resection |
4. Supraglottic Laryngectomy (SGL)
Concept
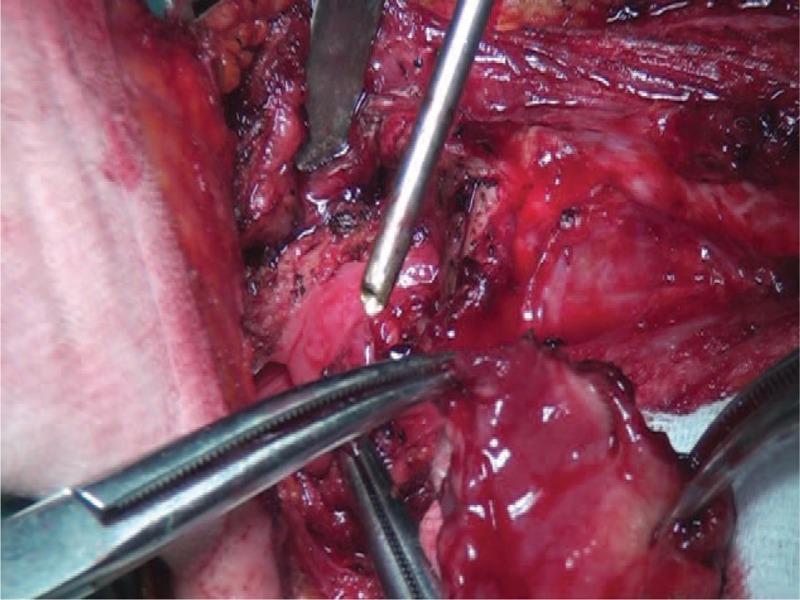
Indications
- T1, T2 supraglottic carcinoma confined to the supraglottis (epiglottis, aryepiglottic folds, false cords)
- Adequate pulmonary reserve (essential — transient aspiration is universal post-operatively)
- No true cord involvement, no impaired cord mobility
- Hyoid bone can be spared if pre-epiglottic space is free (confirmed on CT/MRI)
Contraindications
- True vocal cord involvement or impaired mobility (extension to glottis — statistically significant predictor of glottic invasion)
- Interarytenoid mucosa involvement
- Massive pre-epiglottic space invasion (may require total laryngectomy)
- Bilateral base-of-tongue involvement
- Poor pulmonary function (forced vital capacity <50% predicted, significant COPD)
- Circumferential piriform sinus involvement
- T4 disease with cartilage invasion
Surgical Steps
- Apron incision in line with tracheotomy; routine bilateral modified neck dissection.
- Superior laryngeal nerve main trunk spared bilaterally (essential for sensory rehabilitation of swallowing).
- Strap muscle fascia divided at the superior thyroid cartilage level; thyroid isthmus transected and ligated.
- Sternohyoid and thyrohyoid muscles divided at the superior border of thyroid cartilage — care taken laterally to avoid superior laryngeal nerves.
- Pharyngeal constrictor muscles sharply cut at the posterior/superior thyroid cartilage edge up to the superior cornuа.
- External thyroid cartilage perichondrium elevated halfway down the cartilage from its superior edge.
- Horizontal thyrotomy (sagittal saw) at the level just above the anterior commissure.
- Hyoid bone management:
- If pre-epiglottic space is free (T1–T2 endolaryngeal): hyoid is spared and skeletonized from below with electrocautery.
- If vallecula is involved: hyoid is resected en bloc.
- Vallecula entered, epiglottis pulled externally with Allis clamp; resection proceeds close to the epiglottis, sparing both arytenoid cartilages and the mucosa over the arytenoids bilaterally.
- Piriform sinuses freed bilaterally (endolaryngeal tumors) or unilaterally (piriform sinus involvement).
- Tracheotomy performed; nasogastric tube placed.
- Closure: Residual pharyngeal/laryngeal mucosa approximated; strap muscles closed. — Cummings Otolaryngology Head and Neck Surgery
Complications
| Complication | Notes |
|---|---|
| Aspiration | Universal early; rehabilitation critical |
| Aspiration pneumonia | Most clinically significant early morbidity |
| Failure to swallow | Rare; may require total laryngectomy |
| Delayed decannulation | Especially in older patients |
| Local recurrence | T1–T2: 0–12.8%; T3–T4: up to 75% — extreme caution with T3/T4 |
| Wound fistula | Uncommon |
| Hematoma/seroma | Post-neck dissection |
| Pharyngocutaneous fistula | Uncommon with standard technique |
| Worsened outcomes post-RT | Extended SGL + radiotherapy → decreased function |
Summary Comparison
| Feature | TLM/TELR | VPL | SCPL + CHEP | Supraglottic LX |
|---|---|---|---|---|
| Approach | Transoral endoscopic | Open vertical | Open horizontal | Open horizontal |
| Tumour Stage | T1–T3 (selected) | T1–T2 glottic | T2–T3 glottic/transglottic | T1–T2 supraglottic |
| Structures removed | Variable | Ipsilateral cord(s) | Thyroid cartilage + both cords | Epiglottis + false cords |
| Voice outcome | Good to excellent | Reduced | Hoarse but functional | Excellent (true cords preserved) |
| Swallowing | Near normal | Near normal | Rehabilitation ~weeks–months | Rehabilitation needed |
| Permanent tracheostomy | No | No | No | No |
| Key concern | Access/exposure | Anterior commissure/subglottis | Aspiration, pulmonary reserve | Aspiration, pulmonary reserve |
| Main long-term sequela | Dysphonia (if extensive) | Dysphonia | Dysphonia | Aspiration resolution |
Papillary Carcinoma of Thyroid. Clinical features and Management
papillary thyroid carcinoma histology Orphan Annie nuclei psammoma bodies
Papillary Carcinoma of the Thyroid (PTC)
Overview
Aetiology & Risk Factors
- Female predominance: Occurs 3× more frequently in women; peak incidence in women at 40–50 years, 10–20 years later in men
- Ionizing radiation exposure: The most well-established external causative factor; incidence markedly rose after the Chernobyl nuclear accident
- Familial syndromes: Familial adenomatous polyposis (FAP), Gardner syndrome, and Cowden syndrome are associated with a small subset of PTC
- Molecular alterations: The MAPK (mitogen-activated protein kinase) pathway is altered in ~70% of cases
- BRAF V600E mutation: Most common genetic event, present in ~50% of PTCs; associated with extrathyroidal invasion, higher nodal and distant metastasis rates, and increased recurrence
- Other mutations: RET/PTC rearrangements, NTRK1, RAS
- The majority of PTCs arise spontaneously — K.J. Lee's Essential Otolaryngology, Robbins Pathology
Pathology & Histology
Gross Morphology
- Solitary or multifocal lesions (PTC has a distinct tendency for multifocality within the gland)
- May be well-circumscribed and encapsulated or ill-defined and infiltrative
- Papillary foci may be visible on cut surface
Microscopic Hallmarks (Robbins Pathology)
- Branching papillae with fibrovascular stalks covered by cuboidal epithelial cells
- "Orphan Annie eye" nuclei (ground-glass nuclei): Large nuclei with finely dispersed chromatin giving an optically clear/empty appearance — the single most diagnostic nuclear feature
- Intranuclear inclusions (pseudoinclusions): Cytoplasmic invaginations giving appearance of inclusions
- Intranuclear grooves
- Psammoma bodies: Concentrically calcified structures within papillary cores — virtually diagnostic when seen in the neck; almost never found in follicular or medullary carcinoma
- Lymphatic invasion frequently present; blood vessel invasion uncommon in smaller lesions
These nuclear features (Orphan Annie eye nuclei, inclusions, grooves) are sufficient for diagnosis of PTC even in the absence of papillary architecture — Robbins & Kumar Basic Pathology
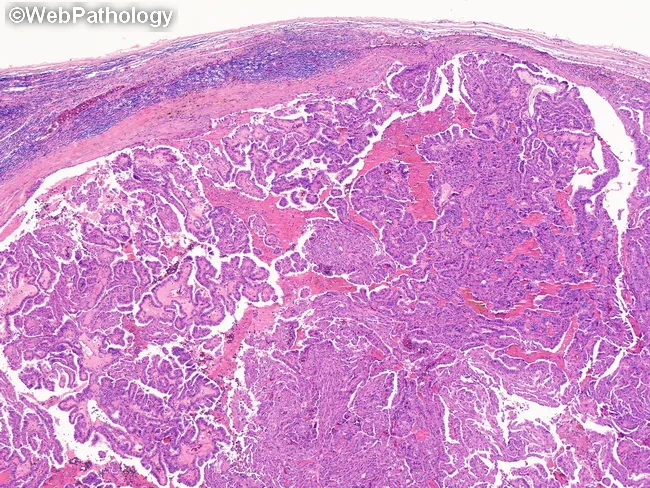
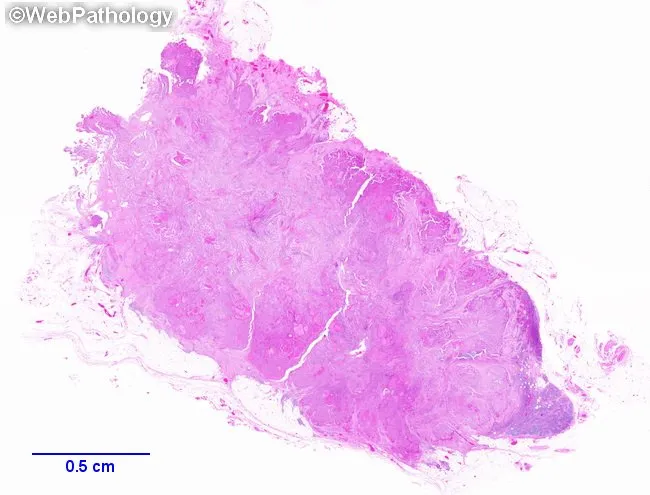
Variants
| Variant | Notes |
|---|---|
| Encapsulated follicular variant | Most common variant; PTC nuclear features + follicular architecture; may carry PAX8-PPARG fusion |
| Diffuse sclerosing | Unfavourable; extensive lymphatic invasion |
| Tall cell variant | Unfavourable; extrathyroidal extension common |
| Columnar cell variant | Unfavourable; aggressive behaviour |
| Microcarcinoma | ≤1 cm; incidental finding; excellent prognosis |
Clinical Features
Presentation
- Painless thyroid nodule or neck mass — the most common presentation
- PTC is non-functional — thyroid function tests are typically normal
- Cervical lymphadenopathy — may be the first presenting sign; nodal metastases can undergo cystic degeneration or appear black in colour
- Rarely: hoarseness (recurrent laryngeal nerve invasion), dysphagia, or dyspnoea in advanced disease
Spread & Metastasis
- Strongly lymphotropic — spreads via intrathyroidal lymphatics then to regional cervical nodes
- At presentation:
- ~30% of adults have clinically evident cervical nodal disease
- ~3% have distant metastases (most commonly lung, followed by bone)
- Paediatric population: Nodal metastatic rate as high as 60% at presentation
- Distant metastases (haematogenous): lung most common; bone also seen
- Microscopic nodal disease has no significant prognostic impact; macroscopic nodal disease increases subsequent recurrence risk — K.J. Lee's Essential Otolaryngology
Staging (AJCC 8th Edition — Age-Stratified)
| Age | T | N | M | Stage |
|---|---|---|---|---|
| <55 years | Any T | Any N | M0 | I |
| <55 years | Any T | Any N | M1 | II |
| ≥55 years | T1–T2 | N0/NX | M0 | I |
| ≥55 years | T1–T2 | N1 | M0 | II |
| ≥55 years | T3a/T3b | Any N | M0 | II |
| ≥55 years | T4a | Any N | M0 | III |
| ≥55 years | T4b | Any N | M0 | IVA |
| ≥55 years | Any T | Any N | M1 | IVB |
Prognosis
- 10-year survival >90–95%; among the most indolent of all malignancies
- Age >40 years → increased recurrence and mortality; risk increases further after age 60
- BRAF mutation, extrathyroidal extension, and distant metastasis are negative prognosticators
- Children tend to present at a more advanced stage but still have excellent long-term outcomes
Investigations
1. Ultrasound (USG Neck)
- Most sensitive and specific imaging modality for evaluating thyroid nodules
- Features suspicious for malignancy: hypoechoic solid nodule, irregular margins, microcalcifications (psammoma bodies), taller-than-wide shape, increased vascularity
- Detects cervical lymph node metastases, altering surgical planning
- Essential for post-treatment surveillance — thyroid bed and neck monitoring
2. Fine-Needle Aspiration Cytology (FNAC)
- Standard diagnostic investigation for thyroid nodules
- Characteristic nuclear features allow preoperative diagnosis (Bethesda classification)
- FNA showing intranuclear inclusions and grooves is highly specific for PTC
3. CT / MRI
- For assessment of extrathyroidal extension, substernal goitre, tracheal/oesophageal involvement, or extensive nodal disease
- CT with contrast is avoided pre-RAI therapy (iodine load)
4. Serum Thyroglobulin (Tg)
- Useful as a tumour marker post-thyroidectomy for surveillance
- Undetectable Tg (<0.2 ng/mL on levothyroxine or <1 ng/mL after TSH stimulation) with negative Tg antibodies + normal neck USG = very low recurrence risk
5. Molecular Testing
- BRAF V600E mutation testing guides prognosis and eligibility for targeted therapy
Management
Step 1 — Surgery
Total Thyroidectomy
- Standard surgical treatment for most PTC
- Rationale: PTC is multifocal in up to 30–85% of cases (contralateral lobe involvement)
- Total thyroidectomy facilitates postoperative RAI therapy and Tg surveillance
- Thyroid lobectomy may be adequate for low-risk, unifocal tumours ≤1–4 cm without extrathyroidal extension, no nodal disease, and no high-risk histologic features
- A paradigm shift toward total or near-total thyroidectomy has occurred with reduced use of subtotal thyroidectomy — Schwartz's Principles of Surgery, Sabiston Textbook of Surgery
Neck Dissection
- Central compartment (Level VI) neck dissection:
- Should be considered in high-risk PTC (T3–T4 tumours) and suspected Hürthle cell carcinoma
- Bilateral central compartment dissection for high-risk cases
- Role of prophylactic (elective) central dissection for clinically N0 disease remains controversial; some series show reduction in locoregional recurrence
- Lateral neck dissection (Levels II–V):
- Performed only when radiographically detectable lateral nodal disease is confirmed
- Elective lateral neck dissection is not recommended in the absence of confirmed lateral disease
- When lateral dissection is performed, a systematic compartment dissection is mandatory — "berry-picking" is not acceptable
- N1a: Metastasis to Level VI (central compartment)
- N1b: Metastasis to lateral neck, retropharyngeal, or superior mediastinal nodes — Cummings Otolaryngology, Schwartz's Principles of Surgery
Step 2 — Radioactive Iodine (RAI / ¹³¹I)
Indications
- Low-risk DTC: RAI is NOT routinely recommended — multiple large studies (including a 2022 RCT) show no benefit in disease-free survival for low-risk PTC treated with total thyroidectomy + RAI vs. surgery alone
- Intermediate-risk DTC: Selective use — a 21,870-patient NCDB study showed a 29% reduction in mortality risk
- High-risk DTC: RAI is routinely recommended to reduce recurrence and mortality
Two Broad Indications for RAI
- Remnant ablation: Destroys residual normal thyroid tissue post-thyroidectomy, increasing specificity of Tg surveillance and ¹³¹I scanning; prevents de novo cancer in remnant
- Treatment of persistent/recurrent disease: For disease not amenable to surgical resection
Administration
- Must be given when patient is in low-iodine state and with elevated TSH to maximise uptake
- TSH stimulation: thyroid hormone withdrawal OR recombinant human TSH (rhTSH/Thyrogen)
- Doses:
- Remnant ablation: 30–50 mCi
- Treatment-level: 100–150 mCi
- Maximum cumulative lifetime dose: ~600 mCi
Adverse Effects of RAI
| Side Effect | Notes |
|---|---|
| Sialadenitis | Common; dose-dependent |
| Nasolacrimal duct obstruction | |
| Transient thyroid/tumour swelling | |
| Infertility | Dose-dependent |
| Secondary malignancy | Particularly leukaemia; dose-dependent |
| Absolute contraindications | Pregnancy and breastfeeding |
Step 3 — TSH Suppression Therapy
- Levothyroxine (T4) administered post-thyroidectomy to suppress TSH, removing growth stimulus for residual or recurrent thyroid cancer cells
- Degree of suppression tailored to risk:
- High-risk: TSH kept <0.1 mU/L
- Low-risk after ablation with undetectable Tg: TSH maintained at low-normal (0.5–2.0 mU/L)
- Long-term risks of TSH over-suppression: atrial fibrillation, osteoporosis — must be balanced against oncologic benefit
Step 4 — Surveillance
- Serum Tg (± anti-Tg antibodies) + neck ultrasound at regular intervals
- Diagnostic ¹³¹I whole-body scan in intermediate/high-risk patients
- Follow-up stratified by ATA risk classification (low / intermediate / high)
Step 5 — Targeted Therapy (Refractory/Metastatic Disease)
- For RAI-refractory metastatic PTC:
- Sorafenib (multikinase inhibitor) — first-line; approved for RAI-refractory DTC
- Lenvatinib — superior progression-free survival; now preferred first-line agent
- BRAF/MEK inhibitors (e.g., dabrafenib + trametinib): for BRAF V600E-mutant PTC
- Selective RET inhibitors (selpercatinib, pralsetinib): for RET-rearranged PTC
- External beam radiotherapy (EBRT): Limited role; used palliatively for unresectable local disease, painful bone metastases, or CNS involvement
Summary Algorithm
Thyroid nodule suspicious on USG
↓
FNAC → Bethesda Category V/VI (suspicious / malignant)
↓
Staging CT / Pre-op neck USG
↓
SURGERY
├── Low-risk (unifocal <4 cm, N0): Thyroid lobectomy OR total thyroidectomy
├── All others: Total thyroidectomy + Level VI neck dissection
└── Lateral N+: + Therapeutic lateral neck dissection (Levels II–V)
↓
Post-op Risk Stratification (ATA: Low / Intermediate / High)
↓
RAI Therapy
├── Low risk → NOT routinely indicated
├── Intermediate risk → Selective
└── High risk → Routinely recommended (100–150 mCi)
↓
TSH Suppression (Levothyroxine)
↓
Long-term Surveillance (Tg + USG ± whole-body scan)
↓
RAI-refractory/distant metastases → Targeted therapy (Lenvatinib / Sorafenib / BRAF inhibitors)
"Image guided surgery in ENT " answer for PG exam under 1000 words with relevant images, diagrams
image guided surgery ENT navigation system intraoperative CT sinus skull base
image guided surgery electromagnetic optical tracking registration fiducial marker ENT
Image-Guided Surgery (IGS) in ENT
Introduction
Principle of Image-Guided Surgery
- A preoperative imaging dataset (thin-cut CT scan, ≤1 mm slices, reformatted in axial, coronal, and sagittal planes)
- The patient's actual anatomy in the operating theatre
- The position of the surgical instrument within that anatomy

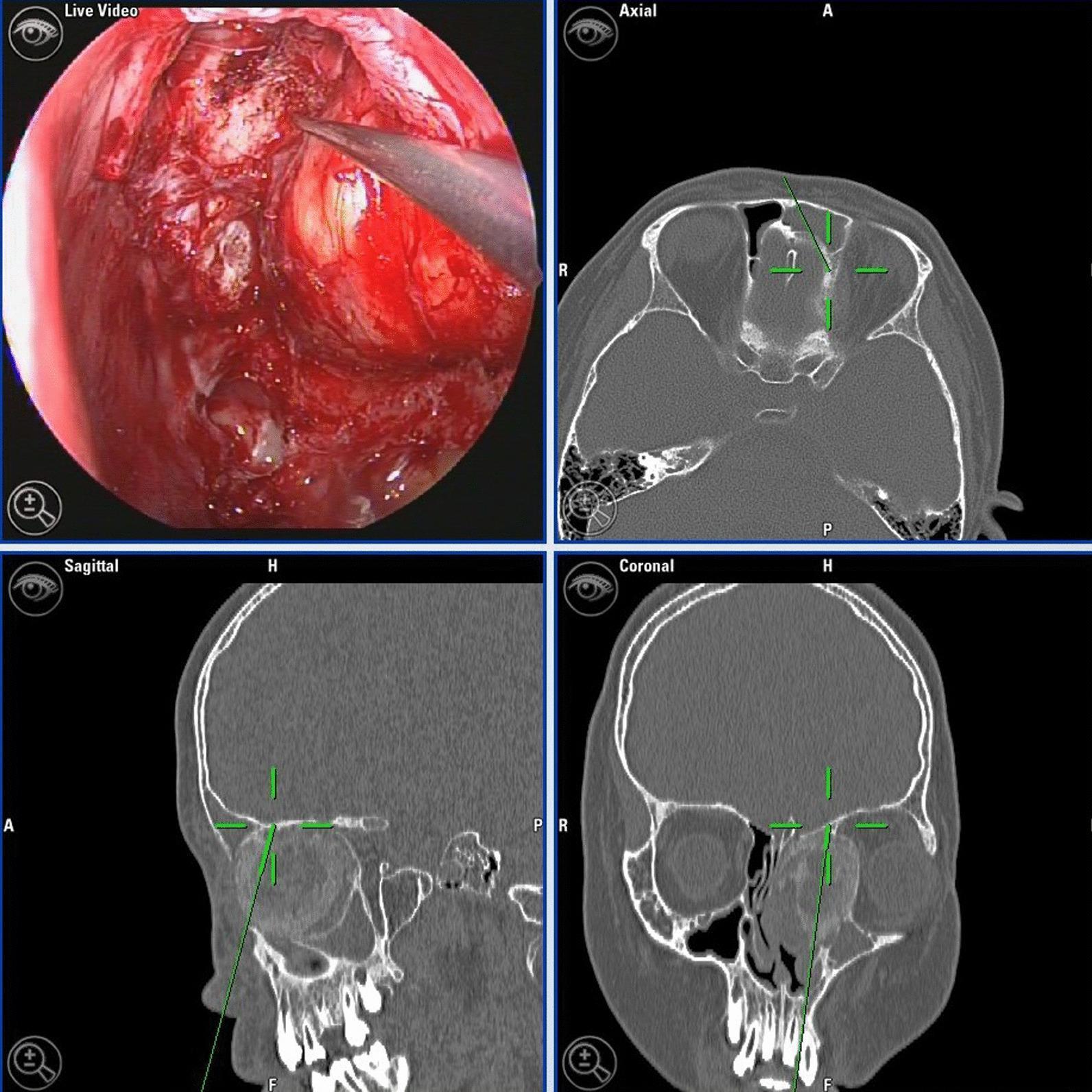
Components of an IGS System
| Component | Role |
|---|---|
| Preoperative CT/MRI | Imaging dataset (thin-cut ≤1 mm, no contrast for CT) |
| Workstation/computer | Image processing, 3D reconstruction, real-time tracking |
| Tracking system | Localises instrument and patient in space |
| Tracked instruments | Probe, suction, dissectors with integrated sensors |
| Reference frame/headset | Fixed to patient; tracks any patient movement |
| Display monitor | Shows live endoscope view + triplanar CT with crosshair |
Tracking Technologies
1. Electromagnetic (EM) Tracking
- A magnetic field generator placed near the patient's head creates a low-intensity magnetic field
- Instruments contain electromagnetic sensors that detect their position within the field
- Advantages: No line-of-sight requirement; instruments can be flexible/curved
- Disadvantages: Metal instruments, implants, and OR equipment cause field distortion (most significant limitation); less accurate near metallic objects
2. Optical (Infrared) Tracking
- Infrared cameras track retro-reflective spheres or LED arrays on instruments and a rigid head-frame fixed to the patient
- Advantages: High accuracy (~1–2 mm); not affected by metal
- Disadvantages: Requires uninterrupted line-of-sight between camera, instrument, and reference frame; any obstruction breaks tracking
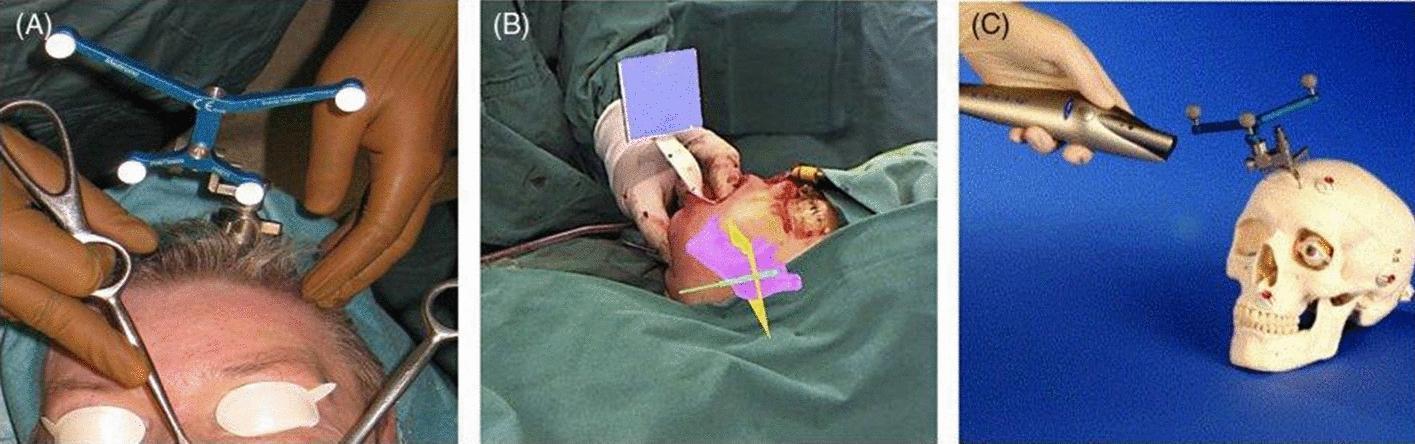
Registration
Methods:
- Fiducial-based registration: Adhesive markers (fiducials) placed on the patient's skin or bony landmarks before CT scanning. The same markers are touched intraoperatively with the navigation probe to establish spatial correspondence. Most commonly used.
- Surface registration (point-to-surface matching): A probe or laser scanner traces the patient's facial surface, which is matched to the CT-derived surface model. More convenient but slightly less precise.
- Anatomical landmark registration: Surgeon touches defined bony landmarks (nasion, medial canthus, etc.) to register the patient.
Indications (AAO-HNS Endorsed)
- Revision sinus surgery — distorted postoperative anatomy
- Distorted anatomy — developmental, traumatic, or prior surgical alteration
- Extensive sinonasal polyposis — obscured landmarks
- Disease involving frontal, posterior ethmoid, or sphenoid sinuses
- Pathology abutting the skull base, orbit, optic nerve, or internal carotid artery (ICA)
- CSF rhinorrhoea or skull base defect repair
- Benign and malignant sinonasal neoplasms
IGS is an adjunct, not a substitute for anatomical knowledge. It should not be used for routine uncomplicated primary ESS. — Cummings Otolaryngology Head and Neck Surgery
Applications in ENT
1. Endoscopic Sinus Surgery (ESS)
- Skull base (cribriform plate / fovea ethmoidalis)
- Orbit and lamina papyracea
- Optic nerve and internal carotid artery in sphenoid surgery
- Frontal recess anatomy in frontal sinusotomy
2. Skull Base Surgery
- Anterior skull base tumours (meningioma, olfactory neuroblastoma)
- Pituitary surgery (endoscopic transsphenoidal approach)
- Optic nerve decompression
- Confirms ICA position in sphenoid/petrous apex surgery
3. Otology / Lateral Skull Base
- Cochlear implant electrode insertion (confirms round window/cochleostomy position)
- Mastoid and posterior fossa approaches
- Petroclival and cerebellopontine angle surgery
4. Head & Neck Oncology
- Orbital decompression in thyroid eye disease
- Craniofacial surgery — orbital wall reconstruction, midface fractures
- Tumour margin confirmation during resection
Sources of Error
| Error Source | Detail |
|---|---|
| CT scanning variables | Slice thickness, gantry tilt, patient movement during scan |
| Fiducial/headset shift | Reference frame movement during surgery invalidates registration |
| Soft tissue deformation | Intraoperative changes not reflected in preoperative CT |
| Operator error | Inaccurate initial registration |
| Brain shift (neurosurgery) | Not applicable to rigid ENT structures but relevant in skull base |
Advantages & Limitations
| Advantages | Limitations |
|---|---|
| Reduces major complication rate (CSF leak, orbital injury, ICA injury) | Not a substitute for anatomical expertise |
| Enhances orientation in distorted anatomy | Accuracy limited to ~2 mm — not sub-millimetre |
| Reduces revision surgery rates | Increases operative time and cost |
| Real-time multiplanar 3D confirmation | Line-of-sight issues (optical) or metal interference (EM) |
| Improves surgeon confidence | Requires preoperative thin-cut CT |
Key Points for PG Exam
| Point | Fact |
|---|---|
| Accuracy threshold | ≤2 mm |
| Primary ENT application | Endoscopic sinus surgery (ESS) |
| Two tracking types | Electromagnetic vs. Optical (infrared) |
| EM disadvantage | Metal distortion of field |
| Optical disadvantage | Line-of-sight requirement |
| Registration | Fiducial-based (most common) |
| AAO-HNS endorsement | 7 specific indications |
| IGS limitation | Cannot replace anatomical knowledge |
| Most dangerous structures protected | ICA, optic nerve, skull base, orbit |
| When NOT to use | Routine, uncomplicated primary ESS |
Essay for "Physiology of Olfaction - pathway & Tests of Olfaction" for ENT exam with flowcharts/diagrams- images under 800 words
olfactory pathway anatomy diagram olfactory bulb cortex cribriform plate nerve
Sniffin Sticks UPSIT olfactory test smell identification threshold
Physiology of Olfaction — Pathway & Tests of Olfaction
Introduction
Receptor Organ — Olfactory Neuroepithelium
Cell Types in Olfactory Epithelium:
| Cell Type | Function |
|---|---|
| Olfactory Sensory Neurons (OSNs) | Bipolar receptor cells; each serves as receptor + 1st-order neuron |
| Basal cells | Stem cells; regenerate OSNs throughout life |
| Sustentacular cells | Secrete mucopolysaccharides → dissolve odorants for receptor binding |
| Bowman's glands | Secrete protective mucus layer |
| Pseudostratified columnar epithelium | Structural support |
Olfactory Transduction (Molecular Mechanism)
Odorant molecule (air phase)
↓
Dissolves into aqueous olfactory mucus
↓
Odorant binds G-protein coupled receptor (GPCR) on OSN dendritic cilia
↓
Gαolf activates adenylyl cyclase → ↑ cAMP
↓
cAMP opens cyclic nucleotide-gated (CNG) cation channels
↓
Influx of Na⁺ and Ca²⁺ → membrane depolarisation
↓
Ca²⁺ opens Cl⁻ channels → outflow of Cl⁻ (amplification)
↓
Action potential generated in OSN axon
Olfactory Pathway
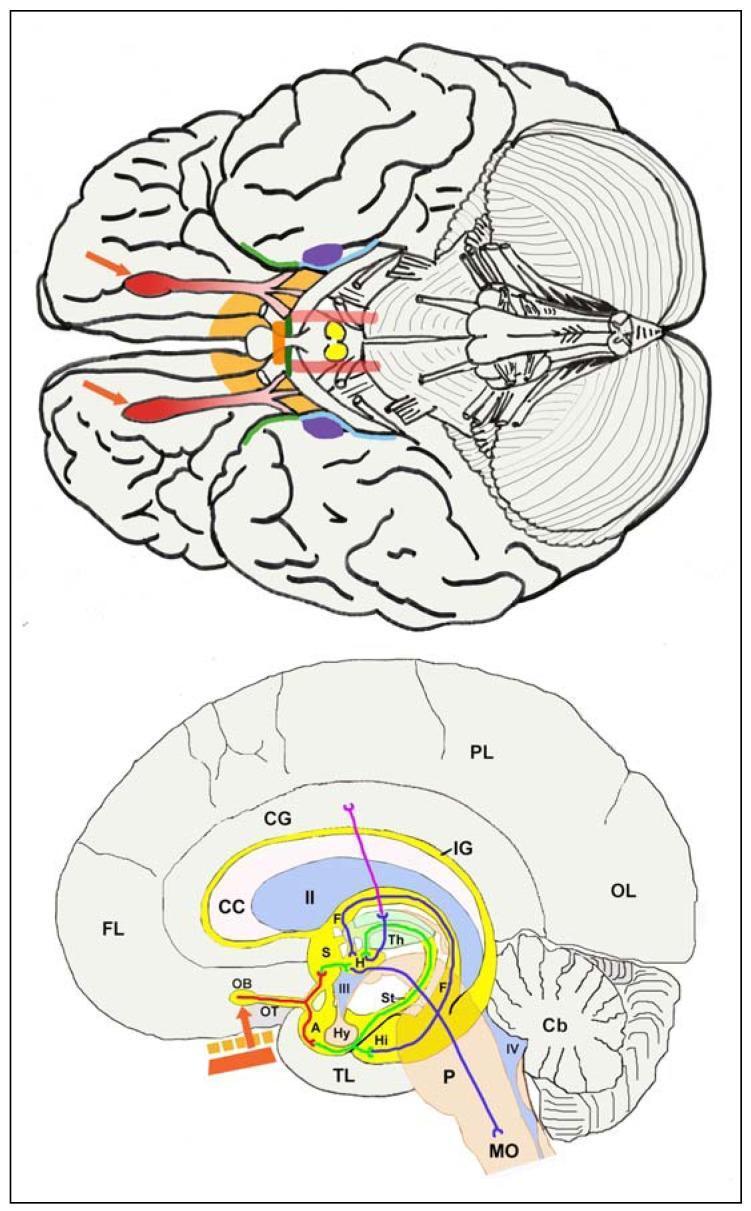
Step-by-Step Pathway:
Odorant binds OSN cilia (Olfactory Neuroepithelium)
↓ [1st-order neurons — CN I fila]
Axons traverse CRIBRIFORM PLATE of ethmoid bone
↓
OLFACTORY BULB
├── Axons synapse in GLOMERULI
├── Mitral cells + Tufted cells (2nd-order neurons)
├── Periglomerular cells (inhibitory, GABA) — lateral inhibition
└── Granule cells (inhibitory, GABA) — sharpens odour signals
↓ [Lateral olfactory stria]
OLFACTORY TRACT → PRIMARY OLFACTORY CORTEX (5 areas):
├── Anterior olfactory nucleus
├── Olfactory tubercle
├── PIRIFORM CORTEX (largest; odour quality, identity, memory)
├── AMYGDALA (emotional/affective responses to odour)
└── ENTORHINAL CORTEX (memory preprocessing → hippocampus)
↓
├── Directly → FRONTAL CORTEX (conscious discrimination)
├── Via thalamus → ORBITOFRONTAL CORTEX (interpretive processing; Brodmann areas 10, 11, 25)
└── → HIPPOCAMPUS (olfactory learning and memory)
Key fact: Olfaction is the ONLY special sense that does NOT relay through the thalamus before reaching primary cortex. This explains its unique emotional and mnemonic significance (Proust phenomenon). — Ganong's Review of Medical Physiology; K.J. Lee's Essential Otolaryngology
Trigeminal Component
Types of Olfactory Sensation
| Type | Route | Association |
|---|---|---|
| Orthonasal | Sniffing → air to olfactory cleft | Detection of environmental odours |
| Retronasal | Chewing/swallowing → oropharynx → nasopharynx → olfactory cleft | Flavour perception (taste + smell) |
Terminology of Olfactory Dysfunction
| Term | Definition |
|---|---|
| Anosmia | Complete loss of smell |
| Hyposmia | Reduced sense of smell |
| Hyperosmia | Increased sensitivity to odours |
| Dysosmia / Parosmia | Distorted smell (present stimulus misidentified) |
| Phantosmia | Olfactory hallucination (no stimulus present) |
| Presbyosmia | Age-related smell decline |
Tests of Olfaction
1. Subjective / Psychophysical Tests
A. University of Pennsylvania Smell Identification Test (UPSIT)
- Gold standard for clinical olfactory testing
- 40-item forced-choice scratch-and-sniff microencapsulated odour booklet
- Patient identifies odour from 4 options per item; scored /40
- Normative data stratified by age and sex (women outperform men at all ages)
- Categories: Normosmia / Microsmia (mild, moderate, severe) / Anosmia / Malingering
B. Sniffin' Sticks (TDI Battery)
- T = Threshold: Minimum concentration detectable (n-butanol dilution series)
- D = Discrimination: Ability to distinguish between pairs of odours
- I = Identification: Naming 16 common odours from 4 options
- TDI composite score ≥30.75 = normosmia; <16.5 = functional anosmia
C. Connecticut Chemosensory Clinical Research Center (CCCRC) Test
- Combines odour threshold (n-butanol) + odour identification tasks
- Scored 0–7; differentiates conductive vs. sensorineural olfactory loss
D. Brief Smell Identification Test (B-SIT / Cross-Cultural Smell Identification Test, CC-SIT)
- 12-item abbreviated version of UPSIT
- Useful for rapid screening in clinical settings
2. Objective Tests
A. Electro-Olfactogram (EOG)
- Records electrical potentials from the olfactory epithelium surface in response to odorant stimulation
- Summated generator potentials from OSN receptor membranes
- Used in research; not routine clinical practice
B. Olfactory Event-Related Potentials (OERPs)
- EEG-based cortical response to carefully controlled olfactory stimuli
- Useful when malingering is suspected (cannot be faked)
- Latency ~400–600 ms; amplitude correlates with odour intensity
C. fMRI / PET Scan
- Imaging of olfactory bulb volume and cortical activation
- Olfactory bulb atrophy correlates with degree of anosmia
- Used in research and in evaluating post-traumatic or neurodegenerative olfactory loss
3. Clinical Bedside Testing
- Patient asked to identify common household odours (coffee, peppermint, clove, lemon) with eyes closed, one nostril at a time
- Avoids trigeminal stimulants (ammonia, alcohol — test CN V, not CN I)
- Documents unilateral vs. bilateral loss
Common Causes of Olfactory Loss (for ENT exam)
| Category | Examples |
|---|---|
| Conductive (transport) | Rhinosinusitis, nasal polyps, septal deviation |
| Sensorineural (neural) | Head trauma (shearing of olfactory fila at cribriform plate), viral URTI, COVID-19, aging |
| Central | Parkinson's disease, Alzheimer's, frontal lobe tumours |
| Endocrine/metabolic | Hypothyroidism, Addison's disease, zinc deficiency |
The three most common identifiable causes of lasting smell loss: URI > Head trauma > Chronic rhinosinusitis — Harrison's Principles of Internal Medicine
Summary Flowchart
ODORANT
↓
Olfactory Epithelium (OSN + sustentacular cells + Bowman's glands)
↓ [GPCR → cAMP → depolarisation]
ACTION POTENTIAL in OSN axon (CN I fila)
↓ [Cribriform plate]
OLFACTORY BULB (glomeruli → mitral/tufted cells)
↓ [Lateral olfactory stria]
PRIMARY OLFACTORY CORTEX
├── Piriform cortex → Odour identity/memory
├── Amygdala → Emotional response
├── Entorhinal → Hippocampus (memory)
└── Orbitofrontal cortex → Conscious discrimination
Essay for "Anatomy of Lateral wall of Nose & Septum Anatomy"
lateral wall nose anatomy turbinates meatus diagram nasal cavity
nasal septum anatomy cartilage quadrangular vomer perpendicular plate ethmoid
ostiomeatal complex hiatus semilunaris uncinate process ethmoid bulla middle meatus diagram
blood supply nasal cavity sphenopalatine artery Kiesselbach plexus Little area nerve supply
Anatomy of the Lateral Wall of the Nose & Nasal Septum
PART 1 — NASAL SEPTUM
Components of the Nasal Septum
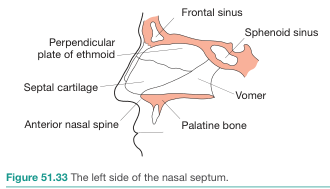
NASAL SEPTUM
├── CARTILAGINOUS
│ └── Quadrangular (septal) cartilage — anterior; forms the mobile columella
│
├── BONY
│ ├── Perpendicular plate of ethmoid — posterosuperior
│ ├── Vomer — posteroinferior
│ ├── Nasal crest of maxilla — inferior (anterior)
│ ├── Nasal crest of palatine bone — inferior (posterior)
│ ├── Rostrum of sphenoid — posterosuperior
│ └── Nasal bones — anterosuperior
│
└── MEMBRANOUS
└── Mobile columella (between anterior septal cartilage and medial crura of alar cartilages)
Key Relationships
- The quadrangular cartilage sits in a groove between the vomer inferiorly and the perpendicular plate of ethmoid superiorly
- It articulates anteriorly with the nasal bones and medial crura of the lower lateral (alar) cartilages
- The nasal spine of the maxilla supports the base of the septal cartilage anteriorly
- Septal deviation most commonly occurs at the junction of the vomer and cartilage — the osseocartilaginous junction — Gray's Anatomy for Students
Blood Supply of the Nasal Septum
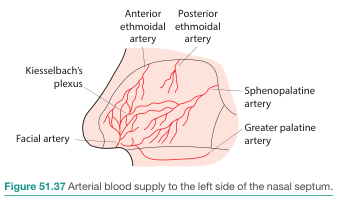
| Artery | Origin | Region Supplied |
|---|---|---|
| Anterior ethmoidal | Ophthalmic (internal carotid) | Anterosuperior septum |
| Posterior ethmoidal | Ophthalmic (internal carotid) | Posterosuperior septum |
| Sphenopalatine | Maxillary (external carotid) | Posterior septum (largest supply) |
| Greater palatine | Maxillary (external carotid) | Inferior septum — enters via incisive foramen |
| Superior labial | Facial (external carotid) | Anteroinferior septum |
Kiesselbach's Plexus (Little's Area)
Nerve Supply of the Nasal Septum
| Nerve | Origin | Region |
|---|---|---|
| Anterior ethmoidal nerve | CN V₁ (nasociliary) | Anterosuperior septum |
| Nasopalatine nerve | CN V₂ (sphenopalatine ganglion) | Posteroinferior septum → incisive foramen |
| Olfactory nerves (CN I) | — | Olfactory area (superior septum) |
PART 2 — LATERAL WALL OF THE NOSE
Bones Forming the Lateral Wall
LATERAL WALL — Bony Support:
├── Ethmoidal labyrinth (superior & middle conchae + uncinate process)
├── Perpendicular plate of the palatine bone (posterior)
├── Medial pterygoid plate of sphenoid (posteriormost)
├── Medial surfaces of lacrimal bones and maxillae (anterolateral)
└── Inferior concha (independent bone — largest turbinate)
The Turbinates (Conchae)
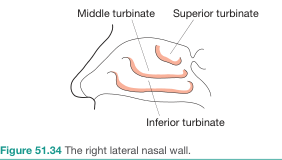
| Turbinate | Origin | Air Channel Below | Key Openings |
|---|---|---|---|
| Inferior turbinate | Independent bone (inferior nasal concha) — largest | Inferior meatus | Nasolacrimal duct (under anterior lip) |
| Middle turbinate | Ethmoid bone | Middle meatus | Frontal, maxillary, anterior ethmoid sinuses |
| Superior turbinate | Ethmoid bone | Superior meatus | Posterior ethmoidal cells |
| (Occasionally: supreme turbinate) | Ethmoid | Spheno-ethmoidal recess | Sphenoidal sinus |
Note: The sphenoidal sinus is the only paranasal sinus that does NOT drain onto the lateral wall — it opens onto the sloping posterior roof of the nasal cavity (sphenoethmoidal recess). — Gray's Anatomy for Students
Middle Meatus — The Most Important Region
Key Structures of the Middle Meatus:
MIDDLE MEATUS (lateral wall, key landmarks)
│
├── UNCINATE PROCESS — thin, hook-like projection of ethmoid bone
│ (anterosuperior landmark; resected first in FESS)
│
├── HIATUS SEMILUNARIS (Semilunar Hiatus)
│ — Curved gutter between ethmoidal bulla (above) and uncinate process (below)
│ — Opens into → ETHMOIDAL INFUNDIBULUM
│
├── ETHMOIDAL INFUNDIBULUM
│ — Funnel-shaped channel at anterior end of hiatus semilunaris
│ — Continues upward as FRONTONASAL DUCT → Frontal sinus
│ — Maxillary sinus ostium opens here (near center of hiatus)
│
├── ETHMOIDAL BULLA
│ — Dome-shaped prominence formed by underlying middle ethmoidal air cells
│ — Largest and most constant ethmoid air cell
│ — Middle ethmoid cells open onto or just ABOVE the bulla
│
└── FRONTAL RECESS
— Narrow passage superiorly leading to frontal sinus
Sinus Drainage Summary:
| Sinus | Drains Into |
|---|---|
| Frontal sinus | Frontonasal duct → Ethmoidal infundibulum → Anterior hiatus semilunaris (middle meatus) |
| Maxillary sinus | Semilunar hiatus (middle meatus) — near roof of sinus |
| Anterior ethmoidal cells | Ethmoidal infundibulum / frontonasal duct |
| Middle ethmoidal cells | On or above ethmoidal bulla (middle meatus) |
| Posterior ethmoidal cells | Superior meatus |
| Sphenoidal sinus | Spheno-ethmoidal recess (above superior turbinate) |
| Nasolacrimal duct | Inferior meatus (under anterior lip of inferior concha) |
Blood Supply of the Lateral Wall
| Artery | Origin | Region |
|---|---|---|
| Anterior ethmoidal | Ophthalmic (ICA) | Anterosuperior lateral wall |
| Posterior ethmoidal | Ophthalmic (ICA) | Posterosuperior lateral wall |
| Sphenopalatine | Maxillary (ECA) | Posterior lateral wall — emerges posterior to middle turbinate |
| Greater palatine | Maxillary (ECA) | Inferior lateral wall |
The sphenopalatine artery is the most common source of posterior epistaxis — emerges through the sphenopalatine foramen posterior to the middle turbinate. — Roberts & Hedges' Clinical Procedures
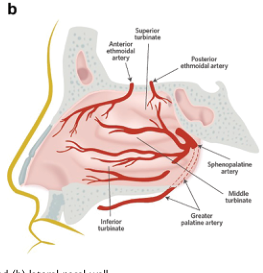
Nerve Supply of the Lateral Wall
| Nerve | Foramen/Route | Area |
|---|---|---|
| Anterior ethmoidal (CN V₁) | Anterior ethmoidal foramen | Anterosuperior lateral wall |
| Lateral posterior superior nasal branches (CN V₂) | Sphenopalatine foramen | Superior + middle turbinates, posterior superior meati |
| Lateral posterior inferior nasal branches (CN V₂) | Greater palatine canal | Inferior turbinate, inferior meatus |
| Olfactory (CN I) | Cribriform plate | Olfactory region (upper 1/3) |
Choanae
- Inferiorly: Horizontal plate of the palatine bone
- Laterally: Medial pterygoid plate of sphenoid
- Medially: Posterior border of the vomer
- Roof: Ala of vomer + vaginal process of medial pterygoid plate (anteriorly); body of sphenoid (posteriorly)
Summary Table
| Feature | Septum | Lateral Wall |
|---|---|---|
| Cartilage | Quadrangular cartilage | Lateral process of septal cartilage, alar cartilages |
| Key bones | Perpendicular plate, vomer | Ethmoid, inferior concha, palatine, lacrimal, maxilla |
| Arterial anastomosis site | Kiesselbach's plexus (anterior) | Sphenopalatine foramen (posterior) |
| Largest artery | Sphenopalatine | Sphenopalatine |
| Olfactory epithelium | Superior septum | Superior turbinate/cleft |
| Most important meatus | — | Middle meatus (OMC) |
| Sinus NOT draining here | — | Sphenoidal sinus (drains to spheno-ethmoidal recess) |