Teach me thallasemia pathology, el5
Reading File
search_medical_text
thalassemia pathophysiology pathology alpha beta globin chain imbalance hemolysis
search_medical_image
thalassemia pathophysiology diagram globin chain imbalance
GradeResponse
GradeResponse
Here's thalassemia pathology, explained like you're five (but not so five that it's useless):
🩸 Thalassemia Pathology — ELI5
The One-Sentence Version
Your red blood cells are supposed to be built from two matching Lego pieces (alpha and beta globin chains). In thalassemia, one piece doesn't get made enough — so you end up with a pile of unmatched pieces that break everything.
Step 1: Normal Hemoglobin = A Balanced Team
Normal hemoglobin (adult Hb = HbA) is made of 2 alpha chains + 2 beta chains, like a 4-person rowing team. Everyone has a partner, the boat moves perfectly.
Step 2: What Goes Wrong in Thalassemia
There are two main types, based on which chain is broken:
| Type | What's deficient | What accumulates |
|---|---|---|
| α-thalassemia | Alpha chains (gene deletion) | Excess beta chains → form HbH (β4 tetramers) |
| β-thalassemia | Beta chains (gene mutation) | Excess alpha chains (the trouble-maker) |
β-thalassemia is the most clinically important, so let's focus there.
Step 3: The Chaos Caused by Unpaired Alpha Chains
(Harrison's, p. 2927)
When beta chains are reduced, alpha chains pile up and have no partner. Here's what happens next:
-
Alpha chains are unstable alone — they can't form a tetramer, so they just... collapse and precipitate (clump up like curdled milk) inside the developing red blood cell (erythroblast).
-
They damage the cell membrane — through lipid oxidation (think rust forming on iron). The cell becomes fragile.
-
The bone marrow destroys them before they even launch — this is called ineffective erythropoiesis (you're making red blood cells, but killing them in the factory before they ship). This is the #1 cause of anemia in β-thalassemia.
-
Some cells escape into the blood, but die early — either inside blood vessels (intravascular hemolysis) or in the spleen (extravascular hemolysis), because they're too rigid and deformed to survive.
Step 4: The Body's Panicked Overreaction
The body sees severe anemia and freaks out:
- Bone marrow expands wildly trying to make more RBCs → causes the classic "chipmunk facies" (frontal bossing, prominent cheekbones from skull marrow expansion)
- Liver and spleen enlarge (hepatosplenomegaly) — they try to compensate by making blood cells extramedullarily
- Hepcidin is suppressed (due to a signal called ERFE from the overactive marrow) → the gut absorbs more iron even though there's no place for it → iron overload accumulates in the heart, liver, and endocrine glands
- This leads to: heart failure, cirrhosis, diabetes, hypogonadism (endocrine stuff)
The Big Picture (Visual)
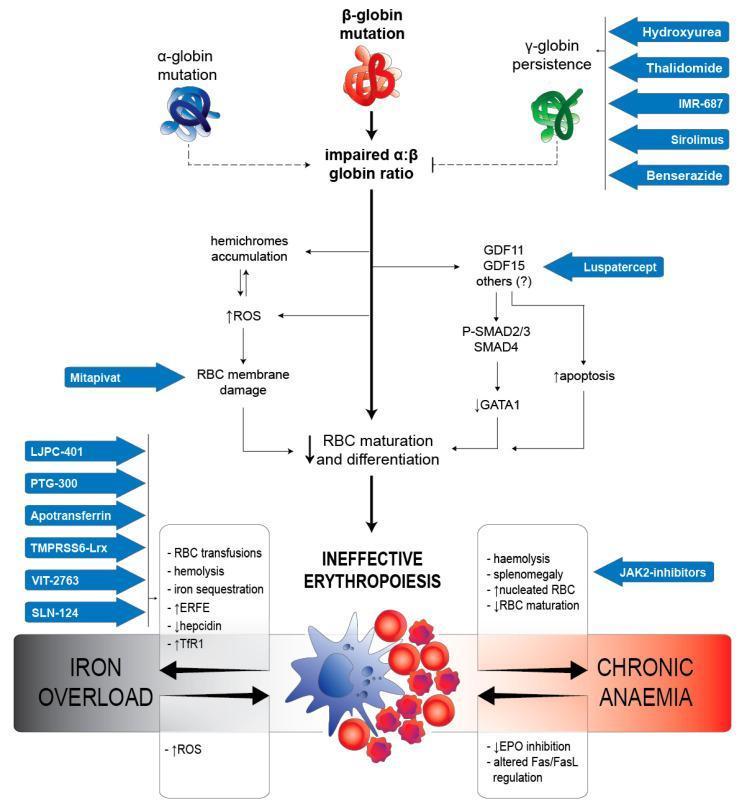
This diagram shows the full cascade: globin mutation → alpha:beta imbalance → two parallel damage highways (ROS/membrane damage + apoptosis via GDF signaling) → both leading to ineffective erythropoiesis → anemia + iron overload.
Quick Summary Table
| Problem | Mechanism | Clinical Result |
|---|---|---|
| Excess unpaired α-chains | Precipitate, oxidize membrane | Cell death in marrow (ineffective erythropoiesis) |
| Fragile RBCs that survive | Hemolysis in spleen/blood | Anemia |
| Body makes more marrow | Massive erythroid expansion | Bone deformity, hepatosplenomegaly |
| Hepcidin suppressed | Gut absorbs more iron | Iron overload → organ damage |
α-Thalassemia in Brief
Alpha genes come in 4 copies (2 per chromosome 16). Severity depends on how many are deleted:
- 1 gene deleted: silent carrier — no symptoms
- 2 genes deleted: mild anemia (α-thalassemia trait)
- 3 genes deleted: HbH disease — moderate hemolytic anemia (β4 tetramers form, unstable)
- 4 genes deleted: Hb Bart's hydrops fetalis — incompatible with life (γ4 tetramers, no functional hemoglobin)
The core concept to remember: thalassemia = globin chain imbalance → the excess chain wrecks the RBC from the inside out. Everything else (anemia, bone changes, iron overload) flows downstream from that one problem.
Tell me about hepcidin
search_medical_text
hepcidin mechanism action iron regulation synthesis
search_medical_image
hepcidin iron regulation pathway diagram
GradeResponse
GradeResponse
Hepcidin — The Master Iron Hormone
What It Is
Hepcidin is a small liver-derived peptide hormone — the body's master regulator of iron. Its name comes from its origin and an early-discovered property: HEPatic bacteriCIDal proteIN. Its iron-regulatory role turned out to be far more important than any antimicrobial activity.
The core job: keep total body iron in balance (normally 3–4 g) by controlling how much iron enters the bloodstream from three sources:
- Intestinal absorption (gut enterocytes)
- Recycling from old RBCs (macrophages in spleen/liver)
- Release from liver storage (hepatocytes)
The Molecular Lock: Ferroportin
Hepcidin works through a single effector protein — ferroportin (FPN1) — the only known iron exporter in mammalian cells.
| When hepcidin is HIGH | When hepcidin is LOW |
|---|---|
| Binds ferroportin → triggers its internalization and degradation | Ferroportin stays on cell surface |
| Iron stays trapped inside enterocytes, macrophages, liver | Iron is exported into plasma freely |
| Serum iron ↓, ferritin ↑ | Serum iron ↑ |
Think of hepcidin as a padlock on the iron door. High hepcidin = door locked shut. Low hepcidin = door wide open.
What Turns Hepcidin ON (Stimuli to Increase It)
(Harrison's, p. 11768)
| Stimulus | Signal | Why it makes sense |
|---|---|---|
| High iron stores | BMP6, BMP2 (from liver sinusoidal cells) via BMP/SMAD pathway | Body has enough iron — close the gate |
| High transferrin saturation | TfR2 + HFE sensing Fe³⁺-Tf | Circulating iron is high |
| Inflammation/infection | IL-6 → JAK2/STAT3 pathway → HAMP gene transcription | Starve bacteria of iron (nutritional immunity) |
What Turns Hepcidin OFF (Stimuli to Suppress It)
| Stimulus | Signal | Why it makes sense |
|---|---|---|
| Iron deficiency | Low BMP6 | Body needs more iron — open the gate |
| Hypoxia | HIF pathway | Need more RBCs → need more iron |
| Increased erythropoiesis | ERFE (erythroferrone, from erythroblasts) | Bone marrow is expanding → feed it iron |
| Anemia | Indirect, via ERFE | Same as above |
ERFE is the key link to thalassemia — in β-thalassemia, the massively overactive (but ineffective) marrow pumps out huge amounts of ERFE, which hammers hepcidin down, causing unregulated iron absorption even when stores are already overloaded.
The Signaling Pathway (Visual)
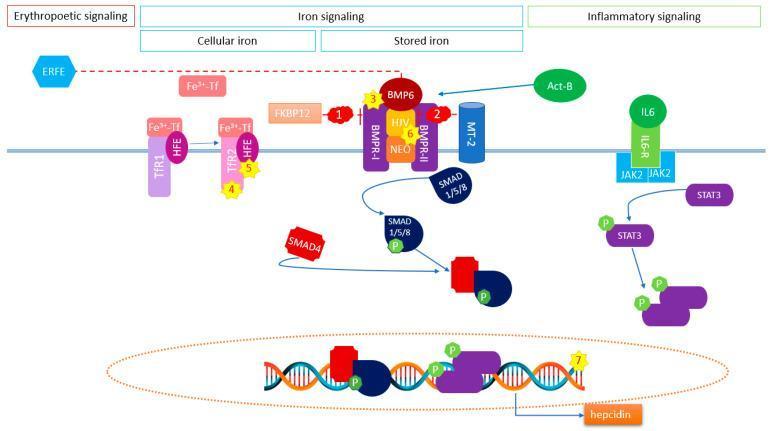
The diagram shows the three converging inputs into hepcidin transcription (the HAMP gene):
- Iron signaling: BMP6/Activin B → BMPR-I/II + HJV co-receptor → SMAD1/5/8 phosphorylation → SMAD4 complex → nucleus
- Inflammatory signaling: IL-6 → IL6-R → JAK2 → STAT3 phosphorylation → nucleus
- Both SMAD complex and pSTAT3 bind the HAMP promoter and drive hepcidin transcription
- ERFE (erythropoietic signal) inhibits this cascade (dotted red line)
Disease States — When Hepcidin Goes Wrong
| Condition | Hepcidin Level | Result |
|---|---|---|
| Hereditary hemochromatosis (HFE mutation, HJV mutation, etc.) | Inappropriately LOW | Iron overload — gut keeps absorbing, stores keep filling |
| Anemia of chronic disease/inflammation | Inappropriately HIGH | Iron trapped in macrophages, unavailable for erythropoiesis → functional iron deficiency |
| β-thalassemia | Suppressed by ERFE | Iron overload despite already high stores |
| IRIDA (Iron-Refractory Iron Deficiency Anemia — TMPRSS6 loss-of-function) | Inappropriately HIGH | Can't suppress hepcidin even when iron-deficient → iron deficiency unresponsive to oral iron |
One-Line Summary
Hepcidin is the liver's way of saying "enough iron" or "we need more" — it locks or unlocks the only iron exit door (ferroportin) based on signals from iron stores, oxygen levels, and the bone marrow's demand.
Iron absorption and utilisation
search_medical_text
iron absorption duodenum enterocyte DMT1 ferroportin dietary iron
search_medical_image
iron absorption intestinal enterocyte diagram
GradeResponse
GradeResponse
Iron Absorption and Utilisation
The Big Picture
The body has no active iron excretion mechanism. The only way to regulate iron is to control how much gets absorbed in the first place — and that's hepcidin's job (as we just covered). Everything downstream is about distribution and recycling.
Step 1: Dietary Iron — Two Forms
| Type | Source | Absorption rate | Notes |
|---|---|---|---|
| Haem iron (Fe²⁺ in haem) | Red meat, liver, blood | ~25–30% | Absorbed directly as intact haem via HCP1; highly bioavailable |
| Non-haem iron (Fe³⁺) | Plants, fortified foods, supplements | ~5–10% | Must be reduced first; affected by diet enhancers/inhibitors |
Enhancers of non-haem absorption: Vitamin C (ascorbate — reduces Fe³⁺ → Fe²⁺), acidic stomach environment, meat factor
Inhibitors: Phytates (whole grains), polyphenols (tea, coffee), calcium, antacids/PPIs (raise gastric pH)
Step 2: Absorption in the Duodenal Enterocyte
(Harrison's, p. 11766)
This is where the action happens — specifically the duodenum and upper jejunum.
Apical (luminal) side:
- Dietary Fe³⁺ is reduced to Fe²⁺ by DCYTB (duodenal cytochrome B, a ferrireductase on the brush border)
- Fe²⁺ enters the enterocyte via DMT1 (divalent metal transporter 1)
Inside the enterocyte:
- Iron is either stored temporarily in ferritin (if body doesn't need it — lost when the enterocyte is shed ~3 days later)
- Or trafficked to the basolateral membrane for export
Basolateral (blood) side:
- Iron exits via ferroportin (FPN1) — the only iron exporter
- Fe²⁺ is re-oxidised to Fe³⁺ by hephaestin (a copper-dependent ferroxidase)
- Fe³⁺ binds transferrin in the plasma
Step 3: Transport in the Blood — Transferrin
- Transferrin is the plasma iron carrier (made in the liver)
- Each transferrin molecule carries up to 2 Fe³⁺ ions
- Normal transferrin saturation = ~20–35% (lots of spare capacity)
- When saturation exceeds ~75–80% (iron overload states), NTBI (non-transferrin-bound iron) appears → freely enters cells → generates free radicals → organ damage
Step 4: Utilisation — Where Does Iron Go?
| Destination | Amount | Purpose |
|---|---|---|
| Bone marrow erythroblasts | ~20–25 mg/day | Haemoglobin synthesis (the dominant consumer) |
| Liver (hepatocytes) | Storage | Stored as ferritin and haemosiderin |
| Muscle | Small amount | Myoglobin synthesis |
| All cells | Tiny amount | Mitochondrial cytochromes, enzymes (catalase, ribonucleotide reductase) |
The bone marrow is by far the biggest iron consumer — ~70% of total body iron is locked inside haemoglobin in circulating RBCs at any given time.
Step 5: Recycling — The Iron Economy
This is the most important concept: the body is extraordinarily efficient at recycling iron.
Daily iron need for erythropoiesis: ~20–25 mg/day
Daily dietary absorption: ~1–2 mg/day
→ The other ~23 mg comes from RECYCLING senescent RBCs
How recycling works:
- RBCs live ~120 days, then get phagocytosed by macrophages in the spleen, liver (Kupffer cells), and bone marrow
- Macrophages break down haemoglobin → haem is catabolised by haem oxygenase → releases Fe²⁺
- Iron is exported back to plasma via ferroportin (again, the gatekeeper)
- Transferrin picks it up → back to the bone marrow
Hepcidin controls this recycling too — high hepcidin locks ferroportin on macrophages, trapping iron inside them (this is exactly what happens in anaemia of chronic disease).
Step 6: Storage
| Protein | Location | Function |
|---|---|---|
| Ferritin | Liver, spleen, bone marrow, serum | Soluble, safe iron storage; serum ferritin reflects stores |
| Haemosiderin | Liver, spleen (when overloaded) | Insoluble aggregate of degraded ferritin; marker of iron overload |
Serum ferritin: ~12–300 µg/L (men), ~12–150 µg/L (women). But ferritin is also an acute phase reactant — it rises in inflammation regardless of iron stores, which complicates interpretation.
Full Iron Flow Summary
Diet (Fe³⁺)
↓ DCYTB reduces → Fe²⁺
↓ DMT1 into enterocyte
↓ Ferroportin + hephaestin out → Fe³⁺
↓ Binds transferrin in plasma
↓
├──→ Bone marrow → Haemoglobin synthesis (70% of body iron)
├──→ Liver → Ferritin/haemosiderin storage
└──→ Muscle → Myoglobin
↓
RBC lives 120 days → Macrophage phagocytosis
↓ Haem oxygenase
Fe²⁺ released → Ferroportin → Transferrin → Cycle repeats
Tying It Back to Thalassemia
In β-thalassemia, this system breaks down at the hepcidin-ERFE axis:
- Ineffective erythropoiesis → massive ERFE release → hepcidin suppressed → ferroportin stays open on both enterocytes AND macrophages → iron floods in from both diet and recycling → progressive iron overload even without transfusions
This is why thalassaemia patients develop iron overload even before they receive any blood transfusions — the absorption pathway is simply stuck open.
This is a shared conversation. Sign in to Orris to start your own chat.