CIN AND CERVICAL CANCER IN DETAIL
Reading File
Finding Sources
Searching Images
cervical intraepithelial neoplasia CIN histology grading cervical cancer
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
I now have comprehensive content from multiple authoritative textbooks. Let me compile the full, detailed response:
Cervical Intraepithelial Neoplasia (CIN) and Cervical Cancer
1. Historical Background
The concept of preinvasive cervical disease was introduced in 1947, when pathologists identified features suggestive of invasive cancer confined entirely to the epithelium. The term cervical intraepithelial neoplasia (CIN) was coined by Richart in 1968, who proposed that dysplasias have the potential to progress to invasive cancer. Historically, carcinoma in situ (CIS) was treated aggressively (often with hysterectomy), while dysplasias were considered less significant. CIN unified these entities into a spectrum.
— Berek & Novak's Gynecology
2. Anatomy: The Transformation Zone
The cervix is composed of two epithelial types:
- Columnar (glandular) epithelium — lines the endocervical canal
- Squamous epithelium — covers the exocervix
The squamocolumnar junction (SCJ) is the point at which these two epithelia meet. It is not static — it migrates in response to puberty, pregnancy, menopause, and hormonal stimulation.
At menarche, estrogen causes vaginal epithelium to fill with glycogen. Lactobacilli act on glycogen to lower pH, stimulating subcolumnar reserve cells to undergo squamous metaplasia. This metaplasia advances from the original SCJ inward, toward the external os and over the columnar villi, creating the transformation zone (TZ).
CIN almost always arises in the transformation zone at the advancing SCJ. The anterior lip is twice as likely to develop CIN as the posterior lip. Once established, CIN may extend horizontally to involve the entire TZ. The healed (mature, glycogenated) transformation zone is relatively resistant to oncogenic stimuli.
— Berek & Novak's Gynecology, p. 834–835
3. Pathology and Grading of CIN
The significant histologic features of CIN are:
- Cellular immaturity
- Cellular disorganization
- Nuclear abnormality (enlargement, hyperchromasia, pleomorphism)
- Increased mitotic activity (abnormal mitotic figures may be seen)
Grading is based on the extent of mitosis and immature cellular proliferation in the epithelial thickness:
| Grade | Epithelial Involvement | Bethesda Equivalent |
|---|---|---|
| CIN 1 | Lower ⅓ of epithelium | LSIL (Low-grade SIL) |
| CIN 2 | Lower ⅔ of epithelium | HSIL (High-grade SIL) |
| CIN 3 | Upper ⅓ to full thickness (includes CIS) | HSIL |
CIN 3 encompasses carcinoma in situ: the full thickness of the epithelium shows abnormal immature cells with mitoses at all levels. The basement membrane remains intact (no invasion).
The ASCCP and College of American Pathologists recommend a two-tier grading system — LSIL and HSIL — for biopsied lesions, reflecting that CIN 2 and CIN 3 share similar management.
— Berek & Novak's Gynecology, p. 834; Sabiston Textbook of Surgery
CIN grading diagram (Pap smear nuclear progression):

4. Human Papillomavirus (HPV) — The Central Cause
Virology
HPV is a small, nonenveloped, double-stranded DNA virus protected by a capsid formed by late proteins L1 and L2. Over 200 strains exist; ~40 infect the genital tract:
- Low-risk HPV (e.g., types 6, 11): causes genital warts
- High-risk (oncogenic) HPV (e.g., types 16, 18, 31, 33, 45, 52, 58): causes anogenital, cervical, and oropharyngeal cancers
HPV 16 and 18 alone account for ~70% of cervical cancers. Types 31, 33, 45, 52, and 58 cause an additional ~20%.
Molecular Oncogenesis
HPV infects basal cells of the stratified squamous epithelium at the TZ. The E6 and E7 oncoproteins drive malignant transformation:
| Oncoprotein | Target | Effect |
|---|---|---|
| E6 | p53 (tumor suppressor) | Binds and degrades p53 → inhibits apoptosis → cellular immortalization |
| E7 | pRB (retinoblastoma protein) + cyclins/CDKs | Degrades pRB → disrupts cell cycle regulation → uncontrolled proliferation |
Both E6 and E7 also have immunosuppressive effects, contributing to immune evasion and peripheral tolerance in cytotoxic T lymphocytes.
Natural History of HPV Infection
- Most infections are transient, asymptomatic, and self-clearing within 1–2 years (9–15 months)
- In ~10% of women, HPV persists and produces viral proteins → persistent infection
- Persistent high-risk HPV infection is a necessary (but not sufficient) cause of cervical cancer
- Average time from HPV infection to invasive cancer: >15 years
- HPV DNA can integrate into host cell DNA or persist as an episome; integration is associated with higher malignant potential
— Goldman-Cecil Medicine, p. 2088–2089; Berek & Novak's Gynecology, p. 835
5. Risk Factors
| Category | Specific Factors |
|---|---|
| HPV acquisition | Early sexual debut, multiple sexual partners, history of STIs (including HIV), genital warts |
| Persistence of HPV | Immunocompromised state (HIV, transplant), cigarette smoking |
| Other | Multiparity, long-term oral contraceptive use, lower socioeconomic status, in utero DES exposure |
| Demographics | Hispanic and Black American women (higher incidence and mortality) |
The lifetime cumulative risk of HPV infection is 84% for those with at least one sexual partner; however, the majority of HPV infections are transient and do not lead to CIN.
6. Cervical Cancer Screening
Cytology — The Pap Smear
The Bethesda System classifies cervical cytology:
- NILM — Negative for Intraepithelial Lesion or Malignancy
- ASC-US — Atypical Squamous Cells of Undetermined Significance
- ASC-H — ASC, cannot exclude HSIL
- LSIL — Low-Grade Squamous Intraepithelial Lesion
- HSIL — High-Grade Squamous Intraepithelial Lesion
- AGC — Atypical Glandular Cells
HPV Testing
- hrHPV testing identifies 90% of CIN 2/3 lesions in women with ASC-US cytology
- Primary hrHPV testing (without concurrent cytology) is now the preferred screening modality in average-risk patients
- Women with ASC-H should be referred directly to colposcopy — hrHPV triage is not appropriate due to the inherent high risk of CIN 2/3
ASCCP Screening Recommendations
| Age Group | Recommendation |
|---|---|
| < 21 years | No screening (regardless of sexual history) |
| 21–29 years | Cytology alone every 3 years |
| 30–65 years | Co-testing (cytology + hrHPV) every 5 years (preferred); or cytology alone every 3 years |
| > 65 years | Discontinue if adequate negative screening history (3 negative cytologies or 2 negative co-tests in 10 years) |
— Berek & Novak's Gynecology, p. 835
7. Colposcopy
Colposcopy is the visual examination of the cervix with magnification, using acetic acid (3–5%) and Lugol's iodine (Schiller's test). It is indicated for:
- LSIL or HSIL cytology
- ASC-H
- Positive hrHPV on primary screening with abnormal cytology
- AGC cytology
Colposcopic Findings
After acetic acid application:
Acetic acid selectively whitens cells with high nuclear/protein content (dysplastic cells), while mature glycogenated squamous cells appear pink.
| Finding | Description | Significance |
|---|---|---|
| Acetowhite epithelium | Dense white discoloration after acetic acid | Hallmark of CIN; density correlates with grade |
| Punctation | Dilated capillaries seen end-on as red dots | High-grade CIN when in acetowhite area |
| Mosaic | Terminal capillaries surrounding polygonal blocks of acetowhite tissue | Associated with CIN 2/3 |
| Leukoplakia | White plaque visible before acetic acid | Keratin layer; must biopsy — may overlie CIN or carcinoma |
| Atypical vessels | Corkscrew, network, hairpin patterns | Raise suspicion for invasive cancer |
IFCPC criteria for CIN 3: Ridge sign (dense acetowhite along cervical ridges) and inner border sign (thick acetowhite with distinct inner border inside finer acetowhite lesion).
After Lugol's iodine: Healthy glycogenated epithelium stains mahogany brown. Dysplastic (glycogen-poor) cells are iodine-negative (yellow/mustard color) = Schiller-positive.
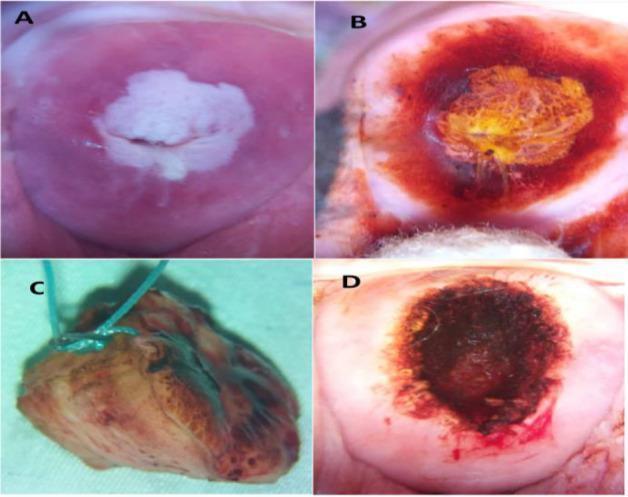
A minimum colposcopic examination includes: visualization of the SCJ, identification of acetowhitening or lesion(s), and an overall impression (normal/benign, low grade, high grade, cancer).
8. Management of CIN
CIN 1 (LSIL)
- Often represents productive HPV infection without obligate cancer precursor potential
- Most regress spontaneously — observation is preferred
- Surveillance with co-testing or hrHPV-based testing at 12 months
- Treatment only for persistent CIN 1 (≥2 years) or if patient is at high risk
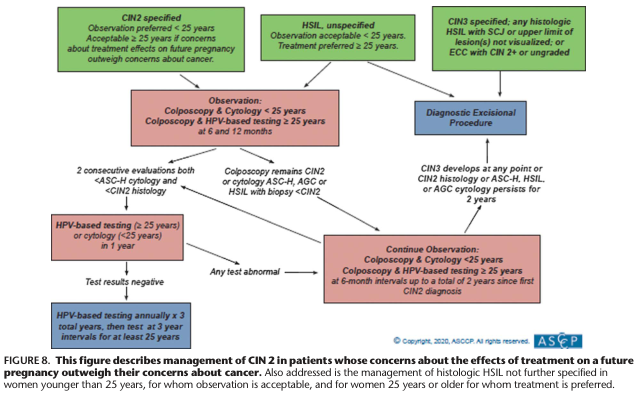
CIN 2 and CIN 3 (HSIL)
CIN 2 (with fertility concerns or age <25)
- Observation preferred for women <25 years
- Observation acceptable ≥25 years if concerns about treatment effects on future pregnancy outweigh cancer concerns
- Follow with colposcopy and cytology/HPV-based testing at 6 and 12 months
- Treat if CIN 3 develops, CIN 2 persists ≥2 years, or cytology remains ASC-H/HSIL/AGC
CIN 3 / Any HSIL with SCJ not visualized / ECC with CIN 2+
- Diagnostic Excisional Procedure is indicated
CIN 2 Management Flowchart (ASCCP):
Treatment Modalities
| Modality | Type | Indications | Notes |
|---|---|---|---|
| Cryotherapy | Ablative | CIN 1–2, lesion fully visible, no suspicion of invasion | Destroys tissue with liquid nitrogen; no specimen |
| Laser ablation | Ablative | CIN 1–2 | More uncomfortable than LEEP; longer operative time |
| LEEP / LLETZ | Excisional | CIN 2–3, preferred HSIL treatment | Fast (mean 16 sec), well-tolerated, provides specimen for margins; ~3.9% recurrence |
| Cold-knife conization (CKC) | Excisional | CIN 3, AIS, SCJ not visualized, prior inadequate colposcopy | Precise margins; preferred for AIS |
| Hysterectomy | Surgical | Recurrent/persistent CIN after excision; AIS; completed childbearing | Not first-line for CIN |
Positive cone margins significantly increase recurrence risk: 31.9% recurrence with positive margins vs. 4.9% with negative margins. — Berek & Novak's Gynecology, p. 867
Adenocarcinoma in situ (AIS) must always be treated with an excisional procedure (not ablation), as it may be multifocal and skip lesions can be missed.
9. Invasive Cervical Cancer
Epidemiology
- Third most common gynecologic cancer in the US
- Over 14,000 new cases and >4,000 deaths annually in the US
-
80% decrease in incidence since the 1950s due to screening
- Remains a leading cancer killer of women in developing countries (Central/South America, sub-Saharan Africa) due to lack of screening access
- Median age at presentation: 48 years
- HPV DNA detected in >99% of cervical cancers
Histologic Types
| Type | Frequency | HPV Association |
|---|---|---|
| Squamous cell carcinoma | ~70–80% | Strongly HPV-driven |
| Adenocarcinoma | ~15–25% | HPV 16, 18, 45 |
| Adenosquamous carcinoma | Rare | HPV |
| Small cell / neuroendocrine | Very rare | Aggressive behavior |
FIGO Staging of Cervical Cancer
| Stage | Description | Approximate 5-year Survival |
|---|---|---|
| I | Confined to cervix | ~85–90% |
| IA | Microscopic invasion ≤5 mm depth, ≤7 mm horizontal | ~95% |
| IB | Clinically visible ≤4 cm (IB1) or >4 cm (IB2) | ~70–80% |
| II | Beyond cervix; not to pelvic wall or lower ⅓ vagina | ~60–75% |
| IIA | Upper ⅔ vagina; no parametrial involvement | |
| IIB | Parametrial involvement | |
| III | Pelvic wall / lower ⅓ vagina / hydronephrosis / pelvic/paraaortic nodes | ~30–50% |
| IVA | Bladder or rectal mucosa invasion | ~15–20% |
| IVB | Distant metastases | <10% |
Clinical Presentation
- Early-stage: often asymptomatic — detected via screening
- Advanced disease:
- Postcoital or intermenstrual bleeding
- Foul-smelling vaginal discharge
- Pelvic pain
- Dyspareunia
- Bladder/bowel dysfunction (hematuria, tenesmus) in advanced disease
Spread is primarily by direct extension to uterine corpus, vagina, bladder, parametria, and pelvic sidewall; and via lymphatic spread to pelvic then paraaortic nodes; hematogenous spread is less common.
Diagnosis and Staging Workup
- Advanced disease: direct visualization + biopsy (Pap smear inadequate)
- Pelvic + rectovaginal examination
- Examination under anesthesia, cystoscopy, proctoscopy
- Imaging: MRI (preferred for local extent), CT (nodal/systemic), PET-CT (metastatic disease)
10. Treatment of Cervical Cancer
Treatment is guided by FIGO stage, patient age, fertility desire, and tumor size.
Early Stage (IA–IB1)
| Scenario | Treatment |
|---|---|
| Microscopic/microinvasive (IA1, no LVSI) | Cone biopsy or simple hysterectomy |
| Fertility desired (IB1) | Radical trachelectomy (removal of cervix only) + pelvic lymphadenectomy |
| Definitive surgical treatment (IB1–IIA) | Radical hysterectomy (open approach preferred over laparoscopic — better disease-free survival) + pelvic lymphadenectomy ± paraaortic lymphadenectomy |
Adjuvant Therapy Indications (Post-Radical Hysterectomy)
Postoperative chemoradiation is indicated for:
- Lymphovascular space invasion (LVSI)
- Deep cervical stromal involvement
- Larger-than-expected tumor size
- Positive surgical margins
- Parametrial involvement
- Nodal involvement
Advanced Stage (IIB–IVA)
Definitive concurrent chemoradiation therapy:
- External beam radiation + brachytherapy
- Radiosensitizing weekly cisplatin (concurrent chemotherapy)
Recurrent / Metastatic Disease
| Scenario | Treatment |
|---|---|
| Central pelvic recurrence | Pelvic exenteration (highly selected; curative in 25–50%) |
| Persistent/recurrent/metastatic | Platinum-taxane doublet chemotherapy |
| Advanced with VEGF expression | + Bevacizumab (anti-VEGF) — incremental survival benefit |
| PD-L1 expressing tumors | + Pembrolizumab (immune checkpoint inhibitor) ± bevacizumab |
— Goldman-Cecil Medicine, p. 2089–2090
11. HPV Vaccination — Prevention
Gardasil 9 covers HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58 (9 types):
- Approved for males and females ages 9–45 years
- Prevents anogenital warts, CIN, and cervical cancer
- Significantly reduces risk of CIN 2/3 and HPV-associated cancers
- Patients with prior HPV infection or history of cervical excision procedures should still receive the vaccine in accordance with guidelines — the vaccine protects against types not yet acquired
— Sabiston Textbook of Surgery
12. CIN in Special Populations
Pregnancy
- Approximately 2–7% of pregnancies complicated by abnormal Pap or HPV result
- Goal: rule out invasive cancer; defer treatment until postpartum
- Cervical dysplasia rarely progresses in immunocompetent pregnant women; regression is common
- Endocervical curettage (ECC) is contraindicated in pregnancy
- Colposcopy is safe (complication rate ~0.6%)
- Observation (no treatment) is appropriate for CIN 2–3 in pregnancy if invasion excluded
- Re-evaluate at least 6 weeks postpartum
— Creasy & Resnik's Maternal-Fetal Medicine, p. 1351
Immunocompromised (HIV+)
- Higher risk of persistent HPV, faster progression, multifocal disease
- More frequent screening and lower threshold for colposcopy
Summary: CIN → Cervical Cancer Continuum
Normal epithelium
↓ (HPV infection at transformation zone)
Productive HPV infection / Koilocytic change
↓
CIN 1 (LSIL) — lower ⅓ dysplasia
↓ ~15% progress; ~57% regress
CIN 2 (HSIL) — lower ⅔ dysplasia
↓ ~35% progress; ~43% regress
CIN 3 / Carcinoma in situ (HSIL) — full thickness dysplasia, BM intact
↓ if untreated over years
Microinvasive carcinoma (Stage IA)
↓
Invasive carcinoma (Stages IB–IV)
Key principle: HPV infection is necessary but not sufficient. Persistent oncogenic HPV + host susceptibility + cofactors (smoking, immunosuppression) over ~15 years → invasive cancer. Early detection via screening and treatment of CIN prevents the majority of cervical cancers.
Sources: Berek & Novak's Gynecology; Goldman-Cecil Medicine (International Ed.); Sabiston Textbook of Surgery; Creasy & Resnik's Maternal-Fetal Medicine
This is a shared conversation. Sign in to Orris to start your own chat.