Q. Mondini Dysplasia in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Searching Images
Mondini dysplasia cochlear malformation CT scan diagram
Reading File
Loading Image
Loading Image
Reading File
Loading Image
I now have comprehensive content from the Cummings textbook. Let me compile the full 50-mark answer.
MONDINI DYSPLASIA
A Comprehensive 50-Mark Answer (RGUHS Pattern)
1. INTRODUCTION & HISTORICAL BACKGROUND
Mondini dysplasia (also called Mondini malformation or Incomplete Partition Type II / IP-II) is one of the most important congenital inner ear anomalies encountered in clinical otolaryngology.
Historical Note: Carlo Mondini, an Italian anatomist, first described the malformation in 1791 when he performed an autopsy on a deaf boy and noted an abnormal cochlea. He found a cochlea with only 1.5 turns instead of the normal 2.5–2.75 turns, with a deficiency in the apical interscalar septum and a dilated vestibule.
2. DEFINITION
Mondini dysplasia is a congenital malformation of the inner ear characterized by:
- A cochlea with only 1.5 turns (normal: 2.5–2.75 turns)
- Absent or deficient interscalar septum (osseous spiral lamina) in the apical portion
- Scala communis (confluence of scala vestibuli and scala tympani at the apex)
- Often associated with dilated vestibule and enlarged vestibular aqueduct (EVA)
"Histologically, incomplete partition appears to be the radiographic correlate of classical Mondini dysplasia." — Cummings Otolaryngology Head and Neck Surgery
3. EMBRYOLOGY & PATHOGENESIS
Normal Cochlear Development
The inner ear develops from the otic vesicle (otocyst) between the 4th and 8th weeks of gestation.
Otic placode (3rd week)
↓
Otic vesicle / Otocyst (4th week)
↓
Cochlear duct begins to coil (6th week)
↓
1.5 turns completed (7th week)
↓
2.5 turns (full complement) by 8th–11th week
↓
Adult size inner ear at birth
Pathogenesis of Mondini Dysplasia
Development is arrested at the 7th week of gestation — at the point when the cochlea has completed only 1.5 turns. Subsequent modiolus and interscalar septum formation fails to occur.
EMBRYOLOGICAL ARREST AT 7TH WEEK
↓
Cochlea halts at 1.5 turns
↓
Absent apical interscalar septum
↓
Scala communis formation at apex
↓
Deficient modiolus (partial/absent)
↓
Associated vestibular dilatation
↓
Enlarged vestibular aqueduct (EVA)
"A majority of combined osseous and membranous malformations appear to arise from a premature arrest in the development of one or more components of the inner ear... the earlier the developmental arrest, the more severe the deformity and the worse the hearing." — Cummings Otolaryngology Head and Neck Surgery
4. INCIDENCE / EPIDEMIOLOGY
| Cochlear Malformation | Incidence |
|---|---|
| Incomplete Partition (Mondini dysplasia) | 55% |
| Common cavity | 26% |
| Cochlear hypoplasia | 15% |
| Complete aplasia (Michel) | <5% |
- Most common cochlear malformation (>50% of all cochlear deformities)
- Can be unilateral or bilateral
- May occur as an isolated anomaly or as part of a syndrome
- Accounts for a significant proportion of cases of congenital sensorineural hearing loss (SNHL)
5. CLASSIFICATION OF COCHLEAR MALFORMATIONS (Sennaroglu & Saatci, 2002)
Sennaroglu and Saatci classified cochlear malformations in order of increasing development:
CLASSIFICATION (Sennaroglu, 2002)
│
├── Michel Aplasia — Complete absence of cochlea + labyrinth (3rd week arrest)
│
├── Cochlear Aplasia — No cochlea, some vestibular structures present
│
├── Common Cavity — Cochlea + vestibule = single cystic cavity (4th–5th week)
│
├── Cochlear Hypoplasia — Small cochlea, <1 turn (6th week arrest)
│
└── Incomplete Partition (IP)
├── IP-I — No modiolus, no interscalar septa, cystic cochlea
├── IP-II (MONDINI) — 1.5 turns, absent apical septum + EVA ← CLASSIC
└── IP-III — X-linked, absent modiolus, dilated IAC
IP-II (Classic Mondini) is characterized by:
- Preserved basal turn (normal)
- Fused/cystic middle + apical turns
- Deficient apical modiolus and interscalar septum
- Enlarged vestibular aqueduct (EVA) — hallmark association
6. GENETICS & ASSOCIATED SYNDROMES
Mondini dysplasia can be:
Isolated (Non-Syndromic)
- Sporadic in most cases
- Some familial cases with autosomal dominant inheritance
- Associated with mutations in SLC26A4 (Pendrin gene) — especially with EVA
- PDS gene mutations → Pendred syndrome + Mondini
Syndromic Associations
| Syndrome | Features |
|---|---|
| Pendred Syndrome | EVA + Mondini + thyroid goiter + euthyroid state; AR; SLC26A4 mutation |
| Waardenburg Syndrome | SNHL + heterochromia iridis + white forelock |
| BOR Syndrome (Branchio-Oto-Renal) | Branchial anomalies + preauricular pits + renal defects |
| DiGeorge Syndrome | 22q11 deletion |
| CHARGE Syndrome | Coloboma, Heart defect, Choanal Atresia, Retardation, Genital, Ear anomalies |
| Treacher Collins Syndrome | Mandibulofacial dysostosis |
| Trisomy 13, 18 | Chromosomal |
7. ANATOMY OF THE MALFORMATION
Normal vs. Mondini Cochlea
NORMAL COCHLEA MONDINI COCHLEA
────────────── ───────────────
2.5–2.75 turns 1.5 turns
Intact modiolus Deficient/absent apical modiolus
Interscalar septa present Absent apical interscalar septum
Separate scalae at apex Scala communis (apical confluence)
Normal vestibule Dilated vestibule
Normal vestibular aqueduct Enlarged vestibular aqueduct (EVA)
Normal endolymphatic sac Dilated endolymphatic sac
Normal IAC May have widened IAC
Histopathology
- Cochlea shows 1.5 turns
- Basal turn is usually well formed
- Middle and apical turns show fusion/confluence
- Absent osseous spiral lamina in the apex
- Scala communis (apical) — the scalae vestibuli and tympani communicate freely
- Endolymphatic hydrops may be present (explains fluctuating hearing)
- Stria vascularis may be abnormal
8. CLINICAL FEATURES
Presentation
- Congenital sensorineural hearing loss — most common presentation
- Ranges from mild to profound
- May be unilateral or bilateral
- Often progressive and fluctuating (due to endolymphatic hydrops)
- Vestibular symptoms — present in ~20% of cases
- Vertigo, imbalance
- Tullio phenomenon (vertigo on loud sound — "third window" effect)
- Delayed motor development — particularly when SCCs are also malformed
- Recurrent bacterial meningitis — important and potentially life-threatening complication
The Classic Triad of Mondini Dysplasia
┌─────────────────────────────────────────┐
│ 1. Sensorineural Hearing Loss (SNHL) │
│ 2. Enlarged Vestibular Aqueduct (EVA) │
│ 3. Incomplete Cochlear Partition │
└─────────────────────────────────────────┘
CSF Perilymph Fistula & Meningitis
- Communication between subarachnoid space and middle ear through:
- Deficient oval window footplate (most common) — stapes gusher
- Round window
- Promontory fistula
- Causative organisms for meningitis:
- Streptococcus pneumoniae — 71%
- Haemophilus influenzae — 33%
- β-hemolytic streptococci — 8%
- Polymicrobial infection noted
"All children with recurrent meningitis without an obvious cause should undergo CT of the inner ear to exclude inner ear malformation." — Cummings Otolaryngology
9. IMAGING DIAGNOSIS
A. High-Resolution CT (HRCT) Temporal Bone — GOLD STANDARD
Findings:
- Small cochlea — 5–6 mm vertically (normal: 8–10 mm)
- 1.5 cochlear turns (difficult to count; not primary criterion)
- Absent interscalar septum — especially at apex
- Cystic apical turn — middle + apical turns appear fused
- Dilated vestibule
- Enlarged vestibular aqueduct (EVA) — >1.5 mm at midpoint (Cincinnati criteria)
"The radiographic diagnosis depends more on cochlear size and the absence of a scalar septum than on the number of cochlear turns perceived." — Cummings Otolaryngology
B. MRI (Gadolinium-enhanced)
- Better for membranous labyrinth assessment
- Shows dilated endolymphatic sac and duct
- Rules out cochlear nerve aplasia/hypoplasia
- Essential for pre-cochlear implant workup
- T2-weighted sequences (CISS/FIESTA) provide excellent inner ear detail
CT vs. MRI Summary
| Feature | HRCT | MRI |
|---|---|---|
| Bony labyrinth | Excellent | Poor |
| Modiolus deficiency | Good | Moderate |
| Endolymphatic sac | Poor | Excellent |
| Cochlear nerve | Poor | Excellent |
| Radiation | Yes | No |
10. IMAGING ILLUSTRATIONS
Figure 1: IP-II (Mondini Deformity) — Axial CT showing cystic cochlear apex (arrow A) and dilated vestibule (arrow B)
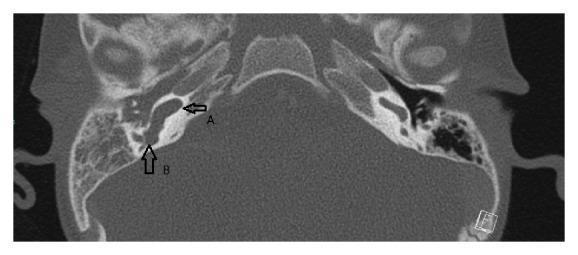
Axial CT temporal bone: Arrow A = absent interscalar septum with cystic apical cochlea; Arrow B = significantly dilated vestibule. Classic Mondini dysplasia (IP-II). Source: PMC Clinical VQA
Figure 2: IP-II (Mondini) — HRCT showing preserved basal turn, cystic middle/apical confluence (arrowhead) and enlarged vestibular aqueduct (arrow)
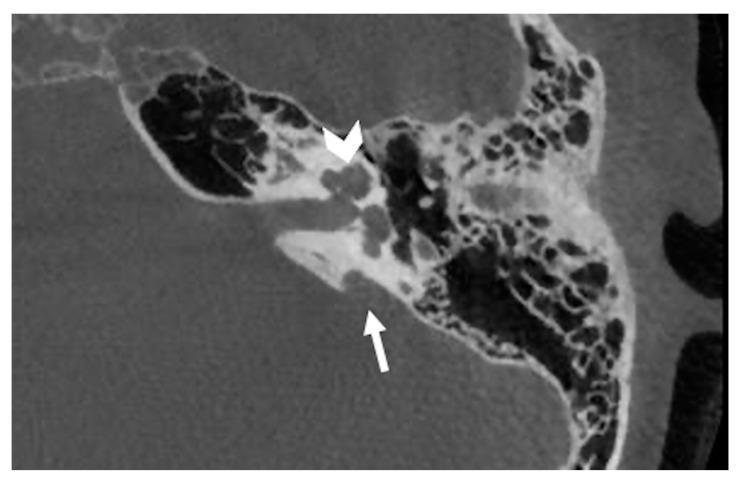
HRCT left temporal bone: Arrowhead = cystic apex (absent apical interscalar septum); Arrow = enlarged vestibular aqueduct. Characteristic IP-II (Mondini deformity). Source: PMC Clinical VQA
11. DIFFERENTIAL DIAGNOSIS
Differential Diagnosis of Cochlear Malformation
│
├── IP-I — Entire cochlea cystic (no modiolus at all); no EVA
├── IP-III — X-linked, full turns but no modiolus, dilated IAC
├── Common Cavity — Cochlea + vestibule = single cyst; no coiling
├── Cochlear Hypoplasia — Tiny cochlear bud, <1 turn
├── Large Vestibular Aqueduct Syndrome (LVAS) — EVA isolated; cochlea normal
└── Michel Aplasia — Complete absence of all inner ear structures
12. AUDIOLOGICAL EVALUATION
AUDIOLOGICAL WORKUP
│
├── Pure Tone Audiometry (PTA)
│ └── SNHL — typically flat or sloping, mild to profound
│
├── Speech Audiometry
│ └── Poor speech discrimination scores
│
├── DPOAE / TEOAE (Otoacoustic Emissions)
│ └── Absent (outer hair cell dysfunction)
│
├── ABR (Auditory Brainstem Response)
│ └── Elevated thresholds / absent waveforms
│
├── Electrocochleography (EcochG)
│ └── Enlarged SP/AP ratio → suggests endolymphatic hydrops
│
└── ASSR (Auditory Steady State Response)
└── For objective thresholds in infants
13. MANAGEMENT
A. Medical Management
- Diuretics (acetazolamide, furosemide) — for endolymphatic hydrops component
- Salt restriction — if hydrops suspected
- Avoidance of head trauma — risk of perilymph fistula
- Pneumococcal vaccination — essential to prevent meningitis
- Haemophilus influenzae type b (Hib) vaccine — mandatory
- Prompt treatment of otitis media — to prevent ascending meningitis
B. Surgical Management
1. Hearing Rehabilitation — Cochlear Implantation
This is the definitive treatment for severe-to-profound SNHL in Mondini dysplasia.
Pre-implant considerations:
- HRCT + MRI to assess:
- Cochlear nerve integrity (must be present)
- Modiolus deficiency (affects electrode stability)
- Risk of CSF gusher
- Facial nerve course anomalies
Operative challenges:
- Perilymph/CSF gusher — occurs due to patent modiolar defect communicating with IAC
- Management: Tight packing around electrode with muscle/fascia; lumbar drain if severe
- Electrode malpositioning — tip may migrate out of cochlea into IAC (malpositioned electrode)
- Facial nerve anomaly — increased incidence
"The anatomy of the cochlea in a mildly abnormal cochlea (Mondini malformation) is often similar to that of a normal cochlea. More severe dysplasias pose additional risk of facial nerve anomalies and inadequate intra-labyrinthine space." — Cummings Otolaryngology
Outcomes: Good outcomes reported in literature; speech perception may be slightly inferior to normal cochlea but significant benefit achieved.
2. CSF Fistula Repair
- Indicated in recurrent meningitis from CSF-middle ear fistula
- Approach: Oval window packing with fat graft + temporalis fascia
- Lumbar drain may be used adjunctively
- Endoscopic approaches increasingly utilized
3. For Associated Conditions
- EVA-related progressive hearing loss: Cochlear implant
- Associated vestibular dysfunction: Vestibular rehabilitation
14. FLOWCHART: DIAGNOSTIC APPROACH TO SUSPECTED MONDINI DYSPLASIA
CHILD WITH CONGENITAL / PROGRESSIVE SNHL
│
▼
History + Physical Examination
(Family history, syndromic features)
│
▼
Audiological Evaluation
(PTA, OAE, ABR, ASSR)
│
▼
SNHL Confirmed?
/ \
NO YES
│ │
Investigate HRCT Temporal Bone
other causes (High-resolution CT)
│
▼
Cochlear Malformation Found?
/ \
NO YES — Classify malformation
│ │
Consider Mondini / IP-II features?
other SNHL / \
causes NO YES
│ │
Other MRI (T2 CISS/FIESTA)
malformation │
┌────┴────┐
│ │
EVA present Cochlear nerve
(SLC26A4 assessment
testing) │
┌────┴────┐
Nerve Nerve
PRESENT ABSENT
│ │
Cochlear Brainstem
Implant Implant (ABI)
Candidate Consider
15. FLOWCHART: MANAGEMENT OF MONDINI DYSPLASIA
CONFIRMED MONDINI DYSPLASIA
│
┌─────┴──────┐
│ │
HEARING LOSS COMPLICATIONS
│ │
▼ ┌────┴────────────────┐
Severity? │ │
/ | \ Recurrent CSF Fistula
Mild Mod Sev Meningitis │
│ │ │ │ Oval window
│ │ │ Vaccinate packing + repair
│ │ │ + treat AOM (fat + fascia graft)
│ │ │ │
│ HA CI Identify
HA │ fistula site
│ (HRCT + intrathecal
▼ fluorescein/metrizamide)
Pre-implant workup
(HRCT + MRI)
│
┌────┴────┐
Anomaly Normal
IAC/CN anatomy
│ │
Counsel Standard
re gusher cochlear
risk implant
│
Intraop gusher management:
- Fascia packing
- Lumbar drain
- Proceed with implantation
16. COMPLICATIONS
| Complication | Mechanism | Management |
|---|---|---|
| Recurrent bacterial meningitis | CSF-middle ear fistula via oval window defect | Fistula repair, vaccination |
| Progressive SNHL | Endolymphatic hydrops, EVA | Cochlear implant |
| Perilymph/CSF gusher (intraoperative) | Patent modiolar-IAC communication | Fascial packing |
| Electrode malpositioning | Absent modiolus → tip migrates into IAC | Careful surgical technique, imaging |
| Vestibular dysfunction | Associated vestibule dilatation | Rehabilitation |
| Facial nerve injury | Anomalous facial nerve course | Pre-op HRCT assessment |
17. RECENT ADVANCES
1. Molecular Genetics
- SLC26A4 (Pendrin) gene mutations — most commonly associated with Mondini + EVA complex
- Next-generation sequencing (NGS) panels now available for comprehensive genetic workup of SNHL
- GJB2/GJB6 (connexin) mutations — rarely associated with inner ear structural anomalies
2. Refined Classification — Sennaroglu (2010, updated)
- IP-I, IP-II, IP-III classification allows targeted surgical planning
- IP-II (Mondini) = preserved basal turn + cystic apex + EVA
3. Advanced Imaging
- 3D-CISS MRI — reconstructed cochleograms for precise turn-counting and cochlear nerve caliber assessment
- Diffusion-weighted MRI — discriminating perilymph/endolymph abnormalities
- Inner ear volumetry — precise measurement of cochlear volume for electrode length selection
4. Cochlear Implantation Advances
- Slim modiolar electrodes (e.g., Cochlear Contour Advance, MED-EL Flex) — better adaptation to malformed cochleae
- Pre-curved perimodiolar electrodes — improved modiolar contact even with deficient septum
- Image-guided cochlear implantation — real-time intraoperative CT to confirm electrode position
- Intraoperative ABR/NRT — neural response telemetry to confirm adequate insertion
- Robotic cochlear implant surgery — precision drilling and electrode insertion
5. EVA Management
- Recognition that EVA + Mondini carries greater fluctuation risk → early implantation recommended
- Head trauma avoidance guidelines refined
- Genetic counseling for SLC26A4-positive families
6. Endoscopic Approaches
- Endoscopic-assisted oval window fistula repair
- Reduced morbidity compared to traditional approaches
18. PEARLS FOR EXAMINATION (RGUHS 50-Mark Answer)
| Point | Key Fact |
|---|---|
| Commonest cochlear malformation | Incomplete partition (Mondini) — 55% |
| Developmental arrest | 7th week of gestation |
| Classical triad | 1.5-turn cochlea + EVA + dilated vestibule |
| Imaging modality | HRCT temporal bone (gold standard) |
| Cochlear size (Mondini) | 5–6 mm (normal: 8–10 mm) |
| Most feared complication | Recurrent bacterial meningitis |
| Commonest organism | Streptococcus pneumoniae (71%) |
| Intraoperative complication (CI) | Perilymph/CSF gusher |
| Genetic association | SLC26A4 gene (Pendrin) |
| Syndrome most associated | Pendred syndrome |
| Definitive hearing rehab | Cochlear implantation |
| Histological finding | Scala communis at apex |
SOURCES & TEXTBOOK REFERENCES
- Cummings Otolaryngology Head and Neck Surgery (6th/7th Ed.) — Chapter 194: Congenital Malformations of the Inner Ear; Jackler, Luxford, House classification; Sennaroglu & Saatci classification
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery — Inner ear malformations; congenital SNHL
- Stell & Maran's Head and Neck Surgery — Cochlear malformations and cochlear implantation
- Dhingra's Diseases of Ear, Nose and Throat — Congenital SNHL; cochlear anomalies
- Hazarika's Textbook of ENT — Congenital ear disorders
- Zakir Hussain's Manual of ENT — SNHL and inner ear malformations
- Sennaroglu L, Saatci I (2002): A new classification for cochleovestibular malformations. Laryngoscope 112:2230
- Jackler RK, Luxford WM, House WF (1987): Congenital malformations of the inner ear: a classification based on embryogenesis. Laryngoscope 97(Suppl 40):2
- O'Donoghue G et al: Cochlear implantation in congenital Mondini dysplasia. Am J Med Genet A 143:13, 2013
Note on image generation: Detailed schematic diagrams (embryological timeline, comparative cochlear cross-sections) cannot be auto-generated in this session as the image generation quota has been reached. Please contact support or upgrade your plan for additional image generation. The CT scan images above from the medical literature directly illustrate the key radiological findings of Mondini dysplasia.
This is a shared conversation. Sign in to Orris to start your own chat.