What is bio film and its role in development of bacterial resistance to antibiotics in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Searching Images
biofilm formation stages bacterial structure diagram
Finding Sources
Reading File
Reading File
Reading File
Reading File
Now I have sufficient material to compose a comprehensive 50-mark RGUHS-style answer. Here it is:
BIOFILM AND ITS ROLE IN BACTERIAL RESISTANCE TO ANTIBIOTICS
OUTLINE (for 50 marks)
- Definition and Introduction
- Historical Background
- Composition of Biofilm
- Stages of Biofilm Formation (with diagram)
- Quorum Sensing
- Mechanisms of Antibiotic Resistance in Biofilm (with flowchart)
- ENT Relevance — Chronic Rhinosinusitis, Otitis Media, Tonsils
- Organisms Involved
- Clinical Implications
- Management Strategies
- Recent Advances
- Summary/Conclusion
1. DEFINITION
A biofilm is an aggregate of interactive bacteria attached to a solid (biotic or abiotic) surface or to each other, enclosed within a self-produced matrix of extracellular polymeric substance (EPS). This mode of growth is fundamentally different from free-living planktonic bacteria and confers extraordinary resistance to host immune defenses and antimicrobial agents.
"A biofilm is an aggregate of bacteria tolerant to treatment and the host defense." — Bjarnsholt et al., cited in Schwartz's Principles of Surgery, 11th Ed.
"Bacteria in the exopolysaccharide matrix may be protected from the host's immune mechanisms. Some of the bacteria within the biofilm show marked resistance to antimicrobials compared with the same strain grown free-living in broth." — Jawetz, Melnick & Adelberg's Medical Microbiology, 28th Ed.
2. HISTORICAL BACKGROUND
- Van Leeuwenhoek (1680s) first described microbial aggregates on tooth surfaces.
- The term "biofilm" was coined by J.W. Costerton in 1978.
- Donlan & Costerton (2002) — landmark review of biofilm in healthcare settings.
- Stoodley et al. (2002) — described biofilm as the predominant mode of microbial life.
- In ENT, Hall-Stoodley et al. (2006) demonstrated biofilms in chronic otitis media with effusion using FISH (fluorescence in situ hybridization).
3. COMPOSITION OF BIOFILM
The EPS matrix consists of:
| Component | Source | Function |
|---|---|---|
| Exopolysaccharides (glycocalyx) | Bacteria | Physical scaffold; diffusion barrier |
| Proteins | Bacteria + host | Structural, enzymatic, adhesive |
| Extracellular DNA (eDNA) | Dying cells | Structural integrity; horizontal gene transfer |
| Lipids | Bacterial membrane | Hydrophobic barrier |
| Water channels | Matrix | Nutrient and oxygen distribution |
"The EPS is composed of secreted proteins, glycans, lipids and the extravasated DNA of dying cells. This supports the microbial community and shields it from antimicrobial defenses, including antibodies, complement, and phagocytic cells." — Janeway's Immunobiology, 10th Ed.
4. STAGES OF BIOFILM FORMATION
DIAGRAM — Five Stages of Biofilm Formation
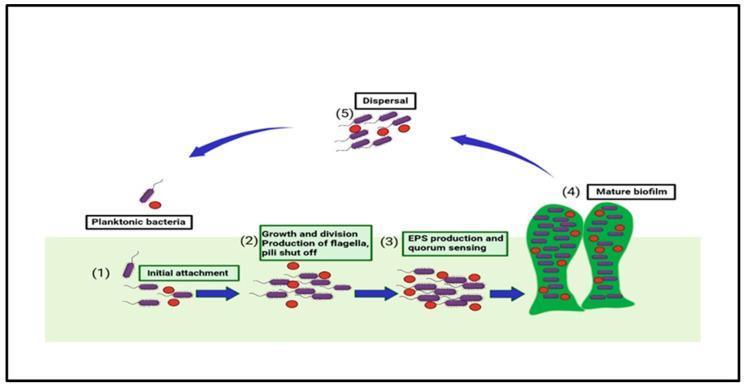
(Stages of biofilm formation: from initial attachment to mature biofilm with dispersal of planktonic cells)
FLOWCHART — Biofilm Life Cycle
PLANKTONIC BACTERIA (Free-floating)
│
▼ [Conditioning film on surface — host proteins, fibronectin]
STAGE 1: REVERSIBLE ATTACHMENT
(Bacteria adhere via flagella/pili/surface adhesins)
│
▼ [Permanent anchor via surface proteins — MSCRAMM in S. aureus]
STAGE 2: IRREVERSIBLE ATTACHMENT / MICROCOLONY FORMATION
(Inhibition of motility; initial EPS secretion begins)
│
▼ [Quorum sensing activated → gene expression shifts]
STAGE 3: MATURATION STAGE I — EPS PRODUCTION
(Extracellular polymeric substance forms protective matrix)
│
▼ [3D mushroom-like structures; water channels form]
STAGE 4: MATURE BIOFILM
(Highly resistant, structured community; 800× more resistant to antibiotics)
│
▼ [Enzymatic degradation of matrix via proteases/nucleases]
STAGE 5: DISPERSAL
(Bacteria shed as planktonic cells → seed new sites → cycle repeats)
Time course:
- Attachment: within minutes
- Microcolony: 2–4 hours
- EPS production + antibiotic resistance: 6–12 hours
- Mature biofilm: 2–4 days
- Recovery after debridement: within 24 hours
(Schwartz's Principles of Surgery, 11th Ed.)
5. QUORUM SENSING (QS)
Quorum sensing is the bacterial cell-to-cell communication system that drives biofilm formation.
Mechanism:
Bacteria produce small chemical signals → AUTOINDUCERS (AIs)
│
▼
AIs accumulate as bacterial density increases
│
▼
Threshold concentration of AIs reached → Binds receptor proteins
│
▼
Gene expression altered in coordinated fashion
│
▼
Biofilm-specific virulence genes activated:
• EPS biosynthesis genes
• Antibiotic efflux pumps
• Toxin production
• Motility suppression
- Gram-negative bacteria: Use N-acyl homoserine lactones (AHLs) as AIs (e.g., Pseudomonas aeruginosa — las/rhl systems)
- Gram-positive bacteria: Use oligopeptide autoinducers (e.g., S. aureus — agr system)
- Universal signal: Autoinducer-2 (AI-2) — cross-species communication
"Quorum sensing, where chemical signal molecules called autoinducers are produced depending on cell density and physiologic conditions, leading to the expression of specific genes in a coordinated fashion." — Rockwood & Green's Fractures in Adults, 10th Ed., 2025
6. MECHANISMS OF ANTIBIOTIC RESISTANCE IN BIOFILM
"Biofilm organisms are up to 800-fold less susceptible to antibiotics than planktonic organisms." — Rockwood & Green's Fractures in Adults, 10th Ed.
FLOWCHART — Mechanisms of Antibiotic Resistance
BIOFILM → ANTIBIOTIC RESISTANCE via FIVE MECHANISMS:
┌─────────────────────────────────────────────────────┐
│ MECHANISM 1: PHYSICAL BARRIER (EPS Matrix) │
│ • EPS acts as diffusion barrier │
│ • Charged antibiotics bind to EPS components │
│ • Hydrophobic drugs excluded by glycan layers │
│ → Antibiotic never reaches bacteria │
└─────────────────────────────────────────────────────┘
│
┌─────────────────────────────────────────────────────┐
│ MECHANISM 2: ALTERED MICROENVIRONMENT │
│ • Oxygen gradients within biofilm │
│ • Anaerobic zones → aminoglycosides ineffective │
│ • Low pH in deeper layers → β-lactam inactivation │
│ • Nutrient depletion → metabolic slow-down │
└─────────────────────────────────────────────────────┘
│
┌─────────────────────────────────────────────────────┐
│ MECHANISM 3: PERSISTER CELLS │
│ • Subpopulation of metabolically dormant cells │
│ • Antibiotic-tolerant (not resistant genetically) │
│ • Antibiotics target dividing cells only │
│ • Persisters survive → repopulate after treatment │
└─────────────────────────────────────────────────────┘
│
┌─────────────────────────────────────────────────────┐
│ MECHANISM 4: HORIZONTAL GENE TRANSFER (HGT) │
│ • eDNA in matrix facilitates conjugation │
│ • Resistance genes spread within biofilm │
│ • Plasmids carrying resistance genes transferred │
│ • Phenotypic AND genotypic plurality │
└─────────────────────────────────────────────────────┘
│
┌─────────────────────────────────────────────────────┐
│ MECHANISM 5: IMMUNE EVASION │
│ • EPS shields bacteria from opsonization │
│ • Antibodies cannot penetrate matrix │
│ • Complement activation blocked │
│ • Phagocytes "frustrated" → release ROS/proteases │
│ → Tissue damage WITHOUT bacterial killing │
└─────────────────────────────────────────────────────┘
"The resistance to antimicrobial agents includes altered composition of extracellular polymeric substances, phenotypic and genotypic plurality — the latter allows virulence and bacterial resistance to drugs. The genetic plurality is passed horizontally among bacteria in the wound." — Schwartz's Principles of Surgery, 11th Ed.
Summary Table — Mechanisms
| Mechanism | Mode | Antibiotics Affected |
|---|---|---|
| EPS diffusion barrier | Physical | Aminoglycosides, β-lactams |
| Low metabolic rate (persisters) | Physiological | All bactericidal agents |
| Low oxygen tension | Microenvironmental | Aminoglycosides |
| Low pH | Microenvironmental | β-lactams, macrolides |
| Horizontal gene transfer | Genetic | All classes |
| Efflux pumps (QS-regulated) | Enzymatic | Fluoroquinolones, tetracycline |
| Immune evasion | Immunological | — (indirect) |
7. ENT RELEVANCE — BIOFILM IN HEAD AND NECK
A. Chronic Rhinosinusitis (CRS)
From Cummings Otolaryngology – Head and Neck Surgery:
"The presence of biofilms on the sinus mucosa of CRS patients has been suggested as a mechanism for antibiotic resistance and the recurrence of inflammation after treatment. The biofilm hypothesis can be considered an offshoot of the staphylococcal superantigen hypothesis, as this is the organism most commonly identified in the biofilms of resistant CRS." — Cummings Otolaryngology, 7th Ed.
- Biofilms found on ethmoid mucosa in >50% of CRS patients undergoing FESS
- Organisms: S. aureus, S. epidermidis, H. influenzae, P. aeruginosa
- FISH and confocal laser scanning microscopy (CLSM) — gold standard for detection
- Explains why CRS recurs despite adequate antibiotic therapy
B. Chronic Otitis Media (COM)
- Hall-Stoodley (2006): Biofilms demonstrated on middle ear mucosa in children with otitis media with effusion (OME)
- H. influenzae, S. pneumoniae, M. catarrhalis — commonest biofilm-forming organisms in otitis
- Biofilm on tympanostomy tubes — contributes to tube blockage and persistent discharge
- Explains persistence of OME despite multiple antibiotic courses
C. Tonsils and Adenoids
- Biofilms found on tonsillar crypts — recurrent tonsillitis organisms (Streptococcus, Bacteroides)
- Adenoid biofilms serve as a reservoir for sinonasal pathogens
- Dhingra emphasizes the role of tonsillar biofilm in recurrent tonsillitis and antibiotic failure
D. Dental Plaque (Oral Biofilm)
From Sherris & Ryan's Medical Microbiology, 8th Ed.:
"Dental plaque is the most prevalent and densest of human biofilms. It forms in stages and layers. The biofilm fastens nutrient and growth regulatory relationships between its members and provides a shield from the outside. There are thought to be 300–400 bacterial species present in mature dental plaque."
8. ORGANISMS COMMONLY FORMING BIOFILM IN ENT
| Organism | Site | Significance |
|---|---|---|
| S. aureus (MRSA) | Sinuses, wounds, implants | Most virulent; superantigen production |
| S. epidermidis | Implants, tympanostomy tubes | Slime-producing; commonest device biofilm |
| Pseudomonas aeruginosa | Sinuses, cystic fibrosis, ear | Las/Rhl QS system; alginate matrix |
| H. influenzae | Middle ear, sinuses | Non-typeable strains; OME biofilm |
| S. pneumoniae | Middle ear | Capsular polysaccharide biofilm |
| M. catarrhalis | Middle ear | Outer membrane vesicle biofilm |
| Streptococcus mutans | Dental | Classic oral biofilm |
| Propionibacterium acnes | Implants | Chronic low-grade infection |
9. CLINICAL IMPLICATIONS
Biofilm Diagram — Immune Evasion
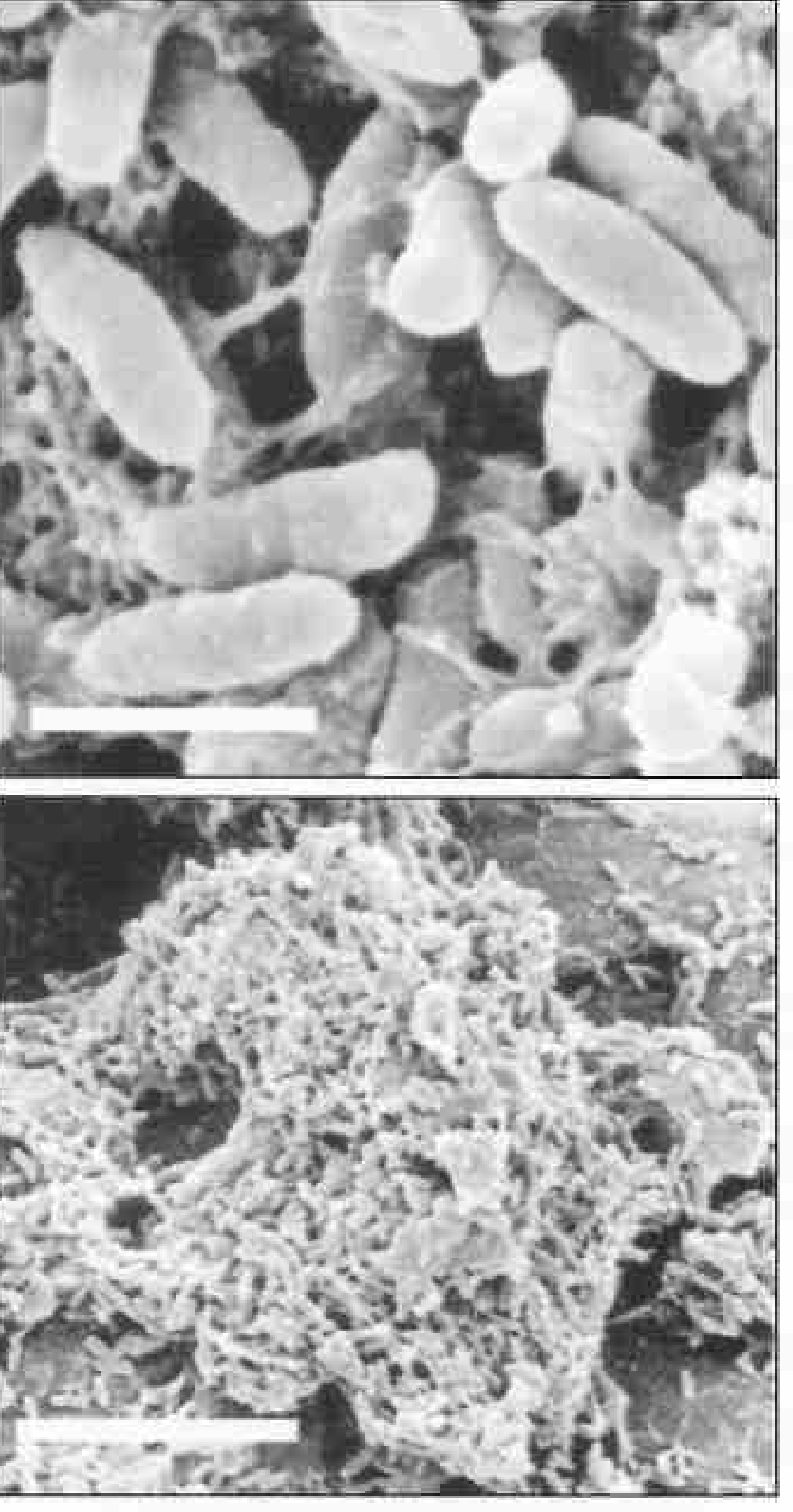
(SEM of P. aeruginosa biofilm showing EPS strands connecting bacteria — Janeway's Immunobiology, 10th Ed.)
- Biofilms explain treatment failure in chronic infections
- In cystic fibrosis: P. aeruginosa biofilm in airways is the principal cause of morbidity and mortality — Jawetz Medical Microbiology, 28th Ed.
- Prosthetic joint and orthopedic implant infections: Up to 65% caused by biofilm-producing organisms — Rockwood & Green's, 10th Ed.
- Catheter-associated infections (CAUTI, CLABSI): S. epidermidis and S. aureus biofilm
- In chronic wounds (diabetic, venous): >60% harbor biofilm — Schwartz's Surgery, 11th Ed.
10. MANAGEMENT OF BIOFILM INFECTIONS
FLOWCHART — Treatment Approach
SUSPECTED BIOFILM INFECTION
│
▼
DIAGNOSIS:
• CLSM (Confocal Laser Scanning Microscopy)
• FISH (Fluorescence In-Situ Hybridization)
• SEM (Scanning Electron Microscopy)
• Synovial fluid culture with sonication
│
▼
STEP 1: PHYSICAL DISRUPTION
• Surgical debridement (most effective)
• Debridement weekly in wounds
• FESS in CRS — removes biofilm-laden mucosa
• Implant removal where feasible
│
▼
STEP 2: ANTIMICROBIAL THERAPY
(Post-disruption — while biofilm disrupted)
• Rifampicin — penetrates biofilm matrix
• Fluoroquinolones — moderate biofilm penetration
• Fosfomycin — inhibits EPS synthesis
• Topical silver/iodine/honey — wound biofilms
• Prolonged IV antibiotics for implant infections
│
▼
STEP 3: PREVENTION OF RECURRENCE
• Anti-biofilm coated implants (silver, rifampicin-coated)
• Nasal irrigation (saline) in CRS
• Probiotics (competitive exclusion)
• Anti-quorum sensing agents (experimental)
11. RECENT ADVANCES (2020–2025)
Relevant for RGUHS long answers — demonstrates updated knowledge:
-
Anti-Quorum Sensing (Anti-QS) Therapy: Quorum quenching enzymes (e.g., lactonases that degrade AHLs); halogenated furanones — block QS signaling to prevent biofilm formation without selective pressure for resistance.
-
Bacteriophage Therapy: Phages can penetrate and dissolve EPS matrix via phage-encoded depolymerases; phage cocktails active against MRSA and P. aeruginosa biofilms in clinical trials (2022–2024).
-
Dispersin B (DspB): Enzyme that degrades poly-N-acetylglucosamine (PNAG) in staphylococcal and H. influenzae biofilms — ongoing trials.
-
Nano-particle-based Drug Delivery: Antibiotic-loaded nanoparticles (silver, chitosan, PLGA) penetrate EPS — overcome diffusion barrier limitation.
-
Photodynamic Inactivation (PDI): Light-activated photosensitizers generate ROS that kill biofilm bacteria — studied in chronic sinusitis and wound biofilms.
-
DNase (Dornase alfa): Degrades eDNA in EPS matrix — used in CF lungs; being studied for sinus and wound biofilms.
-
Biofilm-specific diagnostics: 16S rRNA metagenomic sequencing replacing conventional cultures for detecting multispecies biofilm communities in CRS and OME.
-
MBEC (Minimum Biofilm Eradication Concentration) assay — recognized as superior to MIC for guiding antibiotic dosing in biofilm infections.
12. COMPARISON: PLANKTONIC vs BIOFILM BACTERIA
| Feature | Planktonic | Biofilm |
|---|---|---|
| Growth mode | Free-floating | Surface-attached |
| Antibiotic susceptibility | Standard MIC | 100–1000× MIC needed |
| Host immune clearance | Susceptible | Protected by EPS |
| Genetic exchange | Low | High (HGT) |
| Metabolic activity | High | Variable (persisters dormant) |
| Virulence gene expression | Standard | Quorum sensing-enhanced |
| Clinical relevance | Acute infections | Chronic/recurrent infections |
SUMMARY DIAGRAM — Biofilm & Antibiotic Resistance (Master Flowchart)
BACTERIA ENCOUNTER SURFACE
(sinus mucosa / implant / wound / middle ear mucosa)
│
▼
CONDITIONING FILM — Host proteins (fibronectin, fibrinogen) coat surface
│
▼
INITIAL ATTACHMENT — via adhesins, pili, flagella (minutes)
│
▼
MICROCOLONY FORMATION — irreversible attachment (2–4 hours)
│
▼
QUORUM SENSING ACTIVATED — autoinducers accumulate
│
▼
EPS MATRIX SECRETION — glycocalyx, proteins, eDNA (6–12 hours)
│
┌──────┴──────────────────────────────────┐
▼ ▼
MATURE BIOFILM PERSISTER CELLS FORMED
(mushroom colonies, water channels) (dormant, treatment-tolerant)
│
▼
ANTIBIOTIC APPROACHES BLOCKED BY:
├─→ EPS diffusion barrier
├─→ Anaerobic/low pH microenvironment
├─→ Efflux pumps (QS-regulated)
├─→ Horizontal gene transfer (resistance plasmids)
└─→ Persister cell dormancy
│
▼
HOST IMMUNE RESPONSE FRUSTRATED:
├─→ Complement blocked by EPS
├─→ Phagocytes cannot ingest bacteria
└─→ Frustrated neutrophils release ROS → TISSUE DAMAGE
│
▼
CHRONIC INFECTION ESTABLISHED
(CRS / COM / Chronic wounds / Osteomyelitis / Device infections)
│
▼
DISPERSAL → New planktonic cells shed → Spread to new sites
KEY REFERENCES FOR RGUHS
| Author/Textbook | Relevance |
|---|---|
| Cummings Otolaryngology – Head & Neck Surgery, 7th Ed. | Biofilm in CRS; antibiotic resistance in sinusitis |
| Scott-Brown's Otolaryngology | Biofilm in OME and CRS (referenced in current literature cited in Cummings) |
| Dhingra's Diseases of Ear, Nose & Throat | Tonsillar/adenoid biofilm; recurrent tonsillitis |
| Hazarika's Textbook of ENT | Chronic otitis media and biofilm-related pathology |
| Jawetz, Melnick & Adelberg's Medical Microbiology, 28th Ed. | Biofilm biology; resistance mechanisms |
| Sherris & Ryan's Medical Microbiology, 8th Ed. | Biofilm structure; dental and pathogenic biofilms |
| Rockwood & Green's Fractures in Adults, 10th Ed. (2025) | Biofilm in osteomyelitis; resistance quantification |
| Schwartz's Principles of Surgery, 11th Ed. | Biofilm in chronic wounds; EPS; treatment |
| Janeway's Immunobiology, 10th Ed. | EPS immunological evasion; complement blockade |
| Hall-Stoodley L et al., JAMA 2006 | Biofilm in middle ear mucosa (OME) |
| Costerton JW et al., Science 1999 | Landmark review of biofilm in medicine |
| Bjarnsholt T et al., APMIS 2013 | Biofilm definition and clinical relevance |
CONCLUSION
Biofilm represents the predominant mode of bacterial existence in chronic infections. Its formation is a highly organized, quorum sensing-driven process that culminates in a structured community capable of withstanding antibiotic concentrations 100 to 800 times greater than those needed to kill planktonic bacteria. In ENT practice, biofilm is central to the pathogenesis of chronic rhinosinusitis, chronic otitis media, recurrent tonsillitis, and device-related infections. The multiplicity of resistance mechanisms — physical barrier, metabolic dormancy, horizontal gene transfer, and immune evasion — makes biofilm-related infections among the most challenging clinical scenarios. Current management rests on surgical disruption combined with targeted antimicrobials; however, emerging strategies including anti-quorum sensing agents, bacteriophage therapy, nanoparticle drug delivery, and enzymatic dispersal offer promising advances for the future.
Sources: Cummings Otolaryngology Head & Neck Surgery, 7th Ed. | Jawetz Melnick & Adelberg's Medical Microbiology, 28th Ed. | Sherris & Ryan's Medical Microbiology, 8th Ed. | Rockwood & Green's Fractures in Adults, 10th Ed. (2025) | Schwartz's Principles of Surgery, 11th Ed. | Janeway's Immunobiology, 10th Ed.
This is a shared conversation. Sign in to Orris to start your own chat.