Complications of Peptic Ulcer Disease: (Gastric and duodenal bleeding, perforation. Clinical manifestations. Diagnostic methods. Conservative therapy, indications for hospitalization. Surgical management.)
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from multiple authoritative sources. Let me compile the full answer.
Complications of Peptic Ulcer Disease
Overview
The three most common complications of PUD, in decreasing order of frequency, are bleeding, perforation, and gastric outlet obstruction. Most peptic ulcer-related deaths in the United States are due to bleeding. NSAID and aspirin use are inextricably linked with all major complications, especially in the elderly.
— Schwartz's Principles of Surgery, 11e
I. Gastrointestinal Bleeding (Hemorrhage)
Epidemiology
- The most frequent complication of PUD; annual incidence of ulcer bleeding ranges from 19–57 per 100,000 individuals (~0.02–0.06%)
- Bleeding peptic ulcers are the most common cause of upper GI bleeding in hospitalized patients
- NSAIDs are linked to 50–75% of bleeding peptic ulcers, one-third of hemorrhage deaths, and 30% of hospitalizations
- NSAIDs increase bleeding risk 3-fold under age 65 and 8-fold over age 75 — Mulholland & Greenfield's Surgery, 7e; Sleisenger & Fordtran's GI Disease
Clinical Manifestations
- Melena and/or hematemesis are the classic presentations
- Nasogastric aspiration confirms upper GI bleeding
- Abdominal pain is quite uncommon with bleeding ulcers
- Hypovolemic shock may be present in severe hemorrhage, necessitating aggressive resuscitation and blood transfusion
- Signs/symptoms of chronic anemia may precede acute events
Diagnostic Methods
Endoscopy (EGD) is the gold standard:
- Early endoscopy (within 24 hours) diagnoses the bleeding source and guides hemostatic therapy
- The Forrest Classification stratifies endoscopic stigmata and prognosis:
| Type | Finding | Risk of Rebleeding |
|---|---|---|
| Ia | Spurting hemorrhage | Very high |
| Ib | Oozing hemorrhage | High |
| IIa | Nonbleeding visible vessel | ~50% |
| IIb | Adherent clot | ~30% |
| IIc | Flat pigmentation | Low |
| III | Clean-base ulcer | Very low |
- Active bleeding (Ia/Ib) and nonbleeding visible vessels (IIa) warrant endoscopic therapy
- Adherent clots (IIb): clot removal + endoscopic treatment lowers rebleeding risk from 30% → 5%
- Doppler endoscopic probe can detect arterial flow even under minor stigmata
Risk Stratification Scores:
- Glasgow Blatchford Score (GBS): uses BUN, Hgb, pulse, systolic BP, melena, syncope, cardiac/hepatic failure — predicts need for intervention; GBS ≤1 → outpatient management appropriate
- Rockall Score: composite pre- and post-endoscopy score predicting mortality (max 11; score ≤2 = low risk; score of 0 = negligible mortality, score of 5 = ~30% in-hospital mortality via AIMS65) — Schwartz's; Sleisenger & Fordtran's
Conservative (Medical) Management
- Nil by mouth + IV proton pump inhibitor (PPI): ~75% of patients stop bleeding spontaneously
- High-dose IV PPI (e.g., omeprazole 80 mg bolus → 8 mg/hr infusion) raises gastric pH, stabilizing clot
- Fluid resuscitation and blood transfusion as needed
- Treat H. pylori if positive — eradication dramatically reduces rebleeding risk
- Endoscopic hemostasis: epinephrine injection, electrocautery, hemoclips
Indications for Hospitalization
- Any significant upper GI bleed (hematemesis, melena, hematochezia with hemodynamic instability)
- Glasgow Blatchford Score ≥2 (score ≤1 may allow safe outpatient management)
- Hemodynamic instability: tachycardia, hypotension, shock
- Transfusion requirement
- Active bleeding or high-risk endoscopic stigmata (Forrest Ia, Ib, IIa)
- Significant comorbidities, advanced age, anticoagulant use
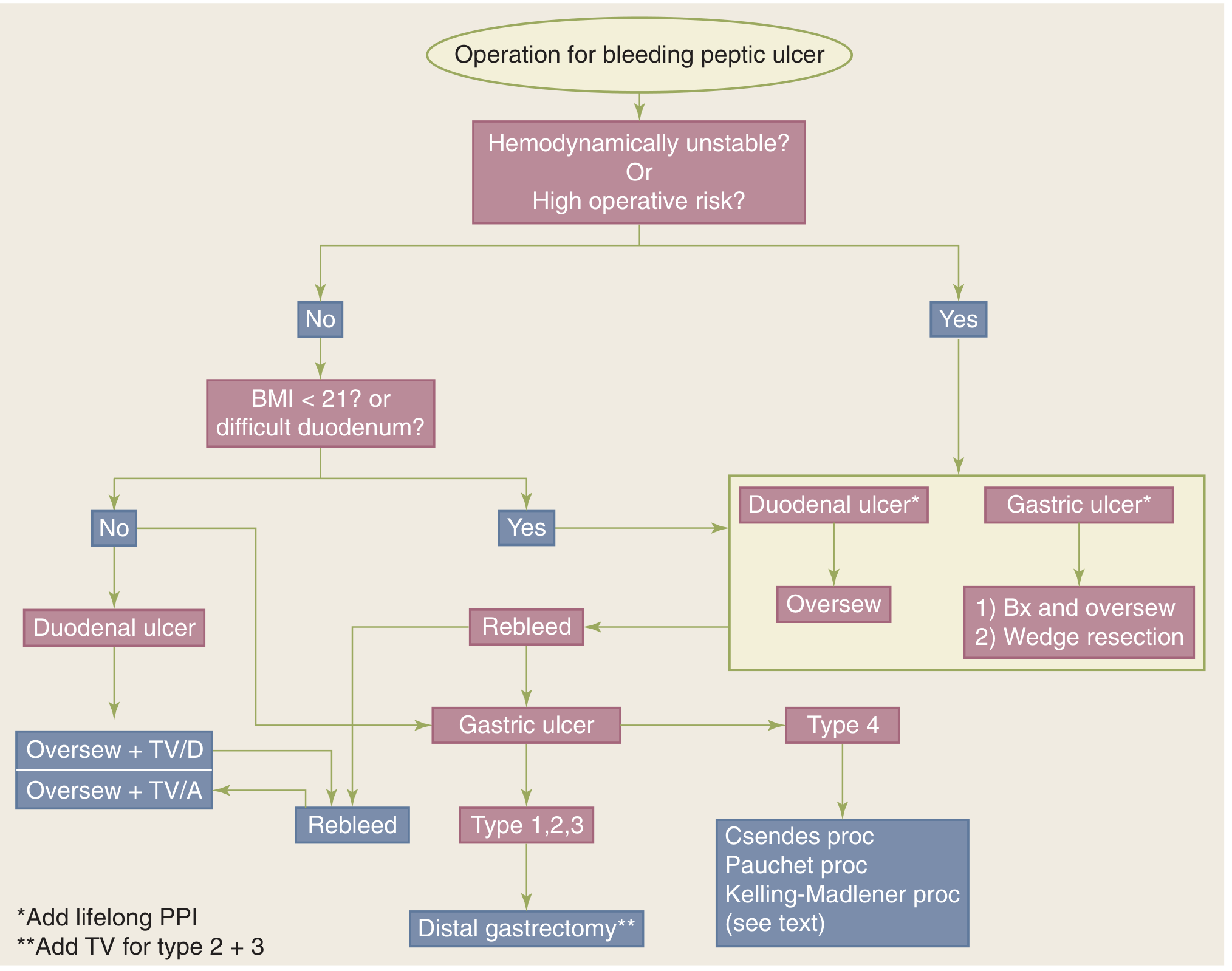
Surgical Management
- Indication: failure of 2 endoscopic attempts; persistent hemodynamic instability; transfusion >4 units/24 hours; rebleeding after initial control; deep ulcers on posterior duodenal bulb or lesser gastric curvature (erode the gastroduodenal artery — not amenable to endoscopy)
- Posterior duodenal ulcer: oversew the bleeding vessel (ligation of gastroduodenal artery), usually combined with pyloroplasty and truncal vagotomy (TV+D)
- Gastric ulcer: excision or distal gastrectomy (Billroth I or II)
- Operations for definitive treatment:
- Truncal vagotomy + antrectomy (TV+A): lowest rebleeding rate
- Truncal vagotomy + drainage (TV+D): pyloroplasty or gastrojejunostomy
- Highly selective vagotomy (HSV): preserves pylorus, less side effects
- Elderly patients and those with multiple comorbidities may benefit from early elective surgery after initially successful endoscopy, particularly with high-risk lesions, as they do not tolerate repeated hemorrhagic episodes — Schwartz's; Mulholland & Greenfield's; Sleisenger & Fordtran's
II. Perforation
Epidemiology
- Second most common complication; incidence 4–14 per 100,000 (0.004–0.014%)
- Now more common as an indication for operation than bleeding (as endoscopy controls most bleeding)
- Strongly associated with NSAID/aspirin use, especially in the elderly
- Many patients asymptomatic until perforation occurs
Clinical Manifestations
- Sudden, excruciating epigastric pain — patients can often give the exact time of onset
- Initially: chemical peritonitis from gastroduodenal secretions
- Within hours: bacterial peritonitis supervenes
- Obvious distress; board-like rigidity (involuntary guarding)
- Rebound tenderness on gentle examination
- Patient may lie motionless; any movement worsens pain
- Referred pain to shoulder (diaphragmatic irritation)
- Tachycardia, fever; hypotension in advanced cases (septic shock)
- Bowel sounds absent or diminished
Diagnostic Methods
- Upright chest X-ray or abdominal X-ray: free subdiaphragmatic air (pneumoperitoneum) in ~80% of cases — key finding
- If X-ray negative and suspicion remains: CT abdomen (more sensitive — detects small amounts of free air, localizes perforation, assesses peritoneal contamination)
- Water-soluble contrast study (Gastrografin): to confirm sealed perforation before nonoperative management
- Serum labs: leukocytosis, elevated amylase/lipase possible (pancreatic irritation), metabolic acidosis in advanced disease
- Endoscopy is contraindicated in suspected free perforation
Conservative Management
- Nonoperative (Taylor's method): reserved for highly selected cases only:
- Hemodynamically stable patient
- Radiologic (contrast study) evidence that the perforation has sealed spontaneously
- Absence of clinical peritonitis
- No signs of systemic sepsis
- Requires: nasogastric decompression, IV fluids, IV antibiotics, IV PPIs, close monitoring
- If patient deteriorates → immediate surgery
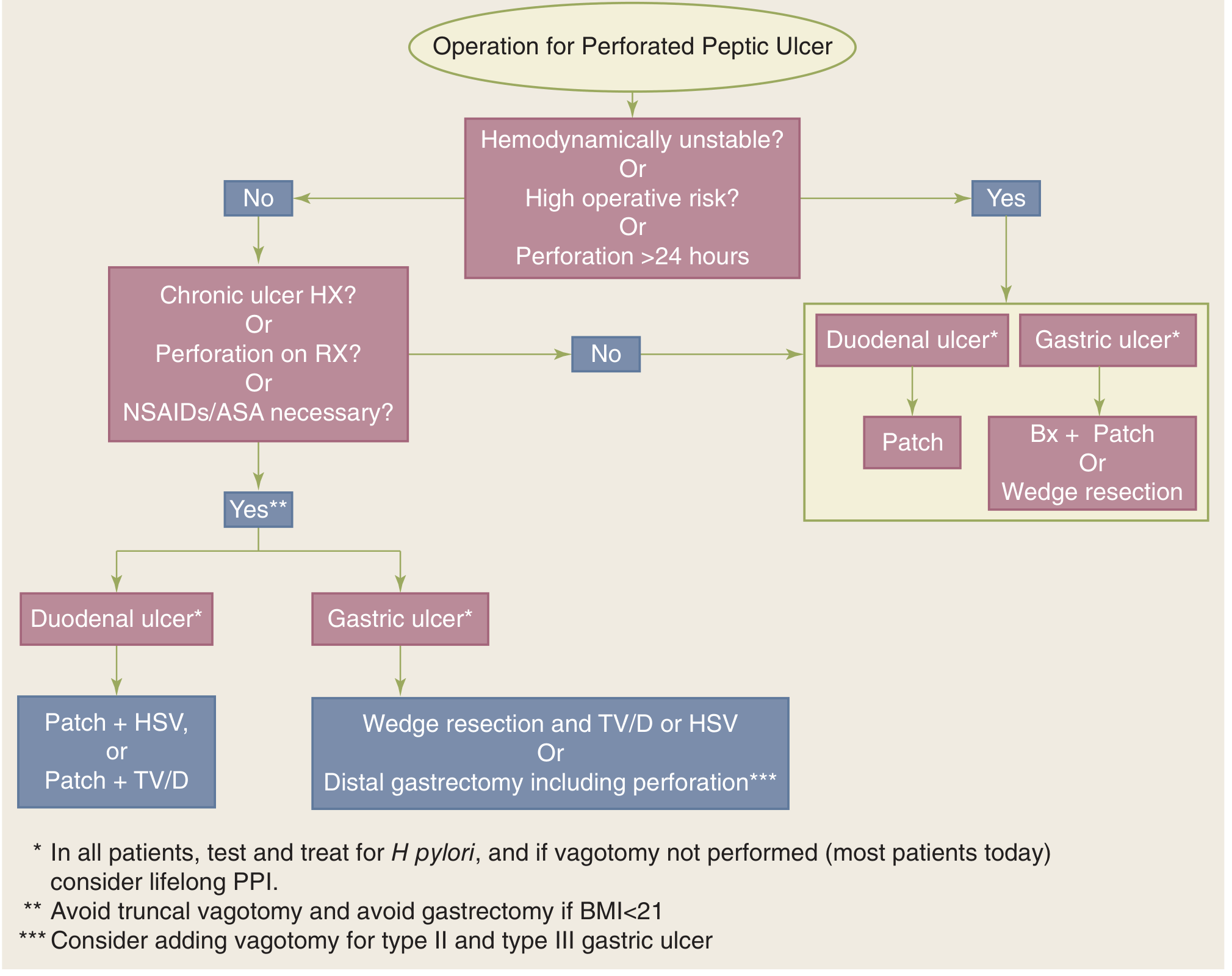
- Surgery is almost always indicated for acute perforation with peritonitis
Indications for Hospitalization
- All suspected perforations require emergency hospitalization
- Emergency resuscitation: IV fluids (significant third-space sequestration into inflamed peritoneum), IV antibiotics, analgesia, NG decompression, urinary catheter
Surgical Management
- Preoperative: IV fluid resuscitation (mandatory — third-space losses can be massive), antibiotics, analgesia
- Approach: open or laparoscopic (laparoscopic approach is increasingly favored in stable patients)
For Duodenal Perforation:
- Simple patch closure (Graham patch/omental patch): most commonly performed; procedure of choice for hemodynamic instability, exudative peritonitis (>24 hours), or significant contamination
- A pedicle of omentum is sutured over the perforation
- Patch closure + Highly Selective Vagotomy (HSV): in stable patients with chronic symptoms or failure of medical treatment
- Patch closure + Truncal Vagotomy + Drainage (TV+D): acceptable definitive operation for perforated duodenal ulcer
- There is a clear trend in the US and Western Europe away from definitive anti-ulcer surgery at the time of perforation (patch + postoperative H. pylori eradication is standard)
For Gastric Perforation:
- Distal gastric resection (preferred in stable patients): Billroth I gastroduodenostomy; vagotomy added for type II and III gastric ulcers
- Patch closure with biopsy or local excision and closure: for unstable/high-risk patients or unfavorable perforation location
- All perforated gastric ulcers must be biopsied (even prepyloric) — to exclude malignancy — Schwartz's Principles of Surgery, 11e; Sleisenger & Fordtran's
Surgical Algorithms:
III. Gastric Outlet Obstruction (for completeness)
- Occurs in ≤5% of PUD patients; caused by duodenal or prepyloric ulcer disease
- May be acute (inflammatory edema, dysmotility) or chronic (cicatrix/fibrosis)
- Presents with non-bilious projectile vomiting, weight loss, early satiety, distension
- Diagnosis: upper endoscopy, upper GI series, gastric emptying study
- Conservative: nasogastric decompression + IV PPIs (acute edematous obstruction)
- Endoscopic balloon dilation: immediate relief in 78–100%; many ultimately fail
- Surgical: vagotomy with drainage (pyloroplasty or gastrojejunostomy) or antrectomy
Summary Table: Complications at a Glance
| Feature | Bleeding | Perforation |
|---|---|---|
| Frequency | Most common | 2nd most common |
| Key symptom | Hematemesis/melena | Sudden severe epigastric pain |
| Key sign | Shock, NG blood | Board-like abdomen, peritonism |
| Key investigation | EGD (Forrest class) | Erect CXR (free air) → CT |
| Risk score | Glasgow Blatchford, Rockall | None standardized |
| Conservative Rx | IV PPI, endoscopic hemostasis | Omental patch + IV PPIs (if sealed) |
| Surgery | After 2 endoscopic failures | Almost always (unless sealed) |
| Main operation | Oversew + TV+D or TV+A | Graham patch ± vagotomy |
Key Sources:
- Schwartz's Principles of Surgery, 11th Edition
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
- Mulholland and Greenfield's Surgery, 7th Edition
Acute Intestinal Obstruction: (Classification. Methods for examining patients. Diagnosis. Surgical tactics. Features of preoperative and postoperative management of patients. Principles of conservative treatment and indications for surgical treatment. Chronic intestinal obstruction. Causes. Clinical presentation. Diagnosis. Conservative and surgical treatment. Adhesive disease)
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content from multiple authoritative sources. Let me compile the full, detailed answer.
Acute Intestinal Obstruction
I. Classification
Intestinal obstruction is classified along several axes:
A. By Mechanism
1. Mechanical Obstruction — a physical barrier to intestinal content movement
- Divided by location relative to the bowel wall:
| Category | Examples |
|---|---|
| Extrinsic (outside the wall) | Postoperative adhesions (~60% of SBO), external hernias, internal hernias, compressing tumors, abscesses, hematomas, volvulus |
| Intrinsic (within the wall) | Primary neoplasms, Crohn's disease, radiation enteritis, intestinal tuberculosis, intussusception, wall hematoma |
| Intraluminal | Bezoars, foreign bodies, gallstone ileus, Ascaris infestation |
2. Functional (Adynamic) Obstruction — Ileus
- No physical blockage; failure of normal coordinated peristalsis
- Causes: post-laparotomy, metabolic disease (especially hypokalemia), narcotics, retroperitoneal/pelvic/intrathoracic infection, abdominal trauma
3. Pseudo-obstruction (Ogilvie's Syndrome)
- Massive colonic dilation without mechanical cause; disruption of interstitial cells of Cajal pacemaker activity; associated with neurogenic/autonomic dysfunction
B. By Degree
- Partial (incomplete): some luminal content passes; less urgent
- Complete: no flatus or stool passes distally; requires prompt intervention
C. By Vascular Status
- Simple obstruction: no compromise of blood supply
- Strangulating obstruction: vascular compromise → ischemia → necrosis → perforation; mortality is double that of simple obstruction
- Closed-loop obstruction: bowel obstructed at two points (e.g., volvulus, internal hernia); intraluminal pressure rises rapidly; arterial obstruction → infarction — surgical emergency
D. By Location
- Small bowel obstruction (SBO): most common; predominantly due to adhesions
- Large bowel obstruction (LBO): most commonly due to colorectal carcinoma, volvulus (sigmoid > cecal), diverticular disease
E. By Acuity
- Acute (rapid onset, full obstruction)
- Chronic (gradual, partial, recurrent)
— Rosen's Emergency Medicine; Current Surgical Therapy 14e
II. Pathophysiology
Following obstruction, the bowel proximal to the blockage dilates due to accumulation of swallowed air, gastric secretions, bile, and pancreatic juice. The bowel wall becomes edematous and absorptive function decreases. Bacterial overgrowth in the normally sterile proximal small bowel explains the feculent vomiting seen in late SBO.
Fluid is lost into the bowel lumen (third-spacing) and transudated into the peritoneal cavity, causing hypovolemia and electrolyte abnormalities. Proximal obstruction produces metabolic alkalosis (from vomiting HCl); distal obstruction produces metabolic acidosis/dehydration.
In closed-loop obstruction, intraluminal pressure escalates rapidly (no retrograde flow), causing venous congestion → arterial compromise → ischemia → gangrene → perforation → peritonitis and sepsis.
III. Methods of Examining Patients
History
- Onset, character, and location of pain (colicky vs. continuous)
- Vomiting (bilious, feculent?)
- Last bowel movement and flatus passage
- Abdominal distension
- Prior abdominal surgeries, hernias, malignancy, IBD
Physical Examination
- Inspection: abdominal distension, visible peristalsis (advanced obstruction), surgical scars, hernias
- Auscultation: high-pitched, tinkling/rushing bowel sounds (mechanical obstruction early); absent bowel sounds (ileus or late strangulation)
- Percussion: tympany over distended loops
- Palpation: tenderness (peritoneal signs suggest strangulation), palpable hernias, masses
- Vital signs: tachycardia, fever, hypotension suggest strangulation/sepsis
- Rectal exam: impaction, blood, rectal mass
Red flags for strangulation / urgent surgery (from imaging + exam):
| Clinical Signs | Radiographic Signs |
|---|---|
| Severe/constant (non-colicky) pain | Free peritoneal fluid |
| Fever > 38.5°C | Mesenteric edema/fat stranding |
| Hypotension / tachycardia | Pneumatosis intestinalis |
| Mental status changes | Portal venous gas |
| Peritoneal signs | Lack of mesenteric vessels in closed loop |
| Non-reducible hernia with erythema | C- or U-shaped dilated loop |
IV. Diagnosis
Laboratory Studies
- CBC: leukocytosis (strangulation, perforation); anemia
- Metabolic panel: electrolyte imbalances (K⁺, Na⁺, Cl⁻, HCO₃⁻), BUN/Cr (renal failure, dehydration)
- Serum lactate: elevated → ischemia / strangulation
- Amylase/lipase: may be elevated (not specific)
- ABG: metabolic alkalosis (proximal) or acidosis (distal, strangulation)
- Type & screen (pre-op preparation)
Imaging
1. Plain Abdominal Radiography (AXR — supine + erect)
- First-line, rapid, available
- SBO: dilated small bowel loops (>3 cm) with multiple air-fluid levels ("step-ladder pattern"), absent colonic gas
- LBO: dilated colon proximal to obstruction (cecum >9 cm = risk of perforation; >12–14 cm = surgical emergency)
- Free air under diaphragm (erect film) → perforation
- Sigmoid volvulus: "coffee bean" sign; cecal volvulus: kidney-bean shape
2. CT Abdomen/Pelvis with Contrast (gold standard)
- Most sensitive and specific modality
- Identifies: site and cause of obstruction, transition point, degree (partial vs. complete), strangulation (mesenteric edema, fat stranding, pneumatosis, portal gas), free fluid, perforation
- Differentiates benign (smooth transition zone, mesenteric vascular changes) from malignant (mass, adenopathy, abrupt transition, irregular thickening) causes
- CT enteroclysis: water-soluble contrast via naso-enteric tube → better definition of low-grade or partial SBO
- Ultrasound: sensitivity ~85%, no radiation; useful in pregnancy; inferior to CT for defining transition zone
3. Water-Soluble Contrast Studies (Gastrografin)
- Oral Gastrografin: both diagnostic (appearance of contrast in colon within 24 h predicts resolution of adhesive SBO) and therapeutic (hyperosmolar effect draws fluid into lumen, reduces edema)
- Contrast enema: for LBO — identifies site, distinguishes from pseudo-obstruction
4. Endoscopy
- Flexible sigmoidoscopy/colonoscopy: in LBO, useful to decompress sigmoid volvulus or place colonic stent
- Contraindicated if peritonitis or perforation suspected
— Rosen's Emergency Medicine; Harrison's 22e; Current Surgical Therapy 14e
V. Principles of Conservative (Non-Operative) Treatment
Indications for conservative management:
- Partial (incomplete) SBO without peritonitis or hemodynamic instability
- Adhesive SBO (most resolve with non-operative management)
- Post-operative ileus
- Early post-operative SBO (within 4–6 weeks of surgery — adhesion edema likely)
- Patient with multiple prior surgeries and known adhesive disease
Components of conservative treatment:
- Nil per os (NPO) — bowel rest
- Nasogastric tube (NGT) decompression
- Removes swallowed air and gastric secretions, reduces distension, relieves vomiting
- Most useful in complete SBO and ileus; role debated in partial SBO
- IV Fluid resuscitation — isotonic crystalloids (NS or Lactated Ringer's) to correct volume depletion and electrolyte deficits
- Electrolyte replacement — especially potassium
- Analgesia — opioids acceptable; do not withhold pain relief
- Antiemetics
- Urinary catheter — to monitor urine output (target ≥0.5 mL/kg/hr)
- Serial clinical reassessment — vital signs, abdominal exam, fluid balance every 4–6 hours
- Water-soluble contrast (Gastrografin) challenge: in adhesive partial SBO — both diagnostic and therapeutic; appearance of contrast in colon within 24 h predicts resolution; also reduces need for surgery and length of stay
Duration of conservative trial: typically 24–72 hours for adhesive SBO; if no improvement → surgery
VI. Indications for Hospitalization
All patients with suspected mechanical intestinal obstruction require hospital admission for:
- IV resuscitation and monitoring
- Serial examination
- Surgical availability if clinical deterioration occurs
- Any complete obstruction
- Any partial obstruction with risk factors for strangulation
Low-risk patients (partial SBO, Blatchford-like scores, reliable follow-up) may rarely be observed as outpatients but this is exceptional.
VII. Indications for Surgical Treatment
Emergency (immediate) surgery:
- Peritonitis or signs of bowel strangulation/ischemia (fever, tachycardia, leukocytosis, lactic acidosis, peritoneal signs)
- Complete obstruction with no improvement after 24–48 h resuscitation
- Closed-loop obstruction (on CT or clinically suspected)
- Non-reducible (incarcerated/strangulated) hernia
- Cecal dilation >12–14 cm (risk of perforation)
- Sigmoid or cecal volvulus not reducible endoscopically
- Perforation (free air)
- Hemodynamic instability attributable to obstruction
Elective/urgent surgery (after optimization):
- LBO due to colorectal cancer (resection + stoma or primary anastomosis)
- Recurrent adhesive SBO with multiple hospital admissions
- Failure of non-operative management after 48–72 hours
VIII. Surgical Tactics
Principles of Surgery for SBO
- Exploration and identification of the transition point (cause of obstruction)
- Assessment of bowel viability: pink color, peristalsis, bleeding at cut edge, Doppler signal in mesentery; if doubtful → warm saline packs + 10–15 min waiting → re-assess; if still not viable → resect
- Relief of obstruction:
- Adhesiolysis: division of adhesive bands (sharp or harmless energy)
- Bowel resection with primary anastomosis (if well-perfused, non-contaminated field) or stoma formation (if ischemic bowel, contamination, hemodynamically unstable → Hartmann's procedure or loop stoma)
- Manual reduction of volvulus ± fixation (pexia) or resection
- Hernia repair with bowel reduction or resection
- Decompression of grossly dilated bowel if needed (milking content proximally into stomach → NGT, or enterotomy decompression)
- Laparoscopic approach: increasingly used for adhesive SBO; reduced morbidity, shorter hospital stay; convert to open if bowel is distended, dense adhesions, or if bowel viability uncertain
SBO with Strangulation
- Urgent resection of non-viable bowel
- Primary anastomosis if: hemodynamically stable, non-contaminated, adequate tissue perfusion
- Stoma if: unstable, contaminated, ischemic/necrotic bowel, nutritionally depleted patient
LBO Surgical Options
- Right-sided obstruction (cecum → transverse colon): right hemicolectomy + primary ileocolic anastomosis (generally safe)
- Left-sided obstruction (descending → sigmoid):
- Hartmann's procedure: resection of sigmoid/left colon, end-colostomy, rectal stump closure — safest in emergency; stoma reversal later
- Primary resection + anastomosis ± on-table lavage: in selected stable patients
- Loop colostomy + deferred resection: for high-risk patients
- Endoluminal stenting: self-expanding metal stent (SEMS) as a bridge to elective surgery — decompresses, allows bowel prep; reduces stoma rate
- Sigmoid volvulus: initial endoscopic decompression/detorsion via rigid sigmoidoscopy or colonoscopy; if successful → elective sigmoid resection to prevent recurrence; if fails or gangrenous → emergency resection + Hartmann's
IX. Pre-Operative and Post-Operative Management
Pre-Operative Management
- Resuscitation: aggressive IV fluids (isotonic crystalloids), correct electrolyte deficits (especially hypokalemia)
- NGT decompression: decompress stomach, reduce aspiration risk, facilitate anesthesia
- Urinary catheter: monitor urine output
- Blood tests: CBC, electrolytes, coagulation, group & screen, lactate, ABG
- IV antibiotics: broad-spectrum cover for gram-negative and anaerobes (e.g., cefazolin + metronidazole or piperacillin-tazobactam) before operation — especially for strangulation, perforation, or prolonged obstruction with bacterial translocation risk
- Analgesia: do not withhold; opioids acceptable
- Thromboprophylaxis: low-molecular-weight heparin (if time permits)
- Informed consent and marking for possible stoma
Post-Operative Management
- Fluid balance: maintain IV fluids until return of bowel function
- NGT: continue decompression until return of bowel function (passage of flatus/stool); remove when output <200–300 mL/day
- Early mobilization: reduces ileus, DVT risk
- Nutrition: early enteral feeding when feasible; TPN for prolonged ileus or short bowel
- Analgesia: multimodal (epidural, regional blocks, NSAIDs, paracetamol) to minimize opioids → reduces post-op ileus
- Antibiotics: continue post-operatively if contaminated field, until clinical improvement
- Monitor for complications: anastomotic leak (day 3–7: fever, tachycardia, peritonism, rising CRP), wound infection, stoma dysfunction, DVT/PE, respiratory complications
- Enhanced Recovery After Surgery (ERAS) pathways: early feeding, early mobilization, minimize opioids, restrict fluids → significantly reduce length of stay
- Stoma care: if formed, educate patient; mark pre-operatively with stoma nurse; reversal planned at 3–6 months
X. Chronic Intestinal Obstruction
Definition
Persistent or recurrent partial obstruction that is longstanding, often incomplete, and may evolve gradually.
Causes
- Adhesions (most common) — recurrent partial SBO from prior surgery or peritonitis
- Colorectal/small bowel malignancy — progressive luminal narrowing
- Crohn's disease — strictures from chronic inflammation + fibrosis
- Radiation enteritis — fibrotic strictures following pelvic/abdominal radiation
- External hernias — intermittent incarceration
- Diverticular disease — stricturing from recurrent diverticulitis
- Extrinsic compression — lymphoma, retroperitoneal fibrosis, endometriosis
- Pseudo-obstruction (Ogilvie's) — functional, no mechanical cause
Clinical Presentation
- Recurrent colicky abdominal pain, often post-prandial (food aversion)
- Abdominal distension — episodic, partially relieving with passage of gas/stool
- Nausea and vomiting — often intermittent, not as acute as in complete obstruction
- Altered bowel habit — alternating constipation and diarrhea; incomplete evacuation
- Weight loss — due to food avoidance (fear of pain), malabsorption
- Borborygmi — loud, hyperactive bowel sounds
- Episodes may partially resolve spontaneously or with conservative measures, only to recur
Diagnosis
- Clinical history is key — recurrent episodes, prior surgeries
- CT abdomen/pelvis: identifies strictures, transition point, degree of dilation, cause (adhesion vs. mass vs. inflammatory)
- Barium small bowel follow-through / CT enterography: excellent for mapping small bowel strictures (especially Crohn's); shows "string sign," mucosal detail
- CT enteroclysis: superior small bowel distension and visualization of partial strictures
- Colonoscopy / flexible sigmoidoscopy: for colonic causes (cancer, diverticular stricture, Crohn's colitis)
- Gastrografin challenge: diagnostic + therapeutic
- Laboratory: CBC (anemia, leukocytosis if active IBD), inflammatory markers (CRP, ESR), nutritional status (albumin, pre-albumin)
Conservative Treatment
- Dietary modification: low-fiber diet to reduce bulk; small, frequent meals
- Liquid/elemental diet: during acute exacerbations to reduce intestinal load
- IV fluids during flares with vomiting
- Corticosteroids (in active Crohn's strictures with inflammatory component): oral prednisolone or IV methylprednisolone
- Biologic therapy: for Crohn's (infliximab, adalimumab) — can reduce inflammatory component of stricture
- Octreotide: reduces GI secretions; useful in malignant partial obstruction
- Endoscopic balloon dilation: first-line for accessible short fibrous strictures (Crohn's, anastomotic, post-radiation); effective in 78–100% immediately; many require repeat sessions
- SEMS (self-expanding metal stents): for malignant colonic strictures as palliation or bridge to surgery
Surgical Treatment
- Adhesiolysis: for adhesive recurrent SBO — laparoscopic preferred if feasible
- Intestinal resection with primary anastomosis: for fibrotic/malignant strictures
- Strictureplasty (Crohn's disease — bowel-sparing):
- Heineke-Mikulicz: for strictures <7 cm — longitudinal incision closed transversely
- Finney: for 8–15 cm strictures — U-shaped closure
- Michelassi (side-to-side isoperistaltic): for long/multiple strictures — advanced technique
- Diverting stoma: palliative decompression in inoperable malignant obstruction
- Colonic resection (for diverticular stricture, cancer): Hartmann's or primary anastomosis based on patient fitness
XI. Adhesive Disease (Peritoneal Adhesions)
Definition
Fibrous bands of scar tissue forming between loops of bowel, or between bowel and peritoneum/abdominal wall, following peritoneal injury.
Pathogenesis
- Normal healing of peritoneal injury involves fibrinous exudate → fibrin deposition → organization by fibroblasts → fibrous adhesion
- Failure of normal fibrinolysis (peritoneal fibrinolytic activity is suppressed by surgery, infection, ischemia, foreign bodies) allows permanent adhesion formation
- Adhesions may be:
- Filmy / avascular: easily divided
- Dense / vascular: difficult, risky to lyse; risk of enterotomy
Causes / Risk Factors
- Prior abdominal surgery (accounts for ~60% of all SBO; ~75% of SBO following colorectal surgery)
- Intra-abdominal infection / peritonitis
- Pelvic inflammatory disease
- Endometriosis
- Abdominal/pelvic radiotherapy
- Inflammatory bowel disease
- Prior peritoneal adhesions (self-perpetuating)
Clinical Consequences
- SBO (acute or recurrent chronic)
- Chronic abdominal/pelvic pain
- Female infertility (pelvic adhesions)
- Difficult re-operative surgery
Diagnosis
- CT scan: shows transition point at adhesion band (no mass lesion); mesenteric vascular changes; may show fixed, angulated bowel loop
- "Diagnosis of exclusion" — no mass, no hernia, history of prior surgery
Treatment
Conservative (initial):
- NGT, IV fluids, bowel rest (as above for SBO)
- Gastrografin challenge: therapeutic + diagnostic; if contrast reaches colon within 24 h → 97% resolve without surgery
- Success rate of non-operative management for adhesive SBO: 70–80% overall
Surgical:
- Adhesiolysis: sharp or energy-device division of adhesive bands
- Laparoscopic adhesiolysis: preferred for limited adhesions; shorter hospital stay, faster recovery; technical difficulty increases with dense adhesions or prior multiple laparotomies
- Open adhesiolysis: for complex, dense adhesions; allows tactile feedback; safer for enterotomy repair
- Noble plication / Baker tube plication: rarely used historical operations to stent bowel in organised position and prevent re-adhesion
- Bowel resection only if bowel is non-viable
Prevention (at surgery):
- Meticulous peritoneal hemostasis
- Minimizing tissue trauma and foreign material (e.g., powder-free gloves, minimal suture)
- Irrigation of peritoneal cavity
- Anti-adhesion barriers: hyaluronic acid–carboxymethylcellulose membrane (Seprafilm) — placed over bowel at closure; shown to reduce adhesion formation and SBO incidence
- Laparoscopic approach (vs. open) significantly reduces adhesion burden
Prognosis
- After a single episode of adhesive SBO requiring surgery: ~30% risk of recurrence within 10 years
- Each reoperation increases adhesion burden and operative risk
- Goal of management is to avoid unnecessary surgery and prevent recurrence
Summary: Acute vs. Chronic Obstruction
| Feature | Acute SBO | Chronic/Recurrent SBO |
|---|---|---|
| Onset | Sudden | Insidious, episodic |
| Pain | Severe colicky | Intermittent post-prandial |
| Vomiting | Prominent, feculent | Intermittent, nausea |
| Most common cause | Adhesions (60%) | Adhesions, Crohn's, cancer |
| First investigation | Erect AXR → CT | CT, barium follow-through |
| Initial management | NGT, IV fluids, Gastrografin | Dietary, conservative, dilation |
| Surgery | Adhesiolysis / resection | Strictureplasty / resection |
Sources:
- Rosen's Emergency Medicine, 10th Edition
- Current Surgical Therapy, 14th Edition
- Harrison's Principles of Internal Medicine, 22nd Edition
- Sleisenger and Fordtran's Gastrointestinal and Liver Disease
Lab test results of necrosis and ischemic
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
I now have solid source material from Tintinalli's and Rosen's Emergency Medicine. Combined with core pathophysiology knowledge, I can deliver a comprehensive, organ-system-organized answer.
Laboratory Test Results in Necrosis and Ischemia
The question is broad — ischemia and necrosis can affect any organ. Below is a systematic, organ-by-organ overview of the key laboratory findings, organized by marker type and clinical context.
I. Understanding the Basis
Ischemia = inadequate blood/oxygen delivery to tissue → anaerobic metabolism → accumulation of metabolic byproducts (lactate, H⁺, etc.)
Necrosis = irreversible cell death → disruption of cell membranes → release of intracellular enzymes, proteins, and ions into the bloodstream
The principle underlying all necrosis markers: intracellular contents leak into circulation when cell membranes rupture.
II. Universal / Non-Organ-Specific Markers
Serum Lactate
- Mechanism: anaerobic glycolysis during tissue hypoxia produces lactic acid
- Normal: < 2 mmol/L
- Elevated in: any ischemia (mesenteric, limb, myocardial, shock), sepsis, liver failure
- Significance: lactate >2 mmol/L = tissue hypoperfusion; >4 mmol/L = severe shock, high mortality
- Serial lactate clearance is used to monitor treatment response
CBC (Complete Blood Count)
- Leukocytosis (WBC >11,000/µL): reactive response to necrosis/inflammation; may signal infarction, infection, or strangulation
- Neutrophilia with left shift: especially in bowel ischemia, mesenteric infarction
- Anaemia: from haemorrhagic necrosis or haemolysis
Metabolic Panel / ABG
- Metabolic acidosis (↓ HCO₃, ↓ pH, ↑ anion gap): hallmark of significant ischemia/necrosis; lactic acidosis drives the anion gap up
- Hyperkalemia: from cellular lysis releasing intracellular K⁺ (massive necrosis, rhabdomyolysis, renal ischemia)
- Elevated BUN and creatinine: renal ischemia or pre-renal failure from shock
LDH (Lactate Dehydrogenase)
- Ubiquitous intracellular enzyme found in heart, liver, kidney, muscle, RBCs
- Released from any necrotic tissue; non-specific
- Elevated in: myocardial infarction, pulmonary infarction, hepatic necrosis, haemolysis, renal infarction, massive tissue necrosis
- LDH isoenzymes:
- LDH-1, LDH-2: cardiac, RBC predominant
- LDH-4, LDH-5: hepatic, skeletal muscle predominant
- "Flipped" LDH-1 > LDH-2 pattern: historically used for myocardial infarction (largely superseded by troponin)
III. Cardiac Ischemia and Necrosis (Myocardial Infarction)
The most extensively studied necrosis biomarker system.
Cardiac Troponins (cTnI, cTnT) — Gold Standard
- Mechanism: Troponin I and T are structural proteins of the cardiac contractile apparatus; released only when myocyte membrane integrity is lost (necrosis)
- Rise: begins 3–6 hours after onset of myocardial necrosis
- Peak: 12–24 hours (cTnI); 12–48 hours (cTnT)
- Duration of elevation: 7–10 days (cTnI); up to 14 days (cTnT) — useful for late presentation
- Diagnostic threshold: >99th percentile of a normal reference population
- High-sensitivity troponins (hs-cTn): detect 90–100% of AMI at presentation; used in 0h/1h or 0h/2h rapid rule-in/rule-out protocols
- Delta troponin: a rising or falling pattern over 1–3 hours distinguishes acute from chronic myocardial injury
- Non-ACS causes of troponin elevation: myocarditis, PE, sepsis, renal failure, heart failure, cardioversion, cardiac contusion, stroke, rhabdomyolysis — troponin reflects myocardial injury, not always ischaemic necrosis — Rosen's Emergency Medicine; Tintinalli's Emergency Medicine
CK-MB (Creatine Kinase MB Isoenzyme)
- More cardiac-specific than total CK; superseded by troponin but still used
- Rise: 3–6 hours after MI
- Peak: 12–24 hours
- Returns to normal: 36–72 hours (earlier than troponin → useful for re-infarction detection)
- Sensitivity: single presentation — ~42%; serial measurement — 69–99%
- CK-MB/total CK ratio >5–6%: suggests cardiac origin vs. skeletal muscle
Total CK (Creatine Kinase)
- Found in heart, skeletal muscle, brain (CK-MM, CK-MB, CK-BB)
- Rises 4–8h post-MI; peaks 24–36h; returns to baseline in 3–4 days
- Low sensitivity and specificity for cardiac necrosis alone (also elevated in rhabdomyolysis, trauma, IM injections)
- Single measurement sensitivity for AMI: only ~37%
Myoglobin
- Earliest marker to rise (1–3 hours after MI)
- Very sensitive, very non-specific (also elevated in skeletal muscle injury)
- Returns to normal within 24 hours
- Useful mainly to rule out MI early if negative
BNP / NT-proBNP (B-type Natriuretic Peptide)
- Released from ventricular myocytes in response to wall stress (volume/pressure overload)
- Elevated in acute MI with left ventricular dysfunction, heart failure
- Prognostic: elevated BNP in ACS = higher risk of adverse cardiovascular events
Summary Table — Cardiac Necrosis Markers
| Marker | Rise | Peak | Normalises | Notes |
|---|---|---|---|---|
| Myoglobin | 1–3 h | 6–9 h | 24 h | First to rise; non-specific |
| CK-MB | 3–6 h | 12–24 h | 36–72 h | Re-infarction detection |
| Total CK | 4–8 h | 24–36 h | 72–96 h | Low specificity |
| Troponin I | 3–6 h | 12–24 h | 7–10 days | Gold standard |
| Troponin T | 3–6 h | 12–48 h | 10–14 days | Gold standard |
| hs-Troponin | 1–3 h | — | — | Rapid rule-in/out |
IV. Skeletal Muscle Ischemia and Necrosis (Rhabdomyolysis / Limb Ischemia)
CK (Total) — Hallmark
- Primary marker for skeletal muscle necrosis
- CK >1,000 U/L = significant muscle injury; >5,000 U/L = rhabdomyolysis
- Rises within hours of injury; peaks at 24–72 hours
Myoglobin
- Released from damaged myocytes → myoglobinuria (urine turns brown/tea-coloured)
- Can cause acute kidney injury by tubular obstruction and direct nephrotoxicity
Urinalysis
- Myoglobinuria: dipstick positive for blood, but no RBCs on microscopy
- Urinary casts, haematuria in renal involvement
Electrolytes
- Hyperkalemia: release of intracellular K⁺ from necrotic muscle
- Hypocalcemia (early): calcium deposits in necrotic muscle
- Hyperphosphatemia: release from necrotic cells
- Hyperuricemia: purine release from cell nuclei
Renal Function
- ↑ Creatinine, ↑ BUN: myoglobin-induced AKI
LDH, AST, ALT
- Also elevated in rhabdomyolysis (released from skeletal muscle); may mimic hepatic injury
V. Hepatic Ischemia and Necrosis ("Shock Liver" / Ischaemic Hepatitis)
AST and ALT (Aminotransferases)
- Released from necrotic hepatocytes
- In acute hepatic ischemia: dramatic elevation — AST and ALT may rise to >1,000–10,000 U/L (100–1,000× normal) within 24–72 hours
- AST > ALT (because AST is also in myocardium and muscle)
- Ratio: AST/ALT > 2:1 suggests ischaemic or alcoholic pattern
LDH
- Markedly elevated in ischaemic hepatitis (LDH >5× normal is characteristic)
- LDH elevation disproportionate to aminotransferase elevation is a clue to ischaemic aetiology vs. viral hepatitis
Bilirubin, Alkaline Phosphatase
- Elevated but usually less dramatically; rise later
Coagulation
- Prolonged PT/INR: synthetic function impaired by hepatic necrosis
- In severe ischaemic hepatitis: coagulopathy can be profound
Ammonia
- Elevated with massive hepatic necrosis (hepatic encephalopathy)
VI. Renal Ischemia and Necrosis (Acute Kidney Injury / Acute Tubular Necrosis)
Creatinine and BUN
- ↑ Serum creatinine (rising by >0.3 mg/dL in 48h or >1.5× baseline in 7 days = AKI by KDIGO criteria)
- BUN:creatinine ratio:
-
20:1 = pre-renal (ischaemia without tubular necrosis yet)
- ~10–15:1 = intrinsic renal (established ATN)
-
Urinalysis and Microscopy
- Granular "muddy brown" casts: pathognomonic of acute tubular necrosis
- Tubular epithelial cells in urine sediment
- Proteinuria (mild–moderate)
- Urine sodium: >40 mEq/L in ATN (tubules cannot reabsorb Na); <20 mEq/L in pre-renal
- FENa (Fractional Excretion of Sodium): >2% in ATN; <1% in pre-renal
Electrolytes
- Hyperkalemia (impaired K⁺ excretion)
- Metabolic acidosis (impaired H⁺ and bicarbonate handling)
- Hyperphosphatemia, hypocalcemia
VII. Bowel / Mesenteric Ischemia and Necrosis
Serum Lactate — Most Important
- Elevated (usually >2 mmol/L); severe ischemia/necrosis → >4 mmol/L
- Lacks sensitivity and specificity early in the disease course; a normal lactate does not exclude mesenteric ischemia
WBC
- Leukocytosis with left shift: common but non-specific; severe leukocytosis (>20,000) with abdominal pain is alarming
Metabolic Acidosis
- Anion gap metabolic acidosis from lactic acid production in ischaemic bowel
LDH, CK, Amylase
- Non-specifically elevated with transmural bowel necrosis and peritonitis
- Amylase may be elevated (from pancreatic and salivary gland stimulation, or intestinal mucosa injury)
D-Dimer
- Often elevated; used as a negative predictor — normal D-dimer makes mesenteric venous thrombosis unlikely
Phosphate
- Hyperphosphataemia: associated with intestinal ischaemia/necrosis in some series
No single lab test is sensitive or specific for mesenteric ischaemia — CT angiography remains the definitive diagnostic modality
VIII. Pulmonary Infarction (Pulmonary Embolism with Infarction)
Troponin
- Elevated in significant PE with right ventricular strain/necrosis
- Prognostic: elevated troponin in PE = higher in-hospital mortality
BNP / NT-proBNP
- Elevated with right ventricular pressure overload
D-Dimer
- Sensitive but non-specific: elevated in PE, but also in MI, infection, post-op, malignancy
- High negative predictive value (Wells score low + negative D-dimer = PE ruled out)
ABG
- Hypoxia (↓ PaO₂), hypocapnia (↓ PaCO₂ from hyperventilation), respiratory alkalosis
- A-a gradient increased
LDH
- Elevated in pulmonary infarction
Summary Table — Key Lab Findings by Organ
| Organ | Primary Markers | Supporting Labs |
|---|---|---|
| Heart | Troponin I/T (↑↑↑), CK-MB, Myoglobin | LDH (↑), BNP, metabolic acidosis |
| Skeletal muscle | Total CK (↑↑↑), Myoglobin | Hyperkalemia, Hyperphosphatemia, ↑ Cr |
| Liver | AST/ALT (↑↑↑), LDH (↑↑) | ↑ PT/INR, ↑ Bilirubin, ↑ Ammonia |
| Kidney | Creatinine (↑), BUN (↑) | Muddy-brown casts, FENa >2%, Hyperkalemia |
| Bowel/Mesentery | Lactate (↑), WBC (↑↑) | Metabolic acidosis, ↑ LDH, ↑ Amylase, ↑ D-dimer |
| Lung | D-dimer (↑), Troponin (↑) | ABG: hypoxia, ↑ A-a gradient, ↑ BNP |
| Universal | Lactate, WBC, Metabolic acidosis | LDH, Anion gap ↑, Electrolyte disturbances |
Sources:
- Rosen's Emergency Medicine, 10th Edition
- Tintinalli's Emergency Medicine, A Comprehensive Study
This is a shared conversation. Sign in to Orris to start your own chat.