Propofol
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Loading Image
Searching PubMed
propofol anesthesia pharmacology
I now have comprehensive information to provide a thorough answer on propofol.
Propofol (2,6-Diisopropylphenol)
Structure & Formulation
Propofol is an alkylphenol — specifically 2,6-diisopropylphenol. It is highly lipid-soluble and insoluble in water, necessitating an emulsion formulation:
- 1% propofol + 10% soybean oil + 1.2% purified egg phospholipid (emulsifier) + 2.25% glycerol (tonicity) + sodium hydroxide (pH adjustment) + EDTA (bacteriostatic)
- Appears as a milky-white viscous emulsion; pH 7
- Stable at room temperature, not light-sensitive; may be diluted with 5% dextrose
Fospropofol (Lusedra): A water-soluble prodrug approved by FDA in 2008 for monitored anesthesia care. Metabolized by alkaline phosphatases to propofol. Avoids injection pain but causes perineal paresthesias and pruritus after bolus dosing.
Mechanism of Action
Propofol's primary mechanism is enhancement of GABA-mediated inhibitory neurotransmission at GABA-A receptors — increasing chloride conductance and hyperpolarizing the neuronal membrane. This leads to CNS depression ranging from sedation to general anesthesia in a dose-dependent manner. Additional actions include inhibition of NMDA receptor activity and modulation of glycine receptors.
Pharmacokinetics
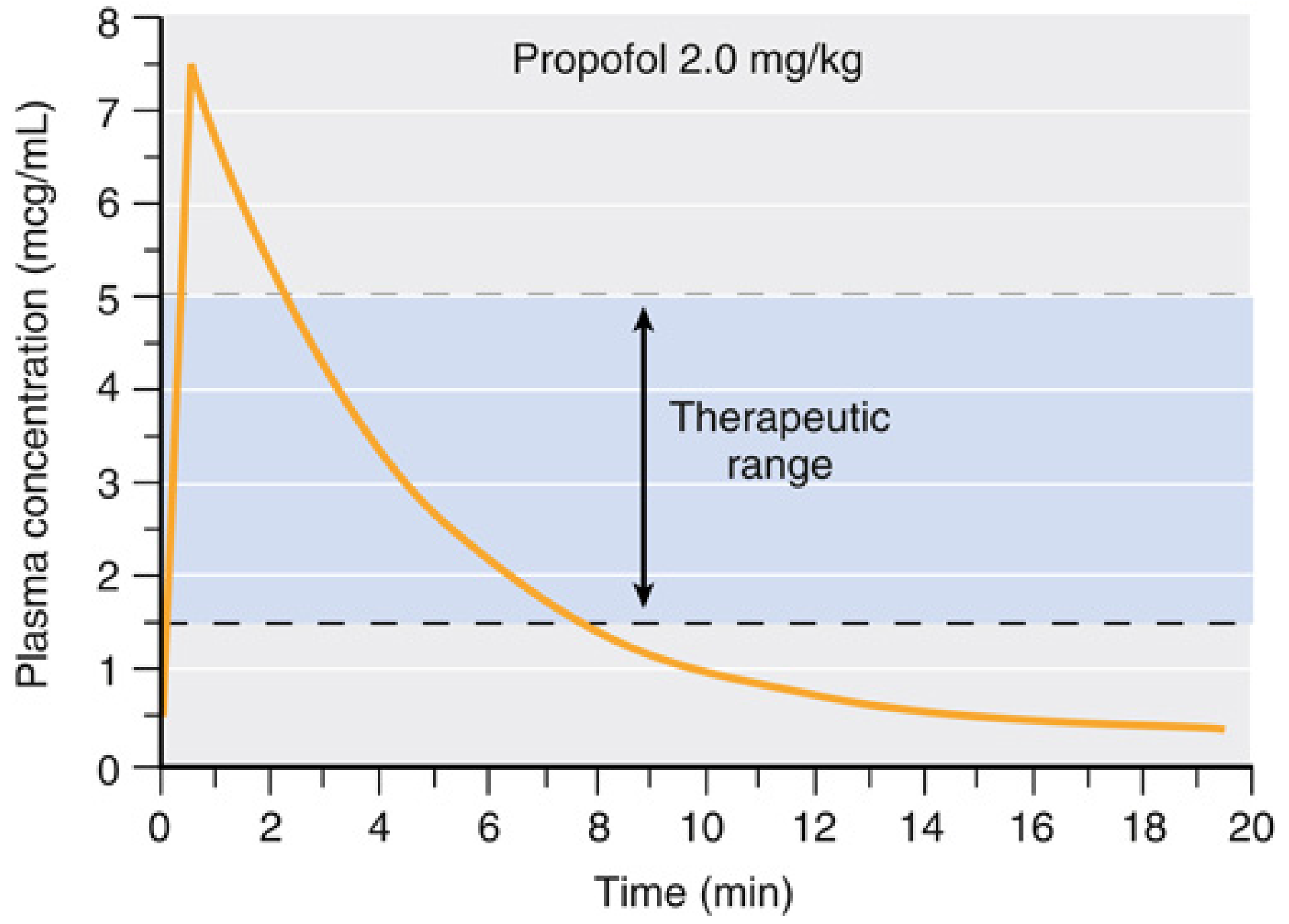
After an induction bolus, plasma levels fall rapidly due to redistribution; levels required for surgical anesthesia are 2–5 mcg/mL, with awakening typically occurring below 1.5 mcg/mL.
| Parameter | Value |
|---|---|
| Initial distribution t½ | 2–8 min |
| Slow distribution t½ | 30–70 min |
| Elimination t½ | 4–23.5 hours |
| Context-sensitive half-time (≤8h infusion) | < 40 min |
| Volume of distribution (central compartment) | 6–40 L |
| Volume of distribution (steady state) | 150–700 L |
| Clearance | 1.5–2.2 L/min (exceeds hepatic blood flow) |
Metabolism: Oxidized in the liver to 1,4-diisopropyl-1,4-quinol (glucuronide conjugate excreted in urine). Extrahepatic metabolism also occurs (~30% first-pass pulmonary uptake). Less than 1% is excreted unchanged. After a 2.5-hour anesthetic, propofol metabolites appear in urine for >60 hours.
Drug interactions: Propofol is a CYP3A4 inhibitor — at blood concentrations of 3 mcg/mL, CYP3A4 activity is reduced ~37%. Midazolam elevates propofol blood levels by ~25% due to competition at this enzyme.
Elderly patients: Reduced cardiac output → smaller central compartment → higher peak plasma concentration; doses should be reduced.
Clinical Uses & Dosing
| Indication | Dose |
|---|---|
| IV induction (adults) | 1.5–2.5 mg/kg IV |
| IV induction (elderly/compromised) | 1–1.5 mg/kg IV (reduced) |
| Maintenance of anesthesia | 100–150 mcg/kg/min infusion |
| ICU/procedural sedation | 25–75 mcg/kg/min infusion |
| Target blood concentration for surgery | 2–5 mcg/mL |
Target-controlled infusion (TCI) systems (e.g., Marsh, Schnider models) are widely used to achieve precise pharmacokinetic targets. A 2025 systematic review (PMID 40289063) confirms that TCI propofol remains highly effective for individualized anesthesia.
Organ System Effects
Central Nervous System
- Decreases cerebral blood flow (CBF) by 53–79% and cerebral metabolic rate (CMR) by 48–58% (PET data in volunteers)
- Reduces ICP and CSF pressure
- CO₂ reactivity and cerebral autoregulation are preserved, even at burst-suppression doses
- Does not have analgesic properties
- Antiemetic at sub-anesthetic doses (a major clinical advantage)
- EEG: Produces dose-dependent slowing → burst suppression at high doses
⚠️ In patients with elevated ICP, propofol can cause a critical reduction in CPP (<50 mmHg) unless mean arterial pressure is supported.
Cardiovascular System
- Hypotension — the most clinically significant adverse effect; results from vasodilation (decreased SVR) and a modest decrease in myocardial contractility
- Decreases myocardial blood flow and oxygen consumption — global O₂ supply-to-demand ratio is likely preserved
- Heart rate is generally unchanged or mildly reduced
- Baroreflex responses are attenuated
Respiratory System
- Dose-dependent respiratory depression — apnea is common after induction bolus
- Reduces tidal volume and respiratory rate
- Bronchodilator in patients with reactive airway disease (effect depends on the preservative; the metabisulfite-containing formulation may be less effective)
- Suppresses upper airway reflexes (facilitates LMA placement)
Other Effects
- No neuromuscular blockade — does not affect the twitch or EMG; however, acceptable intubating conditions have been reported with propofol alone
- Does NOT trigger malignant hyperthermia — safe in susceptible patients
- Anticonvulsant properties (also used in refractory status epilepticus)
- Mild antipruritic effects
Adverse Effects & Contraindications
| Effect | Details |
|---|---|
| Pain on injection | Very common (~70%); reduced by prior IV lidocaine, administering into a large vein, or using lipid-modified formulations |
| Propofol infusion syndrome (PRIS) | Rare but potentially fatal; associated with high-dose, prolonged infusions (>4 mg/kg/h for >48h) — metabolic acidosis, rhabdomyolysis, cardiac failure, lipemic plasma. Most often in critically ill children and adults on ICU sedation |
| Hypotension | Especially at induction; exaggerated in hypovolemic, elderly, or cardiac patients |
| Apnea | Occurs in most patients after induction bolus |
| Hypertriglyceridemia | With prolonged infusions due to lipid vehicle |
| Allergy | True anaphylaxis is rare; egg/soy allergy does not reliably predict propofol allergy (purified phospholipid fraction used) |
| Fetal neurotoxicity | FDA (December 2016) warning: concern about prolonged/repetitive exposure to general anesthetics including propofol affecting fetal brain development (based on animal studies) |
| Green discoloration of urine | Benign; due to phenolic metabolites |
Special Populations
- Pediatric patients: Higher induction dose required (2.5–3 mg/kg); PRIS risk is higher in critically ill children — avoid prolonged high-dose ICU sedation. FDA neurotoxicity warning applies to repeated/prolonged exposures in children <3 years
- Elderly: Reduced dose required; increased sensitivity and hemodynamic depression
- Pregnancy: Used for cesarean section induction; crosses placenta; neonates metabolize propofol; causes variability in neonatal elimination
- Liver/renal disease: Clearance modestly affected; extrahepatic metabolism provides some protection
Propofol vs. Newer Agents
Recent comparative evidence:
- Ciprofol vs. Propofol (2024 meta-analysis, PMID 38412619): Ciprofol shows comparable efficacy for induction with potentially better hemodynamic stability and less injection pain
- Remimazolam vs. Propofol in elderly (2024 meta-analysis, PMID 39069837): Remimazolam associated with less hypotension and respiratory depression in elderly patients; propofol still remains standard
Key Clinical Points Summary
- Most commonly used IV hypnotic worldwide for induction and maintenance of anesthesia and procedural sedation
- Rapid onset (30–60 sec) and rapid recovery (context-sensitive half-time <40 min even after 8-hour infusion) — ideal for ambulatory and day-case surgery
- Antiemetic properties distinguish it from other agents
- No analgesic effect — must combine with opioids or regional techniques
- Avoid in suspected PRIS — monitor triglycerides and urine in prolonged ICU use
- Hemodynamic vigilance required — especially in elderly, hypovolemic, or cardiac patients
— Miller's Anesthesia, 10th ed.
This is a shared conversation. Sign in to Orris to start your own chat.