Current Treatment protocol for NBIA
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of the current treatment protocol for Neurodegeneration with Brain Iron Accumulation (NBIA):
NBIA: Treatment Protocol
Background
NBIA is a heterogeneous group of inherited disorders characterized by pathological iron accumulation in the basal ganglia, presenting with progressive movement disorders (dystonia, parkinsonism, spasticity), neuropsychiatric symptoms, retinal degeneration, and cognitive decline. The most common subtype is Pantothenate Kinase-Associated Neurodegeneration (PKAN), accounting for ~50% of cases, caused by mutations in PANK2.
Other subtypes include:
- PLAN (PLA2G6-associated neurodegeneration)
- MPAN (mitochondrial membrane protein-associated neurodegeneration)
- BPAN (beta-propeller protein-associated neurodegeneration)
- CoPAN (CoA synthase protein-associated neurodegeneration)
- FAHN (fatty acid hydroxylase-associated neurodegeneration)
- Aceruloplasminemia, Neuroferritinopathy
"Eye of the Tiger" Sign (PKAN hallmark on MRI)
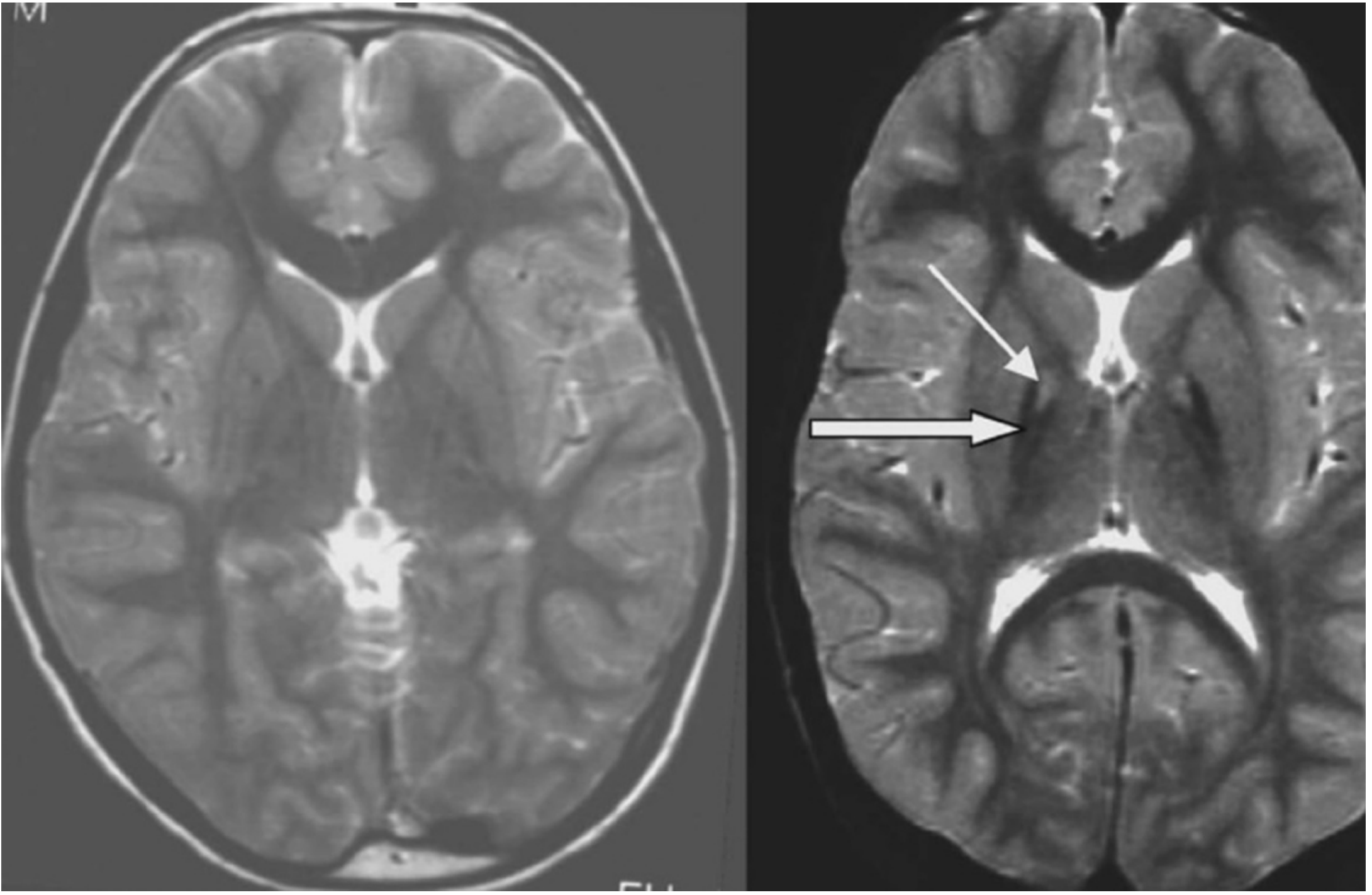
Left: normal control. Right: PANK2 mutation-positive NBIA patient showing hypointensity (thick arrow = iron deposition) with central hyperintensity (thin arrow = axonal spheroids) in the medial globus pallidus — the "eye of the tiger" sign.
Treatment: General Principles
There are currently no disease-modifying treatments proven to halt or reverse NBIA. Management is largely symptomatic, with several subtype-specific strategies under investigation or in limited use.
1. Iron Chelation Therapy
The most investigated disease-directed approach across NBIA subtypes.
- Deferiprone — an oral iron chelator that crosses the blood-brain barrier (BBB). This is the most studied agent in NBIA:
- In classic PKAN (early childhood onset): relatively ineffective at altering disease course.
- In atypical PKAN (teenage/adult onset, slower progression): may slow progression.
- Deferasirox and deferoxamine are other iron chelators (FDA-approved for systemic iron overload) but have limited evidence specific to NBIA; deferoxamine does not cross the BBB well.
- Iron binding has not been definitively established to slow progression in controlled trials.
"Treatment aimed at reducing iron levels using deferiprone, an iron chelator that crosses the blood-brain barrier, has been relatively ineffective in the classic phenotype but may slow progression in the atypical phenotype." — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
"There are no specific treatments; iron binding may help slow progression, but this has not been established." — Harrison's Principles of Internal Medicine 22E
2. Phosphopantothenate Replacement (PKAN-Specific)
- Fosmetpantotetheine (also written fosmetpantotentate / fosmetopentanate) — a prodrug designed to replenish phosphopantothenic acid, bypassing the defective PANK2 enzyme and restoring CoA synthesis.
- A Phase 3 trial (FORT trial) of this agent did not improve function in PKAN patients.
- Earlier case reports and pilot studies suggested possible regression of symptoms after several months.
"A phosphophatonate replacement trial using fosmetpantotentate did not improve function in PKAN patients." — Kaplan & Sadock's Comprehensive Textbook of Psychiatry
"Iron chelation with deferiprone and fosmetopentanate, a phosphopantothenic acid prodrug which aims to replenish phosphopantothenic acid, have been reported to result in regression of symptoms after several months of treatment." — Bradley and Daroff's Neurology in Clinical Practice
3. Symptomatic / Supportive Treatment
These are the mainstays of day-to-day management and are applied across all NBIA subtypes:
| Symptom | Treatment Options |
|---|---|
| Dystonia | Oral medications (trihexyphenidyl, baclofen, clonazepam, levodopa trial); intrathecal baclofen for severe generalized dystonia |
| Parkinsonism | Levodopa — some subtypes (PLAN adult-onset, MPAN, BPAN) show initial levodopa-responsiveness, though benefit often wanes |
| Spasticity | Baclofen (oral or intrathecal), tizanidine |
| Neuropsychiatric symptoms | Standard psychiatric pharmacotherapy (antidepressants, antipsychotics as appropriate) |
| Seizures | Antiepileptic drugs |
| Retinal degeneration | Ophthalmology follow-up; no disease-modifying therapy |
| Dysarthria/Dysphagia | Speech therapy, PEG tube for severe dysphagia |
| Deep Brain Stimulation (DBS) | Globus pallidus internus (GPi) DBS has been used for refractory dystonia in PKAN; results are variable but can provide meaningful improvement in some patients |
4. Subtype-Specific Notes
| Subtype | Key Treatment Consideration |
|---|---|
| PKAN | Iron chelation (deferiprone); fosmetpantetheine (failed Phase 3); DBS for dystonia |
| PLAN (adult-onset) | Levodopa-responsive dystonia-parkinsonism — trial of levodopa warranted |
| MPAN | May present as juvenile-onset levodopa-responsive parkinsonism; levodopa trial warranted |
| Aceruloplasminemia | Iron chelation + fresh frozen plasma (source of ceruloplasmin); some benefit reported |
| Neuroferritinopathy | Iron chelation (deferiprone has been used); supportive care |
5. Emerging / Investigational Approaches
- Gene therapy and molecular targeting of specific NBIA gene mutations are active research areas but not yet clinical standard of care.
- CoA supplementation strategies for CoA-related subtypes (CoPAN) are under investigation.
Summary
| Approach | Status |
|---|---|
| Deferiprone (iron chelation) | Used in practice; benefit modest, mainly in atypical/adult PKAN and neuroferritinopathy |
| Fosmetpantetheine (CoA replenishment) | Phase 3 failed; not in current use |
| Levodopa | Beneficial in PLAN, MPAN, BPAN subtypes with parkinsonism |
| DBS (GPi) | Used for refractory dystonia; variable benefit |
| Symptomatic pharmacotherapy | Core management for all subtypes |
| Disease-modifying therapy | None established |
Sources: Bradley and Daroff's Neurology in Clinical Practice; Harrison's Principles of Internal Medicine 22E; Kaplan & Sadock's Comprehensive Textbook of Psychiatry
This is a shared conversation. Sign in to Orris to start your own chat.