Give me a complete summary of G6PD deficiency involving only the pathology aspects relevant for NEET PG 2026. It should be a summary which will help me to revise in the end by reading it. And it should cover almost 100% of possible pathology questions that is the clinical MCQs which can be asked from G6PD deficiency in the pathology aspect for NEET PG.
G6PD Deficiency — Complete Pathology Summary for NEET PG 2026
1. BASICS (HIGH-YIELD IDENTIFIERS)
| Feature | Detail |
|---|---|
| Type of disorder | RBC enzyme defect (enzymopathy) |
| Inheritance | X-linked recessive |
| Affected sex | Predominantly males (hemizygous) |
| Most common enzymopathy | Most common disease-producing enzyme deficiency in humans |
| Global burden | >400 million affected worldwide |
| Gene locus | X chromosome |
| Enzyme defect | Glucose-6-phosphate dehydrogenase (G6PD) |
| Pathway affected | Pentose phosphate pathway (Hexose monophosphate shunt — HMP shunt) |
NEET MCQ trap: Females are usually carriers (unaffected), but those with "unfavorable lyonization" (large proportion of G6PD-deficient X active in RBCs) can be symptomatic.
2. PATHOGENESIS — THE CORE MECHANISM
G6PD deficiency
↓
↓ NADPH production (HMP shunt is the ONLY source of NADPH in RBCs)
↓
↓ Reduced glutathione (GSH)
↓
Cannot neutralize H₂O₂ and oxidant stress
↓
Oxidation of Hb sulfhydryl groups
↓
Hb denatures → forms HEINZ BODIES (insoluble precipitates)
↓
Heinz bodies attach to RBC membrane → membrane damage
↓
Intravascular hemolysis (severe damage) OR
Splenic macrophages "pluck out" Heinz bodies → BITE CELLS (degmacytes)
↓
Bite cells trapped in splenic cords → extravascular hemolysis
- The pentose phosphate pathway is the only way RBCs generate NADPH (no mitochondria)
- RBCs have no nucleus or ribosomes — cannot synthesize new enzyme to replace deficient/degraded G6PD
3. TRIGGERS OF HEMOLYSIS (PRECIPITATING FACTORS)
Drugs (Oxidants) — "4 As" Mnemonic
| Category | Examples |
|---|---|
| Antimalarials | Primaquine, chloroquine |
| Antibiotics (Sulfa) | Sulfonamides, Dapsone, Nitrofurantoin |
| Analgesics/Antipyretics | Aspirin (large doses), Phenacetin |
| Others | Vitamin K derivatives |
Classic NEET MCQ: Primaquine → hemolysis in G6PD deficiency. Dapsone (used in leprosy/dermatitis herpetiformis) → severe hemolysis, especially in Mediterranean variant.
Foods
- Fava beans (broad beans) → "Favism"
- Favism is seen particularly in the Mediterranean variant
- All patients with favism have G6PD deficiency, but NOT all G6PD-deficient patients have favism
Infections (Most Common Trigger in Practice)
- Viral hepatitis, pneumonia, typhoid fever
- Mechanism: activated leukocytes produce oxygen free radicals → diffuse into RBCs → oxidative damage
- Infection is the most common precipitant of hemolysis in G6PD deficiency
Neonatal period
- Neonatal jaundice (NNJ) appearing 1–4 days after birth with unconjugated hyperbilirubinemia
4. MORPHOLOGY / PERIPHERAL SMEAR FINDINGS
Key Peripheral Smear Features:
| Finding | Significance |
|---|---|
| Heinz bodies | Denatured Hb precipitates; seen with crystal violet or brilliant cresyl blue (supravital stain); NOT seen on routine H&E or Giemsa |
| Bite cells (Degmacytes) | Formed when splenic macrophages pluck out Heinz bodies; seen on routine smear |
| Blister cells | Variant of bite cells |
| Spherocytes | From membrane surface area loss after Heinz body removal |
| Reticulocytosis | In recovery phase; heralds the end of hemolytic episode |
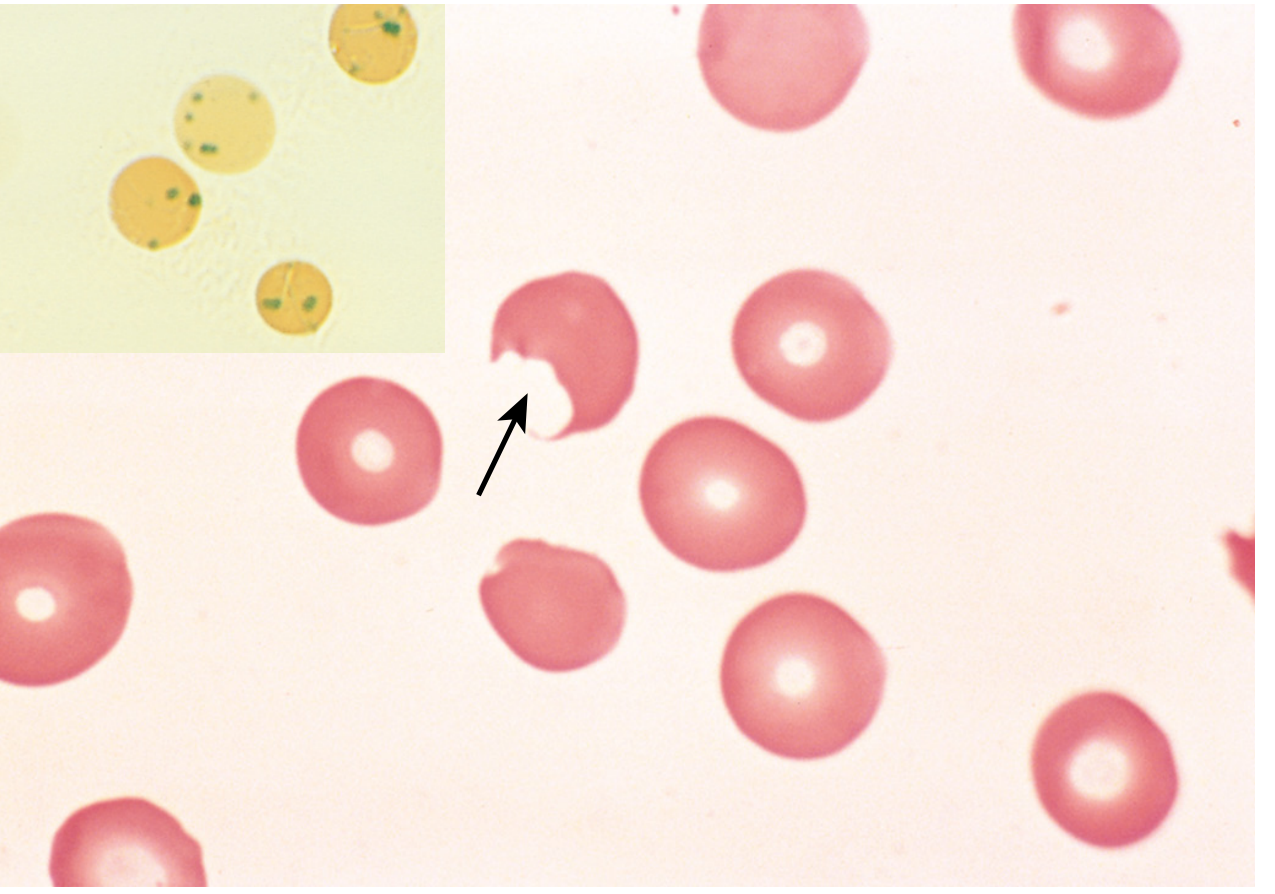
NEET MCQ: Heinz bodies are stained by crystal violet or brilliant cresyl blue (supravital stains). They are NOT visible on routine Leishman/Giemsa stain.
5. CLASSIFICATION OF G6PD VARIANTS (WHO Classes)
| Class | Clinical Features | Residual Enzyme Activity | Notes |
|---|---|---|---|
| I | Chronic Nonspherocytic Hemolytic Anemia (CNSHA) — without oxidant trigger | <10% | Rare; most severe |
| II | Severe intermittent hemolysis (acute) | <10% | G6PD Mediterranean — prototype; *most important for NEET |
| III | Moderate hemolysis only on oxidant stress | 10–60% | G6PD A− (African variant) — prototype; *most common |
| IV | No clinical symptoms | >60% | No disease |
| V | G6PD overproduction | (elevated) | No disease |
NEET MCQ: Class I and II → enzyme activity <10%. Class II and III are the most clinically common. G6PD A− (Class III) is the most prevalent worldwide; G6PD Mediterranean (Class II) is more severe.
6. VARIANTS — HIGH-YIELD COMPARISON
| Feature | G6PD A− (African) | G6PD Mediterranean |
|---|---|---|
| WHO Class | III (Moderate) | II (Severe) |
| Enzyme stability | Mildly unstable | Severely unstable |
| G6PD activity in young RBCs | Near normal | Very low even in young cells |
| Hemolysis pattern | Self-limiting — only old RBCs lyse; young reticulocytes survive | More severe, prolonged |
| Fava bean sensitivity | Less common | Yes — prominent |
| Dapsone sensitivity | Mild | Severe |
| Geography | Sub-Saharan Africa | Mediterranean, Middle East, India |
Why self-limiting in G6PD A−? Only older RBCs have critically low G6PD (the enzyme degrades faster than normal but is initially adequate in young cells). As the hemolytic episode proceeds, older cells are destroyed, leaving only young G6PD-replete reticulocytes — hemolysis ceases even if the drug is continued.
7. CLINICAL FEATURES
Acute Hemolytic Anemia (AHA)
- Onset: 2–3 days after exposure to trigger
- Features: anemia, hemoglobinemia, hemoglobinuria (dark/cola-colored urine), jaundice
- Largely intravascular hemolysis during acute phase
Neonatal Jaundice (NNJ)
- Presents 1–4 days after birth
- Unconjugated hyperbilirubinemia
- Can be severe; management same as other causes of NNJ (phototherapy, exchange transfusion)
Chronic Nonspherocytic Hemolytic Anemia (CNSHA) — Class I only
- Persistent low-grade hemolysis without identifiable trigger
- Rare
- Splenomegaly and cholelithiasis may develop (unlike episodic G6PD — no splenomegaly/cholelithiasis in typical episodic cases)
Favism
- Acute severe hemolysis after fava bean ingestion
- Predominantly Mediterranean variant
- Not all G6PD-deficient persons develop favism, but all favism patients have G6PD deficiency
8. LABORATORY DIAGNOSIS
| Test | Finding |
|---|---|
| CBC | Normocytic/normochromic anemia; ↑ reticulocytes (recovery phase) |
| Peripheral smear | Bite cells, Heinz bodies (supravital stain), spherocytes |
| LDH | Elevated (hemolysis marker) |
| Indirect bilirubin | Elevated |
| Haptoglobin | Decreased (intravascular hemolysis) |
| Urinalysis | Hemoglobinuria (dark/cola urine) |
| Definitive test | G6PD enzyme assay (spectrophotometric) — quantitative |
| Screening test | Fluorescent spot test (NADPH fluorescence) or Methemoglobin reduction test |
| Heinz body stain | Crystal violet or Brilliant cresyl blue (supravital stains) |
NEET MCQ Trap: G6PD levels may appear falsely normal immediately after a hemolytic episode because the older, G6PD-deficient cells have been destroyed and the remaining cells are younger reticulocytes with relatively normal G6PD. Best time to test: 2–3 months after the episode.
9. GENETICS
- X-linked recessive — most females are carriers, males are affected
-
400 mutations identified — most are missense point mutations
- No large deletions or frameshifts identified → complete absence of G6PD is likely lethal
- Active G6PD enzyme exists as a homodimer or tetramer
- Heterozygous females: two populations of RBCs (normal + deficient) due to lyonization (X-inactivation)
- "Unfavorable lyonization" → symptomatic female
10. EVOLUTIONARY / EPIDEMIOLOGICAL ASPECT
- Protective against Plasmodium falciparum malaria — high prevalence in malaria-endemic regions (Africa, Mediterranean, Middle East, India)
- Mechanism: parasite-induced oxidant stress causes premature lysis of infected RBCs → limits parasitemia
- Distribution of G6PD deficiency parallels the worldwide distribution of falciparum malaria ("malaria map")
11. TREATMENT
| Scenario | Management |
|---|---|
| Acute hemolysis | Remove trigger (stop drug/avoid fava bean); supportive; most self-limiting |
| Severe acute anemia | Blood transfusion (emergency in children) |
| Acute renal failure | Hemodialysis (rare; full recovery usually) |
| Neonatal jaundice | Phototherapy / exchange transfusion (same as other NNJ causes) |
| CNSHA | Folic acid supplements; avoid hemolytic drugs; transfusion for exacerbations |
| CNSHA (severe) | Splenectomy — beneficial in severe CNSHA (unlike typical episodic G6PD, where it has no role) |
| Prevention | Screen before prescribing primaquine/dapsone; avoid fava beans |
12. HIGH-YIELD NEET PG MCQ TRIGGERS — QUICK RECALL
| Question Theme | Answer |
|---|---|
| Most common enzymopathy worldwide | G6PD deficiency |
| Inheritance pattern | X-linked recessive |
| Why RBCs uniquely vulnerable | HMP shunt = only source of NADPH in RBCs; no new enzyme synthesis possible |
| Stain for Heinz bodies | Crystal violet / Brilliant cresyl blue (supravital) |
| Bite cells formed by | Splenic macrophages plucking Heinz bodies |
| Drug → classic G6PD hemolysis | Primaquine |
| Food → hemolysis in Mediterranean variant | Fava beans |
| Most common trigger of G6PD hemolysis overall | Infection |
| Self-limiting hemolysis variant | G6PD A− (African type, Class III) |
| More severe variant | G6PD Mediterranean (Class II) |
| CNSHA without trigger | Class I G6PD deficiency |
| Timing of hemolysis after drug | 2–3 days after exposure |
| Why hemolysis self-limiting in G6PD A− | Only old RBCs lyse; young reticulocytes have adequate G6PD |
| Favism — relationship | All favism = G6PD deficiency; NOT all G6PD = favism |
| Best time for G6PD assay | 2–3 months after hemolytic episode |
| Enzyme activity in Class I & II | <10% |
| Enzyme activity in Class III | 10–60% |
| G6PD deficiency and malaria | Protective against Plasmodium falciparum |
| Heinz bodies = | Denatured Hb precipitates attached to RBC membrane |
| Type of hemolysis | Primarily intravascular (acute); also extravascular (splenic) |
| Splenomegaly/cholelithiasis in G6PD | Absent in episodic form; present only in Class I CNSHA |
| Dapsone — severe hemolysis in | Mediterranean variant |
13. DIFFERENTIATING G6PD FROM OTHER HEMOLYTIC ANEMIAS
| Feature | G6PD | Hereditary Spherocytosis | PK Deficiency | PNH |
|---|---|---|---|---|
| Trigger needed | Yes (usually) | No | No | Complement activation |
| Peripheral smear | Bite cells, Heinz bodies | Spherocytes | Echinocytes | Normal/spherocytes |
| Inheritance | X-linked | AD (mostly) | AR | Acquired (PIGA mutation) |
| Splenomegaly | Only in CNSHA | Yes | Yes | No |
| Hemoglobinuria | Yes (acute) | No | No | Yes (nocturnal) |
| Osmotic fragility | Normal | Increased | Normal | Normal |
| Coombs test | Negative | Negative | Negative | Negative |
Now I want you to convert the above info on G6PD deficiency into an Anki card with the front portion showing G6PD deficiency and the back portion showing the rest, all the details. And in the back portion, there should be proper separation of headings, space between them, so that they can be easily read and it should be legible, it should not be crowded. And it should be a single card. I also want you to make it into an import-ready format so that I can copy the entire text, paste it into a text generation app, and then import it directly to Anki card.
.txt), then import into Anki using File → Import with the separator set to Tab.G6PD Deficiency <h2 style="color:#2c3e50;">G6PD DEFICIENCY — Complete Pathology Summary</h2><br><hr><h3 style="color:#1a5276;">1. BASICS</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Feature</th><th>Detail</th></tr><tr><td>Type</td><td>RBC enzyme defect (enzymopathy)</td></tr><tr><td>Inheritance</td><td><b>X-linked recessive</b></td></tr><tr><td>Most common in</td><td>Males (hemizygous); females usually carriers</td></tr><tr><td>Significance</td><td><b>Most common disease-producing enzyme deficiency in humans</b></td></tr><tr><td>Global burden</td><td>>400 million affected worldwide</td></tr><tr><td>Gene locus</td><td>X chromosome</td></tr><tr><td>Pathway affected</td><td><b>Pentose phosphate pathway (HMP shunt)</b></td></tr></table><br><hr><h3 style="color:#1a5276;">2. PATHOGENESIS CHAIN</h3><p>G6PD deficiency → ↓ NADPH (HMP shunt = <b>only source of NADPH in RBCs</b>) → ↓ Reduced glutathione (GSH) → Cannot neutralize H₂O₂ → Oxidation of Hb sulfhydryl groups → Hb denatures → <b>Heinz bodies</b> (insoluble precipitates on RBC membrane) → Membrane damage → <b>Intravascular hemolysis</b> (severe) OR splenic macrophages pluck out Heinz bodies → <b>Bite cells (Degmacytes)</b> → Extravascular hemolysis</p><p><b>Why RBCs uniquely vulnerable:</b> HMP shunt is the ONLY way RBCs make NADPH. RBCs have no nucleus/ribosomes → cannot synthesize new enzyme.</p><br><hr><h3 style="color:#1a5276;">3. TRIGGERS OF HEMOLYSIS</h3><p><b>Drugs (Oxidants):</b></p><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Category</th><th>Examples</th></tr><tr><td>Antimalarials</td><td>Primaquine, Chloroquine</td></tr><tr><td>Antibiotics/Sulfa</td><td>Sulfonamides, <b>Dapsone</b>, Nitrofurantoin</td></tr><tr><td>Analgesics</td><td>Aspirin (large doses), Phenacetin</td></tr><tr><td>Others</td><td>Vitamin K derivatives</td></tr></table><br><p><b>Foods:</b> Fava beans → <b>Favism</b> (especially Mediterranean variant)</p><p><b>Infection (Most Common Trigger):</b> Viral hepatitis, pneumonia, typhoid fever — activated leukocytes produce free radicals that diffuse into RBCs</p><p><b>Neonatal:</b> Neonatal jaundice 1–4 days after birth (unconjugated hyperbilirubinemia)</p><br><hr><h3 style="color:#1a5276;">4. PERIPHERAL SMEAR FINDINGS</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Finding</th><th>Notes</th></tr><tr><td><b>Heinz bodies</b></td><td>Denatured Hb precipitates; seen with <b>Crystal violet</b> or <b>Brilliant cresyl blue</b> (supravital stain); NOT on routine Giemsa/Leishman</td></tr><tr><td><b>Bite cells (Degmacytes)</b></td><td>Formed after splenic macrophages pluck out Heinz bodies; seen on routine smear</td></tr><tr><td>Spherocytes</td><td>From membrane surface area loss</td></tr><tr><td>Reticulocytosis</td><td>Recovery phase — heralds end of hemolytic episode</td></tr></table><br><hr><h3 style="color:#1a5276;">5. WHO CLASSIFICATION OF G6PD VARIANTS</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Class</th><th>Clinical Features</th><th>Residual Enzyme Activity</th></tr><tr><td><b>I</b></td><td>CNSHA — chronic, no trigger needed (rarest, most severe)</td><td><10%</td></tr><tr><td><b>II*</b></td><td>Severe acute hemolysis — <b>G6PD Mediterranean</b> prototype</td><td><10%</td></tr><tr><td><b>III*</b></td><td>Moderate — <b>G6PD A− (African)</b> prototype; most common worldwide</td><td>10–60%</td></tr><tr><td><b>IV</b></td><td>No symptoms</td><td>>60%</td></tr><tr><td><b>V</b></td><td>G6PD <i>overproduction</i> — no disease</td><td>Elevated</td></tr></table><p><i>* = most clinically common classes</i></p><br><hr><h3 style="color:#1a5276;">6. VARIANT COMPARISON: G6PD A− vs MEDITERRANEAN</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Feature</th><th>G6PD A− (African)</th><th>G6PD Mediterranean</th></tr><tr><td>Class</td><td>III</td><td>II</td></tr><tr><td>Severity</td><td>Moderate</td><td>Severe</td></tr><tr><td>Hemolysis pattern</td><td><b>Self-limiting</b> (only old RBCs lyse)</td><td>Prolonged, more severe</td></tr><tr><td>Fava bean sensitivity</td><td>Mild/absent</td><td><b>Prominent</b></td></tr><tr><td>Dapsone sensitivity</td><td>Mild</td><td><b>Severe</b></td></tr><tr><td>Young RBC G6PD activity</td><td>Near normal</td><td>Very low even in young cells</td></tr></table><p><b>Why self-limiting in G6PD A−?</b> Only old RBCs (critically low G6PD) lyse. Young reticulocytes have adequate G6PD → hemolysis ceases even if drug is continued.</p><br><hr><h3 style="color:#1a5276;">7. CLINICAL FEATURES</h3><p><b>Acute Hemolytic Anemia:</b> Onset 2–3 days after exposure; anemia, hemoglobinemia, hemoglobinuria (dark/cola urine), jaundice</p><p><b>Neonatal Jaundice:</b> 1–4 days after birth; unconjugated hyperbilirubinemia; can be severe</p><p><b>CNSHA (Class I only):</b> Persistent hemolysis without trigger; splenomegaly + cholelithiasis may develop</p><p><b>Favism:</b> Acute severe hemolysis after fava beans. All favism = G6PD deficiency. Not all G6PD deficiency = favism.</p><p><b>Note:</b> Splenomegaly and cholelithiasis are <b>ABSENT</b> in typical episodic G6PD; present only in Class I CNSHA.</p><br><hr><h3 style="color:#1a5276;">8. LABORATORY DIAGNOSIS</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Test</th><th>Finding</th></tr><tr><td>CBC</td><td>Normocytic/normochromic anemia; ↑ reticulocytes</td></tr><tr><td>Peripheral smear</td><td>Bite cells, Heinz bodies (supravital stain)</td></tr><tr><td>LDH</td><td>↑ (hemolysis marker)</td></tr><tr><td>Indirect bilirubin</td><td>↑</td></tr><tr><td>Haptoglobin</td><td>↓ (intravascular hemolysis)</td></tr><tr><td>Urinalysis</td><td>Hemoglobinuria</td></tr><tr><td><b>Definitive test</b></td><td><b>G6PD enzyme assay (spectrophotometric)</b></td></tr><tr><td>Screening test</td><td>Fluorescent spot test (NADPH fluorescence) or Methemoglobin reduction test</td></tr><tr><td>Heinz body stain</td><td><b>Crystal violet</b> or <b>Brilliant cresyl blue</b></td></tr></table><p><b>⚠ MCQ Trap:</b> G6PD levels may appear falsely NORMAL right after an episode (old deficient cells destroyed, young reticulocytes remain). Best time to test: <b>2–3 months after episode.</b></p><br><hr><h3 style="color:#1a5276;">9. GENETICS</h3><ul><li>X-linked recessive; males affected, females usually carriers</li><li>>400 mutations; mostly <b>missense point mutations</b></li><li>No large deletions/frameshifts → complete G6PD absence likely <b>lethal</b></li><li>Active enzyme is a <b>homodimer or tetramer</b></li><li>Heterozygous females: two RBC populations due to <b>lyonization (X-inactivation)</b></li><li>"Unfavorable lyonization" → symptomatic female</li></ul><br><hr><h3 style="color:#1a5276;">10. MALARIA CONNECTION</h3><p>G6PD deficiency is <b>protective against Plasmodium falciparum malaria.</b> Distribution of G6PD deficiency parallels the malaria-endemic belt (Africa, Mediterranean, Middle East, India). Parasite-induced oxidant stress causes premature RBC lysis → limits parasitemia.</p><br><hr><h3 style="color:#1a5276;">11. TREATMENT</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Scenario</th><th>Management</th></tr><tr><td>Acute hemolysis</td><td>Remove trigger; mostly self-limiting; supportive</td></tr><tr><td>Severe acute anemia</td><td><b>Blood transfusion</b></td></tr><tr><td>Acute renal failure</td><td>Hemodialysis (rare; full recovery usual)</td></tr><tr><td>Neonatal jaundice</td><td>Phototherapy / exchange transfusion</td></tr><tr><td>CNSHA</td><td>Folic acid; avoid hemolytic drugs; transfusion for exacerbations</td></tr><tr><td>Severe CNSHA</td><td><b>Splenectomy</b> (beneficial in CNSHA only)</td></tr><tr><td>Prevention</td><td>Screen before prescribing primaquine/dapsone; avoid fava beans</td></tr></table><br><hr><h3 style="color:#1a5276;">12. HIGH-YIELD MCQ RAPID RECALL</h3><table border="1" cellpadding="5" cellspacing="0" style="border-collapse:collapse;width:100%;"><tr><th>Question Theme</th><th>Answer</th></tr><tr><td>Most common enzymopathy</td><td>G6PD deficiency</td></tr><tr><td>Stain for Heinz bodies</td><td>Crystal violet / Brilliant cresyl blue (supravital)</td></tr><tr><td>Bite cells formed by</td><td>Splenic macrophages plucking Heinz bodies</td></tr><tr><td>Classic drug trigger</td><td><b>Primaquine</b></td></tr><tr><td>Classic food trigger</td><td><b>Fava beans</b></td></tr><tr><td>Most common trigger overall</td><td><b>Infection</b></td></tr><tr><td>Self-limiting variant</td><td>G6PD A− (African, Class III)</td></tr><tr><td>More severe variant</td><td>G6PD Mediterranean (Class II)</td></tr><tr><td>CNSHA without trigger</td><td>Class I</td></tr><tr><td>Timing of hemolysis after drug</td><td>2–3 days after exposure</td></tr><tr><td>Favism relationship</td><td>All favism = G6PD deficiency; NOT vice versa</td></tr><tr><td>Best time for G6PD assay</td><td>2–3 months after episode</td></tr><tr><td>Class I & II enzyme activity</td><td><10%</td></tr><tr><td>Class III enzyme activity</td><td>10–60%</td></tr><tr><td>Malaria protection</td><td>Against P. falciparum</td></tr><tr><td>Splenomegaly/cholelithiasis</td><td>Absent in episodic form; only in Class I CNSHA</td></tr><tr><td>Type of hemolysis (acute)</td><td>Primarily intravascular</td></tr><tr><td>Dapsone — severe hemolysis in</td><td>Mediterranean variant</td></tr></table>
- Copy the entire text between the code block delimiters (the single line starting with
G6PD Deficiencyand ending after the last</table>) - Paste it into a plain text editor (Notepad, TextEdit) and save as
g6pd.txt(encoding: UTF-8) - In Anki: File → Import
- Select your
.txtfile - Set Field separator to Tab
- Map Field 1 → Front, Field 2 → Back
- Tick "Allow HTML in fields"
- Click Import
Retry
.txt file, then import into Anki.G6PD Deficiency <div style="font-family:Arial,sans-serif;font-size:14px;line-height:1.6;max-width:900px;margin:auto;padding:10px;"><h2 style="color:#1a5276;border-bottom:2px solid #1a5276;padding-bottom:5px;">G6PD DEFICIENCY — Pathology Summary (NEET PG)</h2><h3 style="color:#1a5276;margin-top:20px;">1. BASICS</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:15px;"><tr style="background:#d6eaf8;"><th>Feature</th><th>Detail</th></tr><tr><td>Type of disorder</td><td>RBC enzyme defect (enzymopathy)</td></tr><tr><td>Inheritance</td><td><b>X-linked recessive</b></td></tr><tr><td>Who is affected</td><td>Predominantly <b>males</b> (hemizygous); females usually carriers</td></tr><tr><td>Global significance</td><td><b>Most common disease-producing enzyme deficiency in humans</b></td></tr><tr><td>Burden</td><td>>400 million affected worldwide</td></tr><tr><td>Gene locus</td><td>X chromosome</td></tr><tr><td>Pathway affected</td><td><b>Pentose Phosphate Pathway / HMP shunt</b></td></tr></table><h3 style="color:#1a5276;margin-top:20px;">2. PATHOGENESIS CHAIN</h3><div style="background:#eaf4fb;padding:10px;border-left:4px solid #1a5276;margin-bottom:15px;"><p style="margin:4px 0;">G6PD deficiency</p><p style="margin:4px 0;">↓ NADPH (HMP shunt = <b>only source of NADPH in RBCs</b>)</p><p style="margin:4px 0;">↓ Reduced glutathione (GSH)</p><p style="margin:4px 0;">Cannot neutralize H₂O₂ and oxidant stress</p><p style="margin:4px 0;">Hb sulfhydryl groups oxidized → Hb denatures → <b>Heinz bodies</b> form</p><p style="margin:4px 0;">Heinz bodies damage RBC membrane</p><p style="margin:4px 0;">→ <b>Intravascular hemolysis</b> (severe damage)</p><p style="margin:4px 0;">→ Splenic macrophages pluck out Heinz bodies → <b>Bite cells (Degmacytes)</b> → Extravascular hemolysis</p></div><p><b>Why RBCs are uniquely vulnerable:</b> HMP shunt is the ONLY pathway to generate NADPH in RBCs (no mitochondria). RBCs have no nucleus or ribosomes — they cannot synthesize new enzyme.</p><h3 style="color:#1a5276;margin-top:20px;">3. TRIGGERS OF HEMOLYSIS</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:10px;"><tr style="background:#d6eaf8;"><th>Category</th><th>Examples</th></tr><tr><td>Antimalarials</td><td>Primaquine, Chloroquine</td></tr><tr><td>Antibiotics/Sulfa drugs</td><td>Sulfonamides, <b>Dapsone</b>, Nitrofurantoin</td></tr><tr><td>Analgesics/Antipyretics</td><td>Aspirin (large doses), Phenacetin</td></tr><tr><td>Others</td><td>Vitamin K derivatives</td></tr><tr><td>Foods</td><td><b>Fava beans → Favism</b> (especially Mediterranean variant)</td></tr><tr><td><b>Most common trigger</b></td><td><b>Infection</b> — viral hepatitis, pneumonia, typhoid fever</td></tr><tr><td>Neonatal</td><td>Neonatal jaundice appearing <b>1–4 days after birth</b></td></tr></table><p style="background:#fef9e7;padding:8px;border-left:4px solid #f39c12;"><b>Note:</b> All favism patients have G6PD deficiency. NOT all G6PD-deficient patients develop favism.</p><h3 style="color:#1a5276;margin-top:20px;">4. PERIPHERAL SMEAR FINDINGS</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:15px;"><tr style="background:#d6eaf8;"><th>Finding</th><th>Notes</th></tr><tr><td><b>Heinz bodies</b></td><td>Denatured Hb precipitates attached to RBC membrane. Stained with <b>Crystal violet</b> or <b>Brilliant cresyl blue</b> (supravital stains). <b>NOT visible</b> on routine Leishman/Giemsa stain.</td></tr><tr><td><b>Bite cells (Degmacytes)</b></td><td>Formed when splenic macrophages pluck out Heinz bodies. Visible on routine smear.</td></tr><tr><td>Spherocytes</td><td>From membrane surface area loss after Heinz body removal</td></tr><tr><td>Reticulocytosis</td><td>Recovery phase — heralds end of hemolytic episode</td></tr></table><h3 style="color:#1a5276;margin-top:20px;">5. WHO CLASSIFICATION OF G6PD VARIANTS</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:10px;"><tr style="background:#d6eaf8;"><th>Class</th><th>Clinical Features</th><th>Residual Enzyme Activity</th></tr><tr><td><b>I</b></td><td>CNSHA — chronic hemolysis without any trigger (rarest, most severe)</td><td><10%</td></tr><tr><td><b>II ★</b></td><td>Severe acute hemolytic anemia — <b>G6PD Mediterranean</b> prototype</td><td><10%</td></tr><tr><td><b>III ★</b></td><td>Moderate hemolysis on oxidant stress — <b>G6PD A− (African)</b> prototype; most common worldwide</td><td>10–60%</td></tr><tr><td><b>IV</b></td><td>No clinical symptoms</td><td>>60%</td></tr><tr><td><b>V</b></td><td>G6PD overproduction — no disease</td><td>Elevated</td></tr></table><p><i>★ = most clinically common classes</i></p><h3 style="color:#1a5276;margin-top:20px;">6. VARIANT COMPARISON: G6PD A− vs MEDITERRANEAN</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:10px;"><tr style="background:#d6eaf8;"><th>Feature</th><th>G6PD A− (African)</th><th>G6PD Mediterranean</th></tr><tr><td>WHO Class</td><td>III</td><td>II</td></tr><tr><td>Severity</td><td>Moderate</td><td>Severe</td></tr><tr><td>Enzyme in young RBCs</td><td>Near normal</td><td>Very low even in young cells</td></tr><tr><td>Hemolysis pattern</td><td><b>Self-limiting</b> — only old RBCs lyse</td><td>Prolonged and more severe</td></tr><tr><td>Fava bean sensitivity</td><td>Mild/absent</td><td><b>Prominent</b></td></tr><tr><td>Dapsone sensitivity</td><td>Mild</td><td><b>Severe</b></td></tr></table><p style="background:#eaf4fb;padding:8px;border-left:4px solid #1a5276;"><b>Why self-limiting in G6PD A−?</b> Only older RBCs (with critically low G6PD) are destroyed. Young reticulocytes retain adequate G6PD activity → hemolysis ceases even if the drug exposure continues.</p><h3 style="color:#1a5276;margin-top:20px;">7. CLINICAL FEATURES</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:15px;"><tr style="background:#d6eaf8;"><th>Presentation</th><th>Details</th></tr><tr><td>Acute Hemolytic Anemia</td><td>Onset <b>2–3 days</b> after trigger; anemia, hemoglobinemia, <b>hemoglobinuria (dark/cola urine)</b>, jaundice; primarily intravascular</td></tr><tr><td>Neonatal Jaundice</td><td>1–4 days after birth; unconjugated hyperbilirubinemia; can be severe</td></tr><tr><td>CNSHA (Class I only)</td><td>Persistent hemolysis without trigger; splenomegaly + cholelithiasis may develop</td></tr><tr><td>Favism</td><td>Acute severe hemolysis after fava beans; predominantly Mediterranean variant</td></tr></table><p style="background:#fef9e7;padding:8px;border-left:4px solid #f39c12;"><b>Key:</b> Splenomegaly and cholelithiasis are <b>ABSENT</b> in typical episodic G6PD. Present only in Class I CNSHA.</p><h3 style="color:#1a5276;margin-top:20px;">8. LABORATORY DIAGNOSIS</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:10px;"><tr style="background:#d6eaf8;"><th>Test</th><th>Finding</th></tr><tr><td>CBC</td><td>Normocytic/normochromic anemia; ↑ reticulocytes</td></tr><tr><td>Peripheral smear</td><td>Bite cells; Heinz bodies on supravital stain</td></tr><tr><td>LDH</td><td>↑ (hemolysis marker)</td></tr><tr><td>Indirect bilirubin</td><td>↑</td></tr><tr><td>Haptoglobin</td><td>↓ (intravascular hemolysis)</td></tr><tr><td>Urine</td><td>Hemoglobinuria</td></tr><tr><td><b>Definitive test</b></td><td><b>G6PD enzyme assay (spectrophotometric)</b></td></tr><tr><td>Screening test</td><td>Fluorescent spot test (NADPH fluorescence) OR Methemoglobin reduction test</td></tr></table><p style="background:#fadbd8;padding:8px;border-left:4px solid #c0392b;"><b>⚠ MCQ Trap:</b> G6PD levels may be falsely NORMAL right after an acute episode — old deficient RBCs are destroyed, leaving only young reticulocytes with relatively normal G6PD. <b>Best time to test: 2–3 months after the episode.</b></p><h3 style="color:#1a5276;margin-top:20px;">9. GENETICS</h3><ul style="line-height:2;"><li>X-linked recessive — males affected, females usually asymptomatic carriers</li><li>>400 mutations identified; mostly <b>missense point mutations</b></li><li>No large deletions or frameshifts → complete absence of G6PD is likely <b>lethal</b></li><li>Active enzyme exists as a <b>homodimer or tetramer</b></li><li>Heterozygous females: two RBC populations due to <b>lyonization (X-inactivation)</b></li><li>"Unfavorable lyonization" → symptomatic female</li></ul><h3 style="color:#1a5276;margin-top:20px;">10. MALARIA CONNECTION</h3><p style="background:#eaf4fb;padding:10px;border-left:4px solid #1a5276;">G6PD deficiency is <b>protective against Plasmodium falciparum malaria.</b> Distribution of G6PD deficiency geographically parallels the malaria-endemic belt (Africa, Mediterranean, Middle East, India). Parasite-induced oxidant stress → premature lysis of infected RBCs → limits parasitemia.</p><h3 style="color:#1a5276;margin-top:20px;">11. TREATMENT</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:15px;"><tr style="background:#d6eaf8;"><th>Scenario</th><th>Management</th></tr><tr><td>Acute hemolysis</td><td>Remove trigger; supportive; mostly self-limiting</td></tr><tr><td>Severe acute anemia</td><td><b>Blood transfusion</b> (emergency, especially in children)</td></tr><tr><td>Acute renal failure</td><td>Hemodialysis (rare; full recovery usual)</td></tr><tr><td>Neonatal jaundice</td><td>Phototherapy / exchange transfusion</td></tr><tr><td>CNSHA</td><td>Folic acid supplements; avoid hemolytic drugs; transfusion for exacerbations</td></tr><tr><td>Severe CNSHA</td><td><b>Splenectomy</b> — beneficial (unlike episodic G6PD where splenectomy has no role)</td></tr><tr><td>Prevention</td><td>Screen before prescribing primaquine/dapsone; avoid fava beans</td></tr></table><h3 style="color:#1a5276;margin-top:20px;">12. HIGH-YIELD MCQ RAPID RECALL TABLE</h3><table border="1" cellpadding="6" cellspacing="0" style="border-collapse:collapse;width:100%;margin-bottom:15px;"><tr style="background:#d6eaf8;"><th>Question Theme</th><th>Answer</th></tr><tr><td>Most common enzymopathy worldwide</td><td>G6PD deficiency</td></tr><tr><td>Inheritance</td><td>X-linked recessive</td></tr><tr><td>Why RBCs uniquely vulnerable</td><td>HMP shunt = only NADPH source; no new enzyme synthesis possible</td></tr><tr><td>Stain for Heinz bodies</td><td><b>Crystal violet / Brilliant cresyl blue</b> (supravital)</td></tr><tr><td>Bite cells formed by</td><td>Splenic macrophages plucking out Heinz bodies</td></tr><tr><td>Classic drug trigger</td><td><b>Primaquine</b></td></tr><tr><td>Classic food trigger</td><td><b>Fava beans</b></td></tr><tr><td>Most common trigger overall</td><td><b>Infection</b></td></tr><tr><td>Self-limiting hemolysis variant</td><td>G6PD A− (African, Class III)</td></tr><tr><td>More severe variant</td><td>G6PD Mediterranean (Class II)</td></tr><tr><td>CNSHA without trigger</td><td>Class I G6PD deficiency</td></tr><tr><td>Timing of hemolysis after drug</td><td>2–3 days after exposure</td></tr><tr><td>Favism relationship</td><td>All favism = G6PD deficiency; NOT all G6PD = favism</td></tr><tr><td>Best time for G6PD enzyme assay</td><td><b>2–3 months after hemolytic episode</b></td></tr><tr><td>Class I & II enzyme activity</td><td><10%</td></tr><tr><td>Class III enzyme activity</td><td>10–60%</td></tr><tr><td>Malaria protection against</td><td>Plasmodium falciparum</td></tr><tr><td>Splenomegaly / cholelithiasis</td><td>Absent in episodic form; only in Class I CNSHA</td></tr><tr><td>Type of hemolysis (acute phase)</td><td>Primarily <b>intravascular</b></td></tr><tr><td>Dapsone — severe hemolysis in which variant</td><td><b>Mediterranean variant</b></td></tr><tr><td>Definitive diagnostic test</td><td>G6PD enzyme assay (spectrophotometric)</td></tr><tr><td>Screening test</td><td>Fluorescent spot test</td></tr></table></div>
- Copy the entire text in the code block above (the single line starting with
G6PD Deficiencyfollowed by a Tab, then all the HTML) - Paste into Notepad (Windows) or TextEdit in plain text mode (Mac) — save as
g6pd.txtwith UTF-8 encoding - In Anki: File → Import
- Select
g6pd.txt - Set Field separator → Tab
- Map Field 1 → Front, Field 2 → Back
- Ensure "Allow HTML in fields" is checked
- Click Import
From the summary you gave regarding G6PD deficiency, if suppose in the next exam, NEET PG, a question comes from G6PD, what is the percentage probability that it can come from this summary? I mean, the question's answer is present in the summary.
Coverage Probability Assessment
What the summary covers completely
- Inheritance pattern
- Pathogenesis chain (G6PD → NADPH → GSH → Heinz bodies → bite cells)
- Why RBCs are uniquely vulnerable
- WHO classification (Class I–V, enzyme activity levels)
- G6PD A− vs Mediterranean comparison
- All major triggers (primaquine, dapsone, fava beans, infection)
- Peripheral smear findings and their stains
- Clinical presentations (AHA, favism, NNJ, CNSHA)
- Lab diagnosis including the MCQ trap (false normal after episode)
- Genetics (X-linked, lyonization, missense mutations)
- Malaria protection
- Treatment including splenectomy in CNSHA
- Favism relationship (one-way, not bidirectional)
Honest Probability Estimate
| Scenario | Probability |
|---|---|
| Question is a standard/repeated G6PD concept | ~95% covered |
| Question is a clinical vignette (drug given → hemolysis) | ~95% covered |
| Question tests a specific drug name already in the list | ~95% covered |
| Question tests an obscure biochemical detail not in standard pathology scope | ~60–70% covered |
| Question is a completely novel framing from a non-standard source | ~70% covered |
Overall realistic coverage: ~90–92%
What could fall outside the summary (the ~8–10%)
- Specific enzyme kinetics — e.g. Km values, exact reaction mechanism of G6PD (more biochemistry than pathology; unlikely in NEET PG pathology)
- Molecular genetics details — exact nucleotide substitution in G6PD A− or Mediterranean variant (rarely asked)
- Neonatal jaundice management specifics — bilirubin threshold for exchange transfusion (neonatal medicine territory, not pathology)
- Comparison with other HMP shunt enzyme deficiencies — e.g. glutathione synthetase deficiency, 6-phosphogluconate dehydrogenase (extremely rare questions)
- Specific geographic prevalence statistics — exact percentages in specific populations (factual recall, not conceptual)