Pathophysiology of hepatobilary system
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Here is a comprehensive overview of the pathophysiology of the hepatobiliary system, drawn primarily from Robbins & Kumar Basic Pathology and Sleisenger and Fordtran's Gastrointestinal and Liver Disease.
Pathophysiology of the Hepatobiliary System
1. Structural Foundation
The adult liver (1,400–1,600 g) has a dual blood supply: the portal vein (60–70% of flow) and the hepatic artery (30–40%). Understanding two organizational models is essential for interpreting patterns of injury:
Lobular model — hepatocytes arranged around a central (terminal hepatic) vein, with portal tracts at the periphery.
Acinar model — three metabolic zones based on proximity to the blood supply:
- Zone 1 (periportal): richest O₂ supply; most vulnerable to toxins metabolized to reactive intermediates
- Zone 3 (centrilobular/pericentral): poorest O₂ supply; most vulnerable to ischemia and alcohol injury
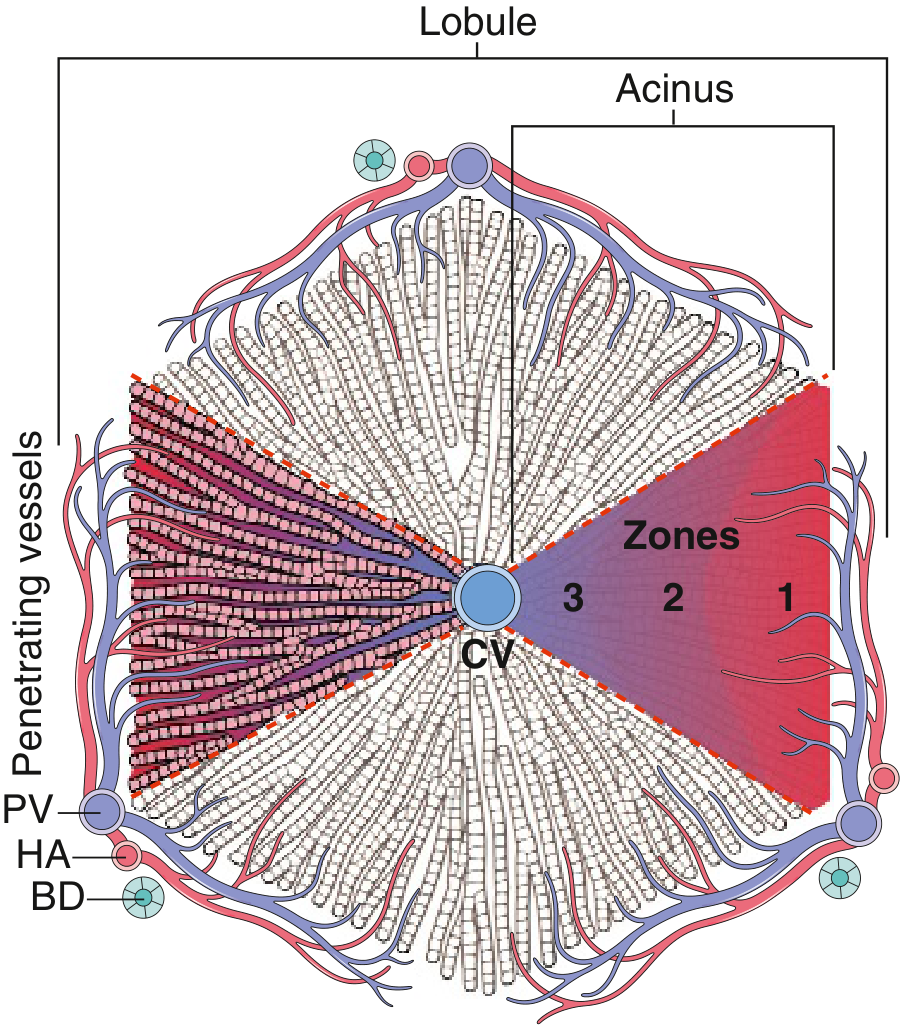
Key microstructural elements:
- Sinusoids: fenestrated endothelium overlying the space of Disse, where hepatocyte microvilli project
- Kupffer cells: sinusoidal macrophages; phagocytose debris, bile pigment
- Hepatic stellate cells (Ito cells): reside in space of Disse; store vitamin A; become activated myofibroblasts during injury → key drivers of fibrosis
- Bile canaliculi: 1–2 µm channels between adjacent hepatocytes, draining into canals of Hering → bile ductules → portal bile ducts
2. General Mechanisms of Hepatocyte Injury
| Mechanism | Example |
|---|---|
| Steatosis (fat accumulation) | Alcohol, obesity, diabetes |
| Cholestasis (bile accumulation) | Obstruction, drug toxicity |
| Necrosis | Ischemia, paracetamol toxicity → zone 3 |
| Apoptosis | Viral hepatitis, autoimmune, drug-induced |
| Confluent necrosis | Bridging between central veins / portal tracts |
| Ductular reaction | Stem cell activation from canal of Hering; marker of severe chronic injury |
Liver disease is typically insidious — the large functional reserve means biochemical abnormalities precede clinical decompensation by months to years. — Robbins & Kumar, p. 596
Key Lab Correlates
| Test | What it reflects |
|---|---|
| ↑ AST / ALT | Hepatocyte membrane disruption |
| ↑ Alkaline phosphatase, GGT | Bile canalicular injury / cholestasis |
| ↑ Bilirubin | Impaired secretion or obstruction |
| ↓ Albumin, ↑ PT | Loss of hepatocyte synthetic function |
| ↑ Serum ammonia | Failure of urea cycle (encephalopathy risk) |
3. Acute Liver Failure
Characterized by sudden loss of hepatic function in a previously healthy liver. Viral hepatitis accounts for ~10% of cases worldwide (HAV is most common globally; HBV predominates in Asia/Mediterranean). Others: drug toxicity (paracetamol), ischemia.
Clinical consequences:
- Jaundice — failure to conjugate or excrete bilirubin
- Coagulopathy — reduced synthesis of clotting factors I, II, V, VII, IX, X
- Hepatic encephalopathy — accumulation of nitrogenous waste (ammonia, false neurotransmitters); cerebral edema in acute failure
- Hypoglycaemia — failure of gluconeogenesis
- Portal hypertension — its major acute consequences are ascites and encephalopathy
4. Chronic Liver Disease & Cirrhosis
4a. Pathogenesis of Fibrosis
Repeated hepatocyte injury → death → inflammatory cytokines (TGF-β, PDGF, TNF-α) → activation of hepatic stellate cells → transformation into myofibroblasts → deposition of collagen (types I and III) in the space of Disse → sinusoidal fibrosis.
Fibrous septa initially link portal tracts to central veins; with progression, nodules of regenerating hepatocytes become encircled by fibrous bands = cirrhosis.
Regression of fibrosis and even established cirrhosis may follow disease remission or cure — scars become thinner and eventually fragment. Adjacent regenerating nodules coalesce. — Robbins & Kumar, p. 598
4b. Morphology of Cirrhosis
- Macroscopic: nodular, shrunken liver; regenerative nodules separated by fibrous bands
- Micronodular (<3 mm): typical of alcohol-related and NAFLD cirrhosis
- Macronodular (>3 mm): typical of viral hepatitis
- End-stage may show "burned-out" appearance devoid of fat and inflammation
4c. Causes
| Cause | Key Mechanism |
|---|---|
| Chronic HBV/HCV | Immune-mediated hepatocyte destruction; persistent inflammation → fibrosis |
| Alcohol-related | Steatosis → steatohepatitis → centrilobular fibrosis → cirrhosis |
| NAFLD/NASH | Similar to alcohol; driven by insulin resistance, oxidative stress, lipotoxicity |
| Hereditary hemochromatosis | Iron deposition in hepatocytes → oxidative injury → fibrosis; Prussian blue–positive periportal deposition |
| Wilson disease | Copper accumulation; ATP7B mutation → hepatic/CNS injury |
| Alpha-1 antitrypsin deficiency | Misfolded protein accumulates in hepatocytes (PAS-positive globules) → ER stress |
| Autoimmune hepatitis | Interface hepatitis; plasma cell infiltrate; anti-SMA/ANA antibodies |
| Primary biliary cholangitis (PBC) | Autoimmune destruction of small intrahepatic bile ducts; anti-mitochondrial antibodies (AMA) in >95%; "florid duct lesion" on histology |
| Primary sclerosing cholangitis (PSC) | Inflammation + obliterative fibrosis of intra- and extrahepatic bile ducts; "beading" on MRCP; strongly associated with ulcerative colitis (~70%) |
5. Portal Hypertension
Definition: Portal pressure >10 mmHg (normal 5–10 mmHg); clinical consequences appear when >12 mmHg.
Causes (classified by site of resistance):
- Pre-hepatic: portal vein thrombosis
- Intrahepatic (most common): cirrhosis → fibrous distortion of sinusoids + stellate cell contraction → ↑ vascular resistance + ↑ nitric oxide–driven splanchnic vasodilation → hyperdynamic circulation
- Post-hepatic: Budd-Chiari syndrome, constrictive pericarditis
Consequences:
| Complication | Mechanism |
|---|---|
| Esophageal/gastric varices | Porto-systemic collaterals; rupture → life-threatening hemorrhage |
| Ascites | ↑ hydrostatic pressure + ↓ oncotic pressure (hypoalbuminemia) + secondary hyperaldosteronism → Na+/H₂O retention |
| Splenomegaly | Splenic congestion → hypersplenism (pancytopenia) |
| Hepatic encephalopathy | Ammonia + other toxins bypass the liver via collaterals |
| Hepatorenal syndrome | Splanchnic vasodilation → ↓ renal perfusion → activation of renin-angiotensin + renal sympathetic system → afferent arteriolar vasoconstriction → oliguria and azotemia |
| Caput medusae | Dilated periumbilical veins from recanalised umbilical vein |
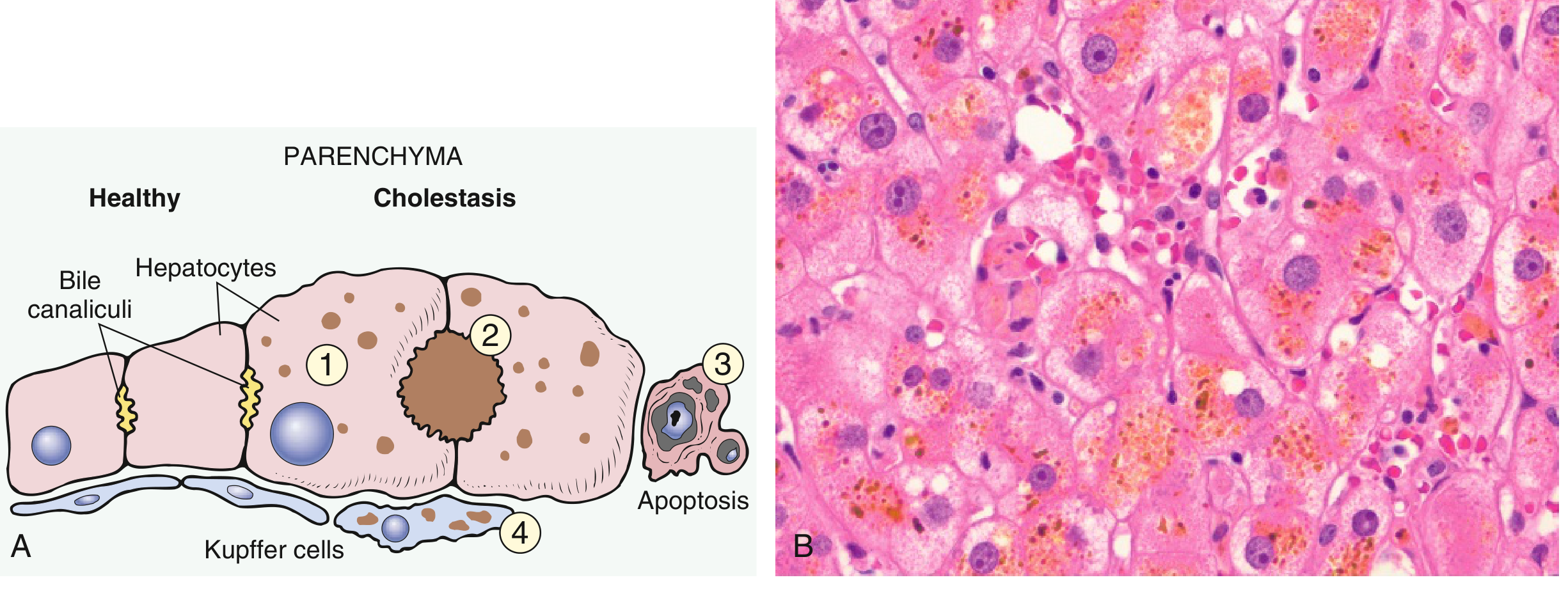
6. Cholestasis & Biliary Pathophysiology
Mechanisms
- Intrahepatic cholestasis: hepatocellular secretory failure (drugs, sepsis, viral hepatitis, PBC) or damage to intrahepatic bile ducts
- Extrahepatic cholestasis: choledocholithiasis (most common in adults), pancreatic carcinoma, strictures, biliary atresia (in children)
Morphology of cholestasis:
- Green-brown bile plugs in dilated canaliculi
- "Feathery degeneration" of hepatocytes (bile droplets)
- Kupffer cell engorgement with bile pigment
- Prolonged obstruction → ductular reactions → biliary cirrhosis
Clinical features: jaundice (conjugated hyperbilirubinaemia), dark urine, pale stool, pruritus (bile salt deposition), xanthomas, fat-soluble vitamin deficiency (A, D, E, K)
Ascending Cholangitis
Bacterial infection (usually gram-negative coliforms) ascending via obstructed biliary tree. Charcot's triad: fever, RUQ pain, jaundice. Suppurative form is life-threatening — dominated by sepsis over cholestasis.
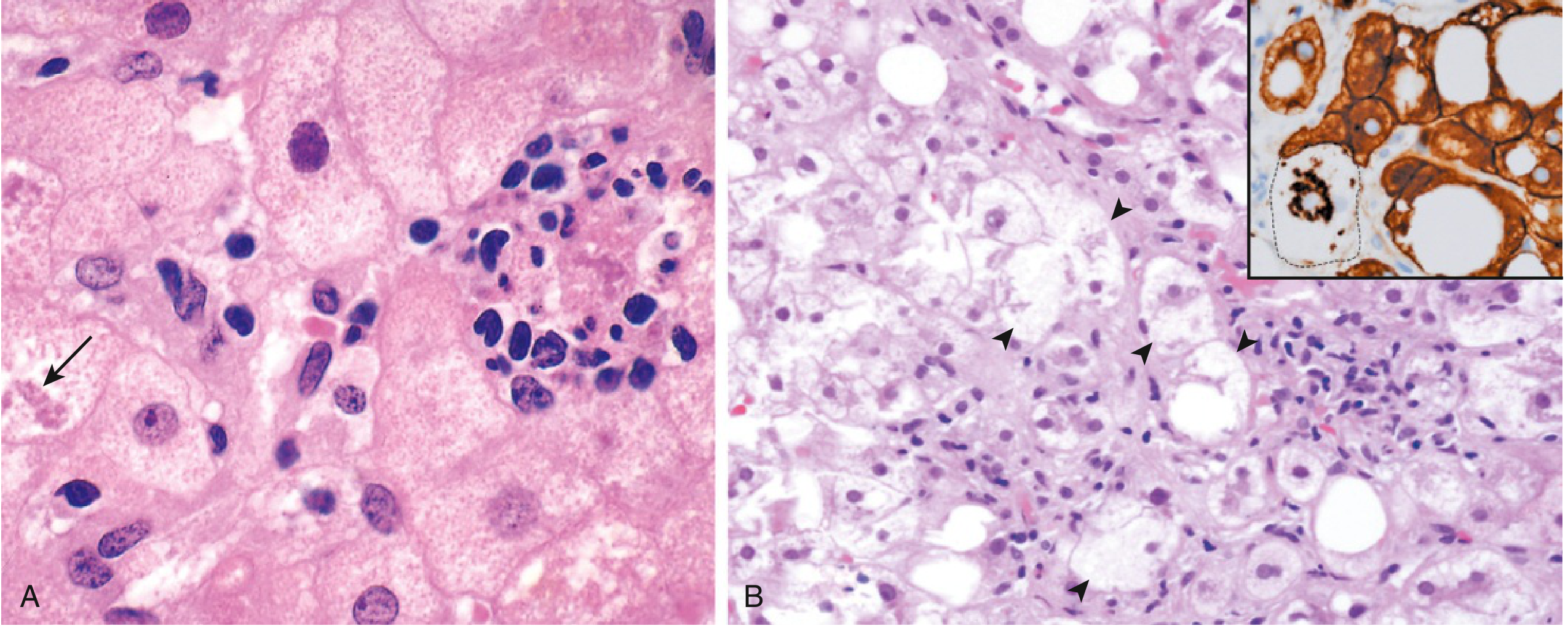
7. Fatty Liver Disease (Steatosis → Steatohepatitis → Fibrosis)
Progression (shared by alcoholic and non-alcoholic NAFLD):
- Steatosis — macrovesicular fat (large droplets displacing nuclei) begins in centrilobular (zone 3) hepatocytes; reversible with abstinence/weight loss
- Steatohepatitis — "second hit" (oxidative stress, inflammatory cytokines, gut-derived endotoxins):
- Hepatocyte ballooning (swelling + necrosis)
- Mallory hyaline bodies (tangled ubiquitinated keratins 8 and 18 in degenerating hepatocytes)
- Neutrophilic infiltration around ballooned cells
- Steatofibrosis — perisinusoidal "chicken-wire" collagen in space of Disse (zone 3) → bridging fibrosis → micronodular cirrhosis
8. Hepatic Encephalopathy
Pathogenesis (multifactorial):
- Ammonia hypothesis: liver failure → failure of urea cycle → systemic hyperammonaemia → astrocyte swelling → cerebral oedema
- Glutamine theory: excess ammonia → astrocytes produce glutamine as osmolyte → astrocyte swelling
- False neurotransmitters: aromatic amino acids (↑ in liver failure) cross BBB and compete with normal neurotransmitters
- Neuroinflammation and GABA receptor upregulation contribute
Precipitants: GI bleeding, infection, hypokalemia, constipation, sedatives, renal failure (hepatorenal syndrome)
9. Viral Hepatitis — Pathophysiological Summary
| Virus | Genome | Transmission | Chronicity | Key Pathophysiology |
|---|---|---|---|---|
| HAV | ssRNA | Fecal-oral | Never | CD8+ T cell–mediated lysis; self-limited |
| HBV | Partially dsDNA | Parenteral/sexual/perinatal | 5–10% adults; >90% neonates | Immune-mediated (CTL attack); cccDNA persistence |
| HCV | ssRNA | Parenteral | ~75–85% | Direct viral cytopathy + immune injury; high mutation rate → immune evasion |
| HDV | Defective ssRNA | Parenteral (coinfection/superinfection with HBV only) | ~70–80% with superinfection | Requires HBV surface antigen (HBsAg) for replication |
| HEV | ssRNA | Fecal-oral | Rare (immunosuppressed) | High mortality in pregnancy (20%); mechanism unclear |
10. Congestive Hepatopathy ("Nutmeg Liver")
Passive congestion from right heart failure → centrilobular sinusoidal congestion → hepatocyte atrophy. Left heart failure or shock → centrilobular ischemic coagulative necrosis (zone 3 most vulnerable due to lowest O₂ tension). Combined → centrilobular hemorrhagic necrosis → grossly mottled "nutmeg" appearance. Chronic severe heart failure → cardiac sclerosis (centrilobular fibrosis) → rarely cirrhosis.
Summary of Key Disease-Pattern Relationships
| Pattern of Injury | Zone Affected | Key Disease |
|---|---|---|
| Ischemic/congestion necrosis | Zone 3 (centrilobular) | Shock, right heart failure |
| Steatosis, alcoholic fibrosis | Zone 3 | Alcohol, NAFLD |
| Toxic injury (paracetamol) | Zone 3 | Drug-induced liver injury |
| Viral hepatitis, autoimmune | Piecemeal/interface | Chronic viral, AIH |
| Biliary cirrhosis | Portal/periportal | PBC, PSC, biliary obstruction |
| Iron deposition | Zone 1 (periportal) | Hereditary hemochromatosis |
Sources: Robbins & Kumar Basic Pathology, 10th Ed. (9780323790185), Chapter 14; Sleisenger and Fordtran's Gastrointestinal and Liver Disease (9780323609623).
This is a shared conversation. Sign in to Orris to start your own chat.