Uterotonics
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
I now have comprehensive information. Here is a structured overview of uterotonics:
Uterotonics
Uterotonics are agents that stimulate myometrial contractions. They are primarily used for:
- Labor induction/augmentation
- Prevention and treatment of postpartum hemorrhage (PPH) — especially from uterine atony (>70% of PPH cases)
- Management of incomplete/missed abortion
Classification
| Class | Agents |
|---|---|
| Oxytocin analogues | Oxytocin (Syntocinon) |
| Ergot alkaloids | Methylergonovine (Methergine) |
| Prostaglandins | Carboprost (15-methyl PGF₂α), Misoprostol (PGE₁), Dinoprostone (PGE₂) |
Individual Agents
1. Oxytocin
- Mechanism: Binds oxytocin receptors on myometrium → ↑ intracellular Ca²⁺ → uterine contraction. Receptor density increases at term.
- Dose (for PPH / cesarean):
- Prophylactic bolus: 1–3 IU IV
- Infusion: 2.5–15 IU/h (elective cesarean) or 18–36 IU/h (intrapartum cesarean)
- Alternatively: 10 IU IM
- Side effects: Hypotension, tachycardia (from rapid IV bolus — antidiuretic effect at high doses, water intoxication with prolonged use)
- First-line agent — given prophylactically on delivery of the placenta
- Note: Lower bolus doses used for elective cesarean; higher infusion rates for intrapartum cesarean
2. Methylergonovine (Ergometrine)
- Mechanism: Ergot alkaloid; acts on α-adrenergic and serotonin receptors → sustained uterine tetany
- Dose: 0.2 mg IM every 2–4 hours
- Side effects: Hypertension, vasoconstriction, coronary vasospasm
- Contraindications: Hypertension, pre-eclampsia, cardiovascular disease, Raynaud's phenomenon
- Second-line/additional agent for uterine atony
3. Carboprost (15-Methyl PGF₂α)
- Mechanism: Synthetic prostaglandin F₂α analogue → myometrial contraction
- Dose: 0.25 mg IM every 15–60 minutes (max 8 doses / 2 mg)
- Side effects: Bronchospasm, ↑ pulmonary vascular resistance, nausea, vomiting, diarrhea, flushing
- Contraindications: Asthma (relative — bronchospasm risk), active cardiovascular/pulmonary disease
4. Misoprostol (PGE₁ analogue)
- Mechanism: Prostaglandin E₁ analogue → uterine contraction and cervical ripening
- Dose: 800–1,000 μg PR/PO (sublingual or buccal) every 2 hours
- Side effects: Fever, nausea, shivering
- Advantages: Cheap, stable at room temperature, multiple routes (oral, sublingual, buccal, rectal, vaginal) — useful in resource-limited settings
- Also used for: cervical ripening, labor induction, incomplete abortion (with mifepristone)
Mechanism Summary
Both oxytocin and prostaglandins control bleeding by inducing myometrial contractions, reducing blood flow to the placental bed. Oxytocin receptor upregulation occurs near term, making the uterus maximally responsive.
Endogenous uterine stimulants also include endothelin and epidermal growth factor, which promote contractility by increasing intracellular calcium and stimulating prostaglandin synthesis — though these are not used therapeutically.
Clinical Use in PPH
The stepwise approach to uterotonic therapy in PPH:
- Oxytocin (first-line, prophylactic on placental delivery)
- Add methylergonovine if atony persists (avoid in hypertension)
- Add carboprost (avoid in asthma)
- Add misoprostol (especially when parenteral agents unavailable)
- If all uterotonics fail → intrauterine balloon tamponade, bimanual compression, pelvic vessel embolization, B-Lynch suture, or hysterectomy
Tranexamic acid (1 g IV) should be given alongside uterotonics in all PPH cases to reduce fibrinolysis.
Note: In uterine rupture, uterotonic agents are contraindicated as they may exacerbate the rupture. — Rosen's Emergency Medicine, p. 3087
Sources: Barash, Cullen, and Stoelting's Clinical Anesthesia, 9e (Table 41-5, p. 3517–3518); Rosen's Emergency Medicine, 10e (p. 3036–3038); Creasy & Resnik's Maternal-Fetal Medicine (Box 6.1, p. 133)
Tocolytics
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
The Creasy & Resnik section was cut off (it ran into references). The actual tocolytic content (lines 558–681) was truncated mid-way. I have enough from Goodman & Gilman plus Rosen's, and the diagram is excellent. I have comprehensive detail to compose a full answer.
Tocolytics
Tocolytics are agents that inhibit myometrial contractions to suppress preterm labor. The primary goal is short-term prolongation of pregnancy (48–72 hours) — sufficient time to administer corticosteroids for fetal lung maturation and/or arrange in utero transfer to an appropriate center. Tocolytic therapy does not prevent premature birth or improve neonatal outcomes such as respiratory distress syndrome directly.
"Although tocolytic agents delay delivery in about 80% of women, they neither prevent premature births nor improve adverse fetal outcomes such as respiratory distress syndrome." — Goodman & Gilman's Pharmacological Basis of Therapeutics, p. 1000
Mechanism Overview
The key event in myometrial contraction is myosin light-chain kinase (MLCK) activation, driven by intracellular Ca²⁺ and calmodulin. Tocolytics target this pathway at multiple points:
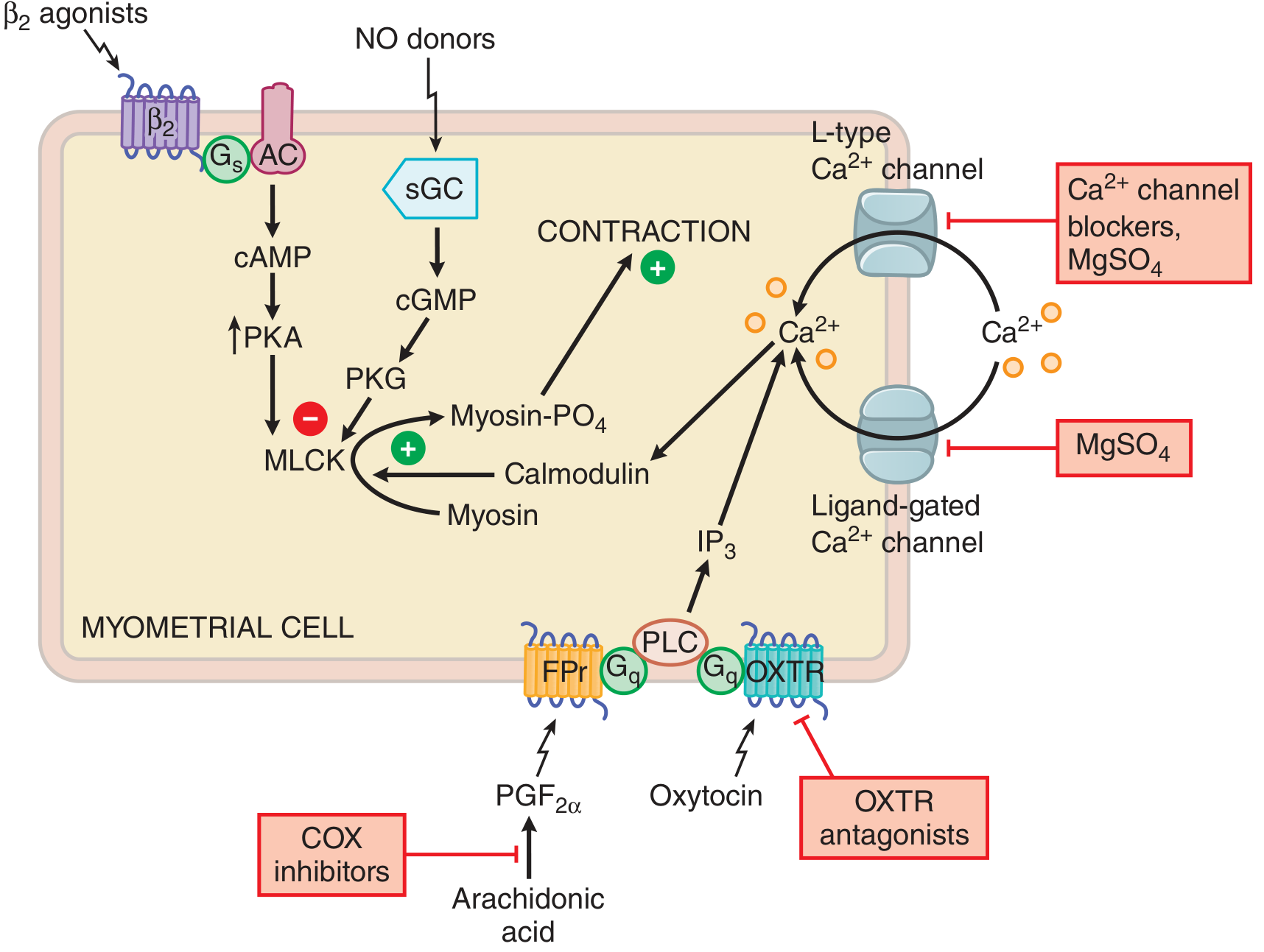
Figure: Sites of action of tocolytic drugs. Ca²⁺ channel blockers and MgSO₄ inhibit Ca²⁺ entry; β₂ agonists elevate cAMP→PKA→inactivate MLCK; NO donors elevate cGMP→PKG→inactivate MLCK; COX inhibitors block PGF₂α synthesis; OXTR antagonists block oxytocin receptor signaling. — Goodman & Gilman's, Fig. 48-7
Drug Classes
1. β₂-Adrenergic Agonists
Agents: Terbutaline, ritodrine (withdrawn from US market), isoxsuprine
Mechanism: β₂ receptor → Gs → adenylyl cyclase → ↑cAMP → PKA → phosphorylates/inactivates MLCK → relaxation
Doses (Terbutaline):
- SC: 0.25 mg every 20 min
- PO: 5–10 mg every 4–6 h
- IV: 2.5–5 mcg/min, up to 25 mcg/min
Efficacy: Delay delivery 48 h and 7 days; Cochrane reviews confirm effective but with the greatest side-effect burden of all tocolytics
Side effects: Tachycardia, hypotension, pulmonary edema (most serious), hypokalemia, hyperglycemia, tremor, palpitations; fetal tachycardia
Terbutaline should be limited to short-term inpatient use due to serious maternal cardiovascular effects and potential deleterious behavioral effects in offspring after in utero exposure. — Rosen's Emergency Medicine
Contraindications: Cardiac disease, uncontrolled diabetes, thyrotoxicosis
2. Calcium Channel Blockers
Agent: Nifedipine (most commonly used)
Mechanism: Blocks voltage-sensitive L-type Ca²⁺ channels in myometrial cell membrane → ↓ intracellular Ca²⁺ → ↓ MLCK activation → relaxation
Dose:
- 10–30 mg PO every 15–20 min for the first hour, then 10–20 mg PO every 4–8 h
Efficacy: Delay delivery 48 h; Cochrane reviews favor nifedipine — more likely to improve fetal outcomes and less likely to cause maternal side effects than β₂ agonists; currently the most widely preferred first-line agent
Side effects: Hypotension, flushing, headache, reflex tachycardia, peripheral edema
Advantages: Oral administration, favorable safety profile, inexpensive
3. MgSO₄
Mechanism: Competes with Ca²⁺ at voltage-gated and ligand-gated channels → reduces Ca²⁺ entry into myometrial cells → reduces MLCK activity
Dose:
- Loading: 4–6 g IV over 20 min
- Maintenance: 1–2 g/h IV infusion
Efficacy: Cochrane reviews conclude MgSO₄ is ineffective as a tocolytic for delaying delivery; however, it is used extensively for fetal neuroprotection (reduces risk of cerebral palsy) before 32 weeks
Side effects: Flushing, nausea, diplopia, loss of deep tendon reflexes (early toxicity), respiratory depression, cardiac arrest (severe toxicity)
- Antidote: Calcium gluconate 1 g IV
Monitoring: DTRs, respiratory rate, urine output, serum Mg levels
4. COX Inhibitors (NSAIDs)
Agent: Indomethacin (most studied)
Mechanism: Inhibits cyclooxygenase (COX) → ↓ prostaglandin (PGF₂α) synthesis → ↓ IP₃-mediated Ca²⁺ release → relaxation
Efficacy: Some data suggest reduction in preterm births; Cochrane found insufficient data due to small sample sizes
Side effects / fetal risks:
- Premature closure of the ductus arteriosus (serious — increases pulmonary vascular resistance)
- Oligohydramnios (reversible — reduced fetal renal prostaglandin-dependent perfusion)
- Impaired platelet function; NEC risk if used near delivery
Contraindications: Gestational age >32 weeks, renal/hepatic dysfunction, peptic ulcer disease, platelet disorders, suspected fetal cardiac or renal anomalies
5. Oxytocin Receptor Antagonists
Agent: Atosiban (not FDA-approved in USA; used in Europe)
Mechanism: Competitive antagonist at the myometrial oxytocin receptor (OXTR) → blocks Gq/PLC/IP₃ pathway → ↓ intracellular Ca²⁺ release → relaxation
Efficacy: Cochrane reviews conclude atosiban is ineffective compared to placebo for improving neonatal outcomes; non-inferior to β₂ agonists with fewer side effects
Side effects: Nausea, vomiting, headache — generally very well tolerated; minimal cardiovascular effects
6. NO Donors
Agent: Nitroglycerin (transdermal or IV)
Mechanism: NO → activates soluble guanylyl cyclase (sGC) → ↑cGMP → PKG → inactivates MLCK → relaxation
Use: One placebo-controlled trial of transdermal nitroglycerin before 28 weeks showed significant decrease in composite neonatal morbidity; also used for acute uterine relaxation (e.g., retained placenta, uterine inversion)
Side effects: Hypotension, headache, tachycardia
Summary Table
| Drug | Mechanism | Key Side Effects | Notes |
|---|---|---|---|
| Terbutaline | β₂ → ↑cAMP → ↓MLCK | Pulmonary edema, tachycardia | Short-term inpatient only |
| Nifedipine | CCB → ↓Ca²⁺ entry | Hypotension, flushing | Most-favored first-line |
| MgSO₄ | ↓Ca²⁺ entry/release | Toxicity → resp. depression | Better for neuroprotection |
| Indomethacin | COX inhibitor → ↓PGF₂α | Ductus arteriosus closure | Avoid >32 weeks |
| Atosiban | OXTR antagonist | Minimal | Not available in USA |
| Nitroglycerin | NO donor → ↑cGMP → ↓MLCK | Hypotension | Acute uterolysis |
Contraindications to Tocolysis
Maternal: Severe pre-eclampsia/eclampsia, hemodynamic compromise, placental abruption, chorioamnionitis, significant cardiac disease
Fetal: Fetal compromise/distress, lethal congenital anomalies, gestational age ≥34 weeks (generally), intrauterine demise
Relative: Placenta previa with bleeding (β-mimetics and CCBs may hamper cardiovascular response to hypotension; NSAIDs impair platelet function)
Key Clinical Points
- No tocolytic is FDA-approved specifically for preterm labor (ritodrine was the only one; voluntarily withdrawn)
- Tocolysis buys ~48–72 h — the window for betamethasone (corticosteroids for fetal lung maturity) to act
- Nifedipine is the preferred agent at most centers (best efficacy/safety ratio)
- Indomethacin is often preferred at very early gestational ages (<28 weeks) when ductus arteriosus risk is low
- Tocolysis fails in >25% of preterm patients for whom it is attempted
- No role for maintenance oral tocolysis — benefit is limited to the acute 48-hour window
Sources: Goodman & Gilman's Pharmacological Basis of Therapeutics, 13e (pp. 999–1000); Creasy & Resnik's Maternal-Fetal Medicine (pp. 900–901); Rosen's Emergency Medicine, 10e (Box 176.3)
This is a shared conversation. Sign in to Orris to start your own chat.