Carcinoma of rectum
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive, authoritative content to construct a detailed answer. Let me synthesize everything.
Carcinoma of the Rectum
Epidemiology
Rectal cancer accounts for approximately 31% of all colorectal cancers (CRCs), with ~46,050 new cases diagnosed annually in the United States (2023 data). The proportion of CRCs occurring in the rectum has steadily increased over the past two decades. Notably, an approximate 2% annual increase is observed in patients under 50 years of age. Risk factors mirror those for colon cancer. — Sabiston Textbook of Surgery
Pathogenesis: The Adenoma-Carcinoma Sequence
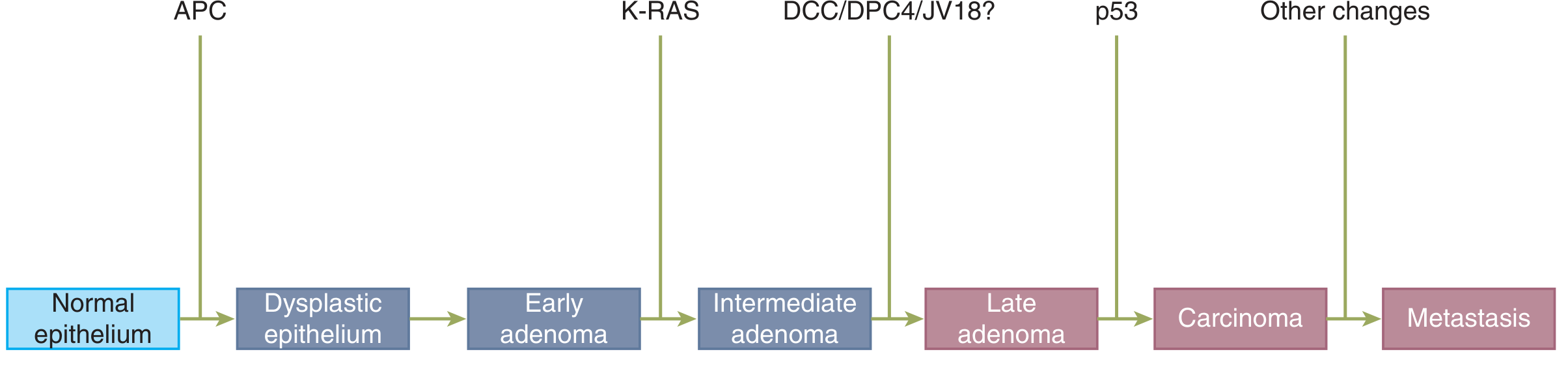
Approximately 80% of CRCs occur sporadically and ~20% arise in patients with a known family history. Three major molecular pathways drive CRC development:
- Chromosomal Instability (CIN) — Most common (65–70%). Follows the classic Fearon–Vogelstein adenoma-to-carcinoma sequence with sequential APC → KRAS → TP53/SMAD4 mutations.
- Serrated/Methylator pathway — BRAF mutation is the inciting event; leads to CpG-island hypermethylation (CIMP phenotype) silencing tumor-suppressor genes (~15–20% of CRC).
- Microsatellite Instability (MSI) — Defective DNA mismatch repair (dMMR); seen in Lynch syndrome and sporadic BRAF-mutated tumors.
Risk Factors
| Category | Factors |
|---|---|
| Dietary | High animal/saturated fat, low fiber, alcohol; oleic acid (olive oil) does NOT increase risk |
| Protective | Vegetable fiber; calcium, selenium, vitamins A/C/E, carotenoids |
| Hereditary | FAP (APC mutation), Lynch syndrome (MMR gene mutations), MYH-associated polyposis |
| IBD | Chronic ulcerative colitis — duration and extent of colitis correlate with risk |
| Other | Obesity, sedentary lifestyle, cigarette smoking, ureterosigmoidostomy, pelvic irradiation, acromegaly |
Clinical Presentation
Key symptoms and signs include:
- Rectal bleeding (most common)
- Tenesmus and rectal pain (suggest low tumor close to sphincter)
- Change in bowel habits — constipation, diarrhea, pencil-thin stools
- Neuropathic pain — indicative of locally invasive tumor involving sacral nerve roots
- Urinary/sexual dysfunction — from local invasion
- Weight loss, anemia — systemic features
Clinical Evaluation and Staging
Initial Workup
- Digital rectal examination (DRE) — assess tumor distance from anal verge and dentate line, mobility, fixation to adjacent structures (prostate, vagina, sphincter complex)
- Rigid/flexible proctoscopy with biopsy — confirm adenocarcinoma histology; IHC for MMR protein expression (dMMR/pMMR)
- Complete colonoscopy — assess synchronous cancers (~3%) and synchronous polyps (~30%)
- Serum CEA — baseline tumor marker; prognostic and useful for postoperative surveillance
Imaging Modalities
| Modality | Use |
|---|---|
| Pelvic MRI | Standard of care — assesses mesorectal (circumferential radial) margin, nodal status, tumor-free distance; best for predicting local recurrence risk |
| Endorectal ultrasound (ERUS) | Highly accurate for T and N staging (uT/uN classification); best for early lesions |
| CT chest/abdomen/pelvis | Evaluates distant metastases (liver, lung) |
| PET scan | Selected cases — equivocal CT findings; before high-morbidity surgery (pelvic exenteration, sacrectomy) |
TNM Staging (AJCC)
| Stage | Criteria | Approx. 5-yr Survival |
|---|---|---|
| I | T1–T2, N0, M0 (submucosa or muscularis propria; no nodes) | Excellent (>90%) |
| II | T3–T4, N0, M0 (through bowel wall; no nodes) | Good (~70–80%) |
| III | Any T, N+, M0 (nodal metastases) | Moderate (~40–60%) |
| IV | Any T, Any N, M1 (distant metastases) | Poor (~5–15%) |
The circumferential/radial margin (CRM) — best assessed by MRI — is a critical prognostic factor. CRM involvement (≤1–2 mm clearance) predicts high local recurrence and poor prognosis. — Schwartz's Principles of Surgery
Old Dukes' classification (now largely replaced by TNM):
- A – confined to bowel wall
- B – through bowel wall, no nodes
- C – nodal involvement
- D – distant metastases
Treatment
Localized Disease (Stage I–III)
Surgical Options
- Low Anterior Resection (LAR) — sphincter-preserving resection; anastomosis performed above the sphincter complex; suitable for mid- and upper-rectal tumors
- Abdominal-Perineal Resection (APR) — resection of rectum and anal sphincter with permanent end colostomy; required for low rectal tumors with sphincter invasion
Total Mesorectal Excision (TME) is the gold-standard surgical technique, consisting of sharp dissection of the entire mesorectal envelope under direct vision, removing all mesorectal lymph nodes in continuity.
Neoadjuvant Therapy (Stage II–III)
Rectal cancer (defined as tumor at or below the peritoneal reflection) has a higher risk of local recurrence than colon cancer after surgery alone. Neoadjuvant treatment reduces local recurrence risk:
- Standard chemoradiation — fluoropyrimidine (5-FU or capecitabine) + pelvic radiation → surgery → adjuvant chemotherapy
- Total Neoadjuvant Therapy (TNT) — all chemotherapy and radiation given preoperatively; associated with better compliance and similar local control
- For average-risk stage II/III: fluoropyrimidine + oxaliplatin (FOLFOX/CAPOX) → surgery (comparable local control to chemoradiation without radiation toxicity)
- For high-risk: chemoradiation + consolidation FOLFOX or CAPOX
- Nonoperative management ("watch and wait") — for patients achieving complete clinical response after total preoperative therapy; applicable to ~60–70% of such patients who can be cured without LAR/APR. However, 30–40% recur and need intensive follow-up; the majority of local recurrences after nonoperative management can be salvaged with surgery. — Harrison's Principles of Internal Medicine 22E
Radiation side effects to consider: radiation proctitis (especially in IBD), small-bowel obstruction, secondary malignancy (~1% at 10 years), and infertility in women (even without ovarian irradiation, due to uterine radiation effects).
Local Excision
Indicated for carefully selected early T1 (occasionally T2) lesions:
- Transanal excision
- Transanal endoscopic microsurgery (TEM)
- Transanal minimally invasive surgery (TAMIS) Salvage radical surgery is required if high-risk pathologic features are found.
Metastatic Disease (Stage IV)
- Complete resection of metastatic disease is curative, particularly for isolated hepatic or pulmonary metastases — multidisciplinary/tumor board consultation is essential
- MSI-high (dMMR) tumors: First-line checkpoint inhibitor immunotherapy (pembrolizumab); long-term survival and complete responses possible
- MSS (pMMR) tumors: Combination chemotherapy regimens:
- FOLFOX (5-FU + oxaliplatin + leucovorin) ± bevacizumab
- FOLFIRI (5-FU + irinotecan) ± bevacizumab
- FOLFIRINOX-bevacizumab
- Median survival: ~2–3 years; ~25% survive ≥5 years on systemic therapy
- KRAS/NRAS wild-type tumors: Anti-EGFR agents (cetuximab, panitumumab) can be added
- Primary tumor resection in unresectable metastatic disease: does NOT improve overall survival; reserved for symptomatic obstruction or bleeding
Prognostic Factors
| Factor | Impact |
|---|---|
| Nodal status (N) | Single most important prognostic factor |
| Circumferential radial margin (CRM) | Involvement → high local recurrence |
| Tumor differentiation | Poor differentiation → worse prognosis |
| Histology | Mucinous or signet-ring cell type → worse prognosis |
| Vascular/perineural invasion | Adverse |
| Preoperative CEA | Elevated → adverse prognosis |
| MSI status | MSI-high → better prognosis (sporadic); responds to immunotherapy |
| DNA aneuploidy | Adverse |
Postoperative Surveillance
- Stage I: No tumor marker or imaging follow-up recommended
- Stage II/III: History, physical, and CEA every 3–6 months × 2 years, then every 6 months × 5 years total; CT chest/abdomen/pelvis every 6–12 months × 5 years; colonoscopy at 1 year post-resection
- Stage IV (curative-intent resection): Same frequency as stage II/III for clinical and CEA; imaging every 3–6 months × 2 years, then every 6–12 months × 5 years total — Sabiston Textbook of Surgery
Key Points Summary
| Most common histology | Adenocarcinoma |
| Molecular pathway | Chromosomal instability (CIN) 65–70% → APC → KRAS → p53 |
| Most important prognostic factor | Lymph node involvement |
| Standard surgical technique | TME (total mesorectal excision) |
| Sphincter preservation | LAR (when feasible) |
| Gold-standard staging tool | Pelvic MRI |
| Neoadjuvant standard (stage II/III) | Chemoradiation or TNT |
| Watch-and-wait | After complete clinical response to total neoadjuvant therapy |
| MSI-high treatment | Checkpoint inhibitor (pembrolizumab) |
| Curative metastasectomy | Isolated liver/lung metastases |
This is a shared conversation. Sign in to Orris to start your own chat.