Clinical and pharmacological characteristics of antifungal and antiviral agents. Pharmacology
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
I now have comprehensive content from multiple textbooks. Let me synthesize a thorough answer.
Antifungal and Antiviral Agents: Clinical & Pharmacological Characteristics
PART I — ANTIFUNGAL AGENTS
Mechanisms of Action
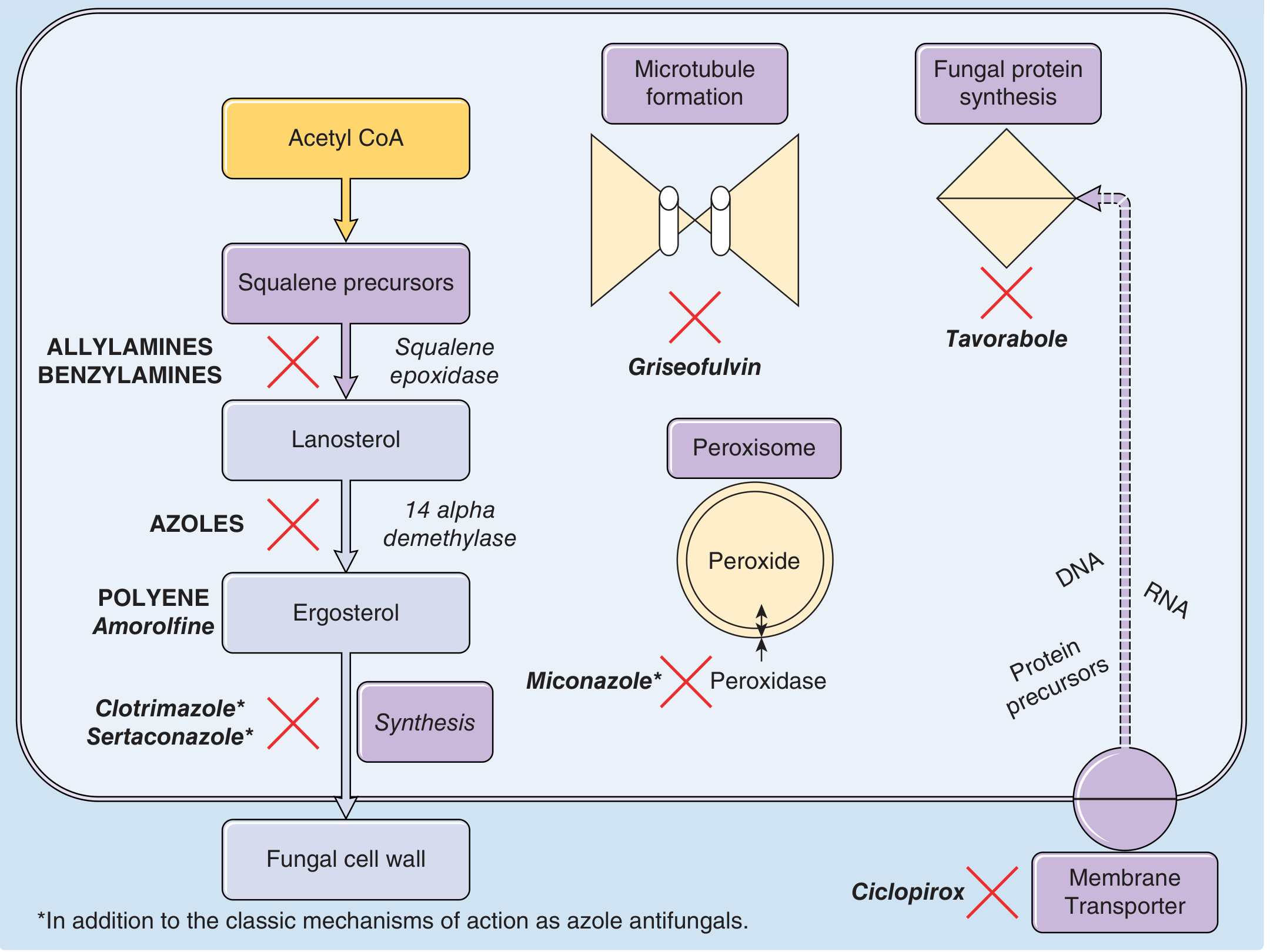
Antifungals exploit the unique biochemistry of fungal cells, principally the ergosterol biosynthesis pathway and the cell wall, which differ from mammalian cell membranes.
1. Polyenes (Amphotericin B, Nystatin)
Mechanism: Bind directly to ergosterol in the fungal cell membrane, forming transmembrane channels that cause ion leakage and cell death. Fungicidal.
Amphotericin B deoxycholate (D-AmB)
- Broad-spectrum: active against Candida, Aspergillus, Cryptococcus, endemic mycoses (histoplasmosis, coccidioidomycosis, blastomycosis)
- IV administration only for systemic infection
- Toxicity: Nephrotoxicity (dose-limiting), infusion-related reactions (fever, rigors, hypotension), hypokalemia, hypomagnesemia, anemia
- Lipid formulations (liposomal AmB, AmB lipid complex, AmB colloidal dispersion) are equally or more effective with significantly less nephrotoxicity; preferred when renal function is compromised
- Cryptococcal meningitis induction: 0.7–1 mg/kg/day IV + flucytosine 100 mg/kg/day for ≥2 weeks (AIDS), or 3 mg/kg/day liposomal formulation
Nystatin
- Topical only (too toxic for systemic use); used for oropharyngeal and cutaneous candidiasis
2. Azoles
Mechanism: Inhibit fungal cytochrome P450 enzyme 14α-demethylase (CYP51), blocking conversion of lanosterol → ergosterol. Ergosterol depletion disrupts membrane integrity. Generally fungistatic (fungicidal against some Candida spp.).
Imidazoles (topical)
| Drug | Key Use | Notes |
|---|---|---|
| Clotrimazole | Tinea corporis/cruris/pedis, vaginal candidiasis | OTC cream/lotion; twice daily 2–4 weeks |
| Miconazole | Dermatophytoses | OTC; twice daily 2–4 weeks |
| Ketoconazole | Tinea versicolor, seborrheic dermatitis | Cream/shampoo; systemic use largely abandoned due to hepatotoxicity |
| Econazole, Oxiconazole, Sertaconazole | Dermatophytoses, tinea versicolor | Rx topical agents |
Miconazole additional mechanism: Also inhibits fungal peroxidase, causing accumulation of peroxides toxic to the cell.
Triazoles (systemic) — preferred over imidazoles for systemic infections due to better selectivity
Fluconazole
- Water-soluble; oral bioavailability nearly 100% (unaffected by food or gastric pH)
- Good CNS penetration — drug of choice for cryptococcal meningitis consolidation (400 mg/day × 8 weeks) and maintenance (200 mg/day × 6–12 months)
- Metabolized via CYP3A; inhibits CYP3A4 and CYP2C9 — multiple drug interactions (warfarin, phenytoin, cisapride, astemizole → risk of fatal arrhythmias)
- Adverse effects: Headache, GI upset, prolonged QTc, rare Stevens-Johnson syndrome/toxic epidermal necrolysis, neutropenia
- Pregnancy Category C
Itraconazole
- Highly lipophilic; capsules require gastric acidity (take with food); cyclodextrin solution absorbed on empty stomach
- Accumulates in nails (reservoir 6–9 months post-treatment) — useful for onychomycosis
- Half-life ~21 hours; metabolized by CYP3A4 (dose adjust in liver disease)
- Broad-spectrum including Aspergillus, dimorphic fungi, dermatophytes
- Drug interactions: Major CYP3A4 inhibitor
Voriconazole
- First-line for invasive aspergillosis (superior to D-AmB in randomized trials)
- Active against Aspergillus, Candida (including fluconazole-resistant spp.), Fusarium, dimorphic fungi
- Visual disturbances (transient photopsia) common; hepatotoxicity, QTc prolongation
- Avoid if substantially elevated aminotransferases or prolonged QTc
- Not recommended in patients <2 years; unpredictable PK in children
Isavuconazole
- Equally effective as voriconazole for invasive aspergillosis with fewer adverse effects and more predictable pharmacokinetics — increasingly considered treatment of choice
- Also active against mucormycosis (advantage over voriconazole)
Posaconazole
- Noninferior to voriconazole for invasive aspergillosis (all-cause mortality); less toxic
- Extended-spectrum: only triazole with activity against Mucor
- Used for prophylaxis in high-risk neutropenic patients and GVHD patients
3. Allylamines (Terbinafine, Naftifine)
Mechanism: Inhibit fungal squalene epoxidase, blocking early ergosterol synthesis; squalene accumulates and is directly toxic. Fungicidal against dermatophytes.
Terbinafine
- Oral: excellent for onychomycosis and tinea capitis (first-line)
- Highly lipophilic → concentrates in skin, nails, hair follicles; half-life ~17 hours
- Oral absorption not affected by food; metabolized by CYP2D6; eliminated in urine
- Dose adjustment required in renal or hepatic impairment
- Topical form: 3–5% systemic absorption
- Adverse effects: GI upset, taste disturbance (dysgeusia), rare hepatotoxicity, rare severe skin reactions
Naftifine
- Topical only; persists in stratum corneum up to 5 days after single application; 3–6% systemic absorption
- Indications: tinea pedis, cruris, corporis, versicolor, cutaneous candidiasis
- Side effects: local dryness, pruritus, irritation, erythema
4. Benzylamines (Butenafine)
Mechanism: Also inhibit squalene epoxidase (similar to allylamines).
- Topical; OTC (Lotrimin Ultra); once–twice daily for tinea infections and tinea versicolor
5. Echinocandins (Caspofungin, Micafungin, Anidulafungin)
Mechanism: Inhibit β-1,3-glucan synthase, blocking fungal cell wall synthesis (unique fungal target → minimal mammalian toxicity). Fungicidal against Candida; fungistatic against Aspergillus.
- IV only; active against Candida (including azole-resistant) and Aspergillus
- Caspofungin is the only echinocandin licensed for salvage treatment of invasive aspergillosis
- Anidulafungin + voriconazole combination may reduce 6-week mortality in invasive aspergillosis (hematologic malignancy patients)
- Not active against Cryptococcus neoformans — should not be used for cryptococcal meningitis
- Generally well-tolerated; liver enzyme elevation possible
- Minimal drug interactions (not CYP substrates)
6. Flucytosine (5-FC)
Mechanism: Converted intracellularly by fungal cytosine deaminase to 5-fluorouracil, which inhibits fungal DNA/RNA synthesis. Active only intracellularly.
- Used almost exclusively in combination (with amphotericin B) to prevent resistance
- Key use: cryptococcal meningitis induction (100 mg/kg/day in 4 divided doses)
- Adverse effects: Bone marrow suppression (leukopenia, thrombocytopenia), GI toxicity, hepatotoxicity — monitor levels and CBC
- Requires renal dose adjustment
7. Griseofulvin
Mechanism: Disrupts fungal microtubule formation (binds tubulin); arrests mitosis; also deposits in keratin precursor cells making them resistant to fungal invasion.
- Oral; only for dermatophyte infections (tinea capitis, tinea corporis, onychomycosis)
- Fungistatic; requires prolonged treatment
- Induces CYP450 (reduces effectiveness of warfarin, oral contraceptives)
8. Ciclopiroxamine
Mechanism: Inhibits membrane transport (chelates polyvalent cations vital for fungal enzyme function) — unique mechanism among topical antifungals.
- Topical for dermatophytoses; also available as nail lacquer for onychomycosis
9. Tavaborole
Mechanism: Inhibits fungal protein synthesis (inhibits leucyl-tRNA synthetase).
- Topical solution for onychomycosis
PART II — ANTIVIRAL AGENTS
General Principles
Most antivirals are nucleoside/nucleotide analogs or enzyme inhibitors that target virus-specific steps in replication while sparing host cells. Selectivity relies on viral-specific enzymes (thymidine kinase, viral DNA polymerase, protease, integrase, etc.).
1. Antiherpesvirus Agents
Nucleoside Analogues (Acyclovir Class)
Acyclovir (Aciclovir)
- Mechanism: Guanosine analog; selectively phosphorylated by HSV/VZV thymidine kinase (TK) to acyclovir-monophosphate → then by cellular enzymes to triphosphate → inhibits viral DNA polymerase and terminates DNA chain. Not activated in uninfected cells → excellent selectivity
- Active against HSV-1, HSV-2, VZV (VZV ~10× less sensitive than HSV)
- IV for severe/disseminated infections; oral for moderate disease
- Varicella dosing:
- Immunocompetent child: 20 mg/kg PO 4× daily × 5 days (if initiated within 24h)
- Immunocompromised (severe): 10 mg/kg IV q8h × 7–10 days
- Acyclovir-resistant VZV: foscarnet 40 mg/kg IV q8h until healed
- Resistance mechanism: Mutations in viral TK gene → cross-resistance to ganciclovir, valacyclovir, famciclovir, penciclovir
Valacyclovir
- Prodrug (valine ester of acyclovir); converted to acyclovir after intestinal absorption
- Superior oral bioavailability vs. acyclovir → higher plasma levels, less frequent dosing (3× vs. 5× daily)
- Standard for herpes zoster in adults: 1 g PO q8h × 7 days
Famciclovir
- Prodrug of penciclovir (structurally similar to acyclovir; guanosine analog)
- Same mechanism: activated by viral TK → inhibits viral DNA polymerase
- Better oral absorption than acyclovir; similar antiviral activity against VZV and HSV
- Standard for herpes zoster: 500 mg PO q8h × 7 days
- Contraindication: Do NOT combine with 5-fluorouracil (brivudin only — potentially lethal interaction; famciclovir caution)
Penciclovir
- Active form of famciclovir; available as topical cream for orolabial HSV
- Same mechanism as acyclovir; similar resistance profile
Ganciclovir / Valganciclovir
- Active against CMV (primary indication); also HSV, VZV, EBV
- Mechanism: phosphorylated by CMV-encoded kinase (UL97) → triphosphate inhibits CMV DNA polymerase
- Valganciclovir = oral prodrug with high bioavailability; replaces IV ganciclovir for most CMV indications
- Toxicity: Myelosuppression (neutropenia, thrombocytopenia — dose-limiting); use with caution with other myelosuppressants
Pyrophosphate Analogue
Foscarnet
- Mechanism: Inorganic pyrophosphate analog; directly inhibits viral DNA polymerases (and HIV reverse transcriptase) at the pyrophosphate-binding site without requiring TK phosphorylation
- Active against all herpesviruses including TK-deficient (acyclovir-resistant) mutants
- Second-line agent — more toxic than nucleoside analogs
- Indications: acyclovir-resistant HSV/VZV (especially in advanced AIDS), CMV retinitis, ganciclovir-resistant CMV
- Toxicity: Nephrotoxicity (most common, requires aggressive hydration), electrolyte disturbances (hypocalcemia, hypomagnesemia, hypo/hyperphosphatemia), genital ulceration
Other Herpesvirals
Cidofovir
- Nucleotide analog; directly inhibits viral DNA polymerase (does not require TK activation)
- Third-line; significant nephrotoxicity limits use
- Indicated for CMV retinitis in AIDS patients
Amenamevir
- Novel helicase-primase inhibitor — new class; inhibits viral DNA replication upstream of DNA polymerase
- Potent activity against acyclovir-resistant VZV and HSV
- Single dose 400 mg/day as effective and well-tolerated as valacyclovir 1 g TID for herpes zoster (Japanese trial)
- Licensed in Japan; investigational in USA
2. Antiviral Agents for Influenza
Oseltamivir (Tamiflu), Zanamivir
- Mechanism: Inhibit neuraminidase (sialidase) — a surface enzyme that cleaves sialic acid from host cell receptors, preventing release and spread of new viral particles
- Active against influenza A and B
- Oseltamivir: oral; Zanamivir: inhaled
- Most effective if started within 48 hours of symptom onset
Baloxavir marboxil
- Mechanism: Inhibits cap-dependent endonuclease (polymerase acidic/PA subunit) — blocks viral mRNA transcription; novel class
- Single oral dose; active against influenza A and B including oseltamivir-resistant strains
Amantadine, Rimantadine
- Mechanism: Block M2 ion channel of influenza A, preventing viral uncoating
- Active only against influenza A; widespread resistance limits current use
- CNS side effects (amantadine); rimantadine better tolerated
3. Antiretrovirals (HIV) — Overview of Classes
| Class | Example Agents | Target |
|---|---|---|
| NRTIs | Tenofovir, Emtricitabine, Abacavir | Reverse transcriptase (competitive/chain termination) |
| NNRTIs | Efavirenz, Rilpivirine | Reverse transcriptase (allosteric) |
| PIs | Ritonavir, Darunavir | Protease |
| INSTIs | Dolutegravir, Bictegravir | Integrase |
| Entry inhibitors | Enfuvirtide (T20), Maraviroc | gp41 fusion / CCR5 co-receptor |
All antiretroviral therapy (ART) is given as combination regimens to prevent resistance.
4. Antivirals for Hepatitis
Hepatitis B (HBV): Tenofovir (TDF or TAF), Entecavir — nucleoside/nucleotide analogs inhibiting HBV DNA polymerase/reverse transcriptase; Pegylated interferon-α for immune-mediated viral suppression
Hepatitis C (HCV) — Direct-Acting Antivirals (DAAs):
| Class | Examples | Target |
|---|---|---|
| NS3/4A protease inhibitors | Glecaprevir, Grazoprevir | Viral protease |
| NS5A inhibitors | Pibrentasvir, Ledipasvir | Replication complex |
| NS5B polymerase inhibitors | Sofosbuvir | RNA-dependent RNA polymerase |
- Combination DAA regimens achieve >95% sustained virologic response (SVR/cure)
- Sofosbuvir-based combinations (e.g., Sofosbuvir/Ledipasvir, Glecaprevir/Pibrentasvir) are pangenotypic
5. Antiviral Resistance
Key principles:
- Arises from mutations in viral targets (polymerase, TK, protease, integrase)
- Acyclovir resistance in HSV/VZV: usually TK mutations → cross-resistance to penciclovir/famciclovir/valacyclovir; foscarnet (TK-independent) remains active
- CMV resistance to ganciclovir: UL97 kinase mutations (most common) or UL54 DNA polymerase mutations
- Influenza resistance to oseltamivir: neuraminidase H275Y mutation — baloxavir remains active
- HIV resistance: Prevented by combination therapy; single-drug use rapidly selects resistant virus
- Azole resistance in Aspergillus: Emerging CYP51 mutations threaten utility of voriconazole, isavuconazole, posaconazole
Summary Table: Antifungal Drug Classes
| Class | Target | Examples | Spectrum | Cidal/Static |
|---|---|---|---|---|
| Polyenes | Cell membrane ergosterol | AmB, Nystatin | Broad | Fungicidal |
| Azoles | 14α-demethylase (ergosterol synthesis) | Fluconazole, Voriconazole, Itraconazole, Posaconazole, Isavuconazole | Broad | Fungistatic |
| Allylamines | Squalene epoxidase | Terbinafine, Naftifine | Dermatophytes | Fungicidal |
| Echinocandins | β-1,3-glucan synthase | Caspofungin, Micafungin, Anidulafungin | Candida, Aspergillus | Fungicidal vs Candida |
| Flucytosine | DNA/RNA synthesis | 5-FC | Candida, Cryptococcus | Fungistatic |
| Griseofulvin | Microtubule formation | Griseofulvin | Dermatophytes only | Fungistatic |
Sources: Goldman-Cecil Medicine (International Ed.); Fitzpatrick's Dermatology (Vol. 1–2) — chapters on antifungal and antiviral therapy
This is a shared conversation. Sign in to Orris to start your own chat.