Make easy assignment on leukemia (subject pathology) for pharmacy student
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Searching Images
leukemia blood smear microscopy ALL AML CML CLL
I now have all the information needed to write a comprehensive, well-structured assignment. Let me compile it.
Leukemia — Pathology Assignment
Subject: Pathology | For: Pharmacy Students
Definition
Leukemia is a malignant clonal proliferation of hematopoietic stem cells or their precursors in the bone marrow, leading to uncontrolled production of abnormal white blood cells (blasts or mature-appearing cells) that crowd out normal blood cell production. The leukemic cells spill into the peripheral blood and may infiltrate organs such as the spleen, liver, and lymph nodes.
Classification
Leukemias are broadly classified along two axes:
| Axis | Categories |
|---|---|
| Cell lineage | Myeloid (from granulocyte/monocyte precursors) vs. Lymphoid (from B/T lymphocyte precursors) |
| Clinical course | Acute (blasts predominate, rapid onset) vs. Chronic (mature-appearing cells, insidious onset) |
This gives the four major types:
| Type | Abbreviation | Cell of Origin |
|---|---|---|
| Acute Myeloid Leukemia | AML | Myeloid blast |
| Acute Lymphoblastic Leukemia | ALL | Lymphoid blast |
| Chronic Myeloid Leukemia | CML | Myeloid progenitor |
| Chronic Lymphocytic Leukemia | CLL | Mature B lymphocyte |
1. Acute Myeloid Leukemia (AML)
Definition
AML is an aggressive tumor composed of immature myeloid lineage blasts that replace the marrow and suppress normal hematopoiesis.
Pathogenesis
AML arises from driver mutations that disrupt normal myeloid differentiation and promote uncontrolled proliferation. Two major categories of mutations cooperate:
- Mutations impairing differentiation — affect transcription factors required for myeloid maturation (e.g., fusions like t(8;21) → RUNX1-RUNX1T1; inv(16); t(15;17) → PML-RARA in acute promyelocytic leukemia)
- Mutations promoting growth/survival — activate growth factor receptor signaling pathways (e.g., mutations in FLT3, RAS, KIT)
Mutations in epigenetic regulators (e.g., IDH1, IDH2, DNMT3A, TET2) also contribute to abnormal gene expression in AML.
Acute Promyelocytic Leukemia (APL/M3 AML): A critical subtype caused by t(15;17), creating the PML-RARA fusion protein. This blocks differentiation at the promyelocyte stage. Treatment with all-trans retinoic acid (ATRA) causes the leukemic promyelocytes to differentiate and undergo apoptosis — a landmark example of differentiation therapy in oncology, highly relevant to pharmacy students.
Key Features
- Age: Mainly adults; most common acute leukemia in adults
- Blast cutoff: ≥20% blasts in bone marrow or blood = diagnostic
- Morphology: Large blasts with Auer rods (pathognomonic — pink needle-like cytoplasmic inclusions found in AML only)
- Symptoms: Fatigue/pallor (anemia), infections (neutropenia), bleeding (thrombocytopenia) — the triad of bone marrow failure
Lab Findings
- Elevated WBC (or may be normal/low)
- Decreased RBCs, platelets
- Auer rods on blood smear (pathognomonic for AML)
- Bone marrow biopsy: ≥20% myeloblasts
Treatment (Pharmacology Relevance)
| Drug | Mechanism | Notes |
|---|---|---|
| Cytarabine (Ara-C) | Antimetabolite — inhibits DNA polymerase | Backbone of AML induction |
| Daunorubicin / Idarubicin | Anthracycline — topoisomerase II inhibitor | Combined with Ara-C ("7+3 regimen") |
| ATRA (tretinoin) | Binds PML-RARA → induces differentiation | Used specifically in APL (M3 AML) |
| Arsenic trioxide (ATO) | Degrades PML-RARA protein | Used with ATRA in APL |
| Venetoclax | BCL-2 inhibitor — induces apoptosis | For older/unfit patients |
| Midostaurin / Gilteritinib | FLT3 kinase inhibitors | For FLT3-mutated AML |
2. Acute Lymphoblastic Leukemia (ALL)
Definition
ALL is a neoplasm of lymphoid precursor cells (B-cell or T-cell lymphoblasts). It is the most common cancer in children.
Pathogenesis
- Arises from precursor B or T lymphoblasts
- Common chromosomal abnormalities:
- t(12;21) [ETV6-RUNX1]: Most common in pediatric B-ALL; favorable prognosis
- t(9;22) [BCR-ABL / Philadelphia chromosome]: Present in ~25% of adult ALL; poor prognosis without targeted therapy
- Hyperdiploidy (>50 chromosomes): favorable prognosis in children
Key Features
- Age: Peak in children (2–5 years); also occurs in adults
- Morphology: Lymphoblasts — medium-sized cells with scant cytoplasm, dispersed chromatin, and small nucleoli. No Auer rods (distinguishes from AML)
- Immunophenotype: B-ALL expresses CD19, CD10, TdT; T-ALL expresses CD3, CD7, TdT
- Sanctuary sites: CNS and testes — leukemic cells can hide here (important for therapy)
Symptoms
Same bone marrow failure triad as AML. Additionally: bone pain (lymphoblast infiltration), lymphadenopathy, hepatosplenomegaly.
Treatment (Pharmacology Relevance)
| Drug | Notes |
|---|---|
| Vincristine | Vinca alkaloid — disrupts microtubule polymerization |
| Prednisone / Dexamethasone | Corticosteroids — induce apoptosis in lymphoid cells |
| L-Asparaginase | Depletes asparagine — lymphoblasts cannot synthesize it |
| Methotrexate (intrathecal) | CNS prophylaxis/treatment |
| Imatinib / Dasatinib | BCR-ABL inhibitors for Ph+ ALL |
| Rituximab | Anti-CD20 monoclonal antibody for B-ALL |
3. Chronic Myeloid Leukemia (CML)
Definition
CML is a myeloproliferative neoplasm characterized by overproduction of mature granulocytes arising from a transformed hematopoietic stem cell.
Pathogenesis — The Philadelphia Chromosome
CML is defined by the presence of the BCR-ABL fusion gene. In ~95% of cases, this results from the t(9;22) translocation — creating the Philadelphia chromosome (the abnormally small chromosome 22). The BCR-ABL fusion encodes a constitutively active tyrosine kinase that drives uncontrolled myeloid proliferation.
The BCR-ABL protein:
- Constitutively activates signaling pathways (RAS, PI3K/AKT, STAT5)
- Promotes cell proliferation and survival
- Suppresses apoptosis
The BCR-ABL fusion gene is present in granulocytic, erythroid, megakaryocytic, and B-cell precursors, indicating that CML arises from a transformed hematopoietic stem cell.
Clinical Phases
| Phase | Features |
|---|---|
| Chronic phase | Elevated WBC with mature granulocytes, mild symptoms; responds well to therapy |
| Accelerated phase | Increasing blasts (10–19%), worsening cytopenias, resistance to therapy |
| Blast crisis | ≥20% blasts — resembles acute leukemia (myeloid or lymphoid); very poor prognosis |
Key Features
- Age: Peak incidence in 4th–5th decade; adults 25–60 years
- Blood smear: Marked leukocytosis with full spectrum of myeloid maturation (neutrophils, bands, metamyelocytes, myelocytes, basophilia, eosinophilia)
- Splenomegaly: Often massive (extramedullary hematopoiesis)
- LAP (Leukocyte Alkaline Phosphatase): Low or absent (vs. reactive leukocytosis where LAP is high)
Treatment (Pharmacology Relevance) — Landmark Drug Class
| Drug | Mechanism | Generation |
|---|---|---|
| Imatinib (Gleevec) | BCR-ABL tyrosine kinase inhibitor | 1st generation — revolutionized CML treatment |
| Dasatinib | BCR-ABL + SRC kinase inhibitor | 2nd generation |
| Nilotinib | Highly selective BCR-ABL inhibitor | 2nd generation |
| Bosutinib | BCR-ABL + SRC inhibitor | 2nd generation |
| Ponatinib | Covers T315I mutation (resistance) | 3rd generation |
Pharmacy Note: Imatinib is the prototype tyrosine kinase inhibitor (TKI) and is one of the most significant drugs in oncology history. It transformed CML from a fatal disease into a manageable chronic condition. Understanding TKI resistance mechanisms (e.g., T315I "gatekeeper" mutation) is important.
4. Chronic Lymphocytic Leukemia (CLL)
Definition
CLL is a neoplasm of mature-appearing, functionally incompetent B lymphocytes. It is the most common leukemia in adults in Western countries.
Pathogenesis
- Arises from mature B cells that accumulate because of defective apoptosis (failure to die) rather than rapid proliferation
- Key genetic abnormalities:
- del(13q14): Most common (55%); favorable prognosis
- del(11q): Associated with aggressive disease
- del(17p) / TP53 mutation: Worst prognosis; resistant to chemotherapy
- Trisomy 12: Intermediate prognosis
- Mutational status of IGHV gene: Mutated IGHV = better prognosis
Key Features
- Age: Predominantly elderly (>60 years); rare before 40
- Blood smear: Monomorphic small mature lymphocytes with scant cytoplasm and clumped chromatin; characteristic smudge/basket cells (fragile leukemic cells broken during smear preparation)
- Immunophenotype: CD5+, CD19+, CD23+, weak surface Ig (co-expression of CD5 — a T-cell marker — on B cells is a hallmark)
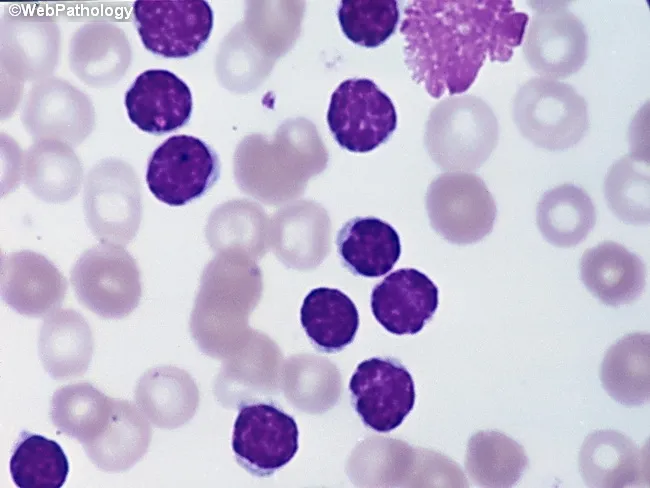
Symptoms
- Often asymptomatic, discovered incidentally on CBC (B symptoms can occur in advanced disease)
- Lymphadenopathy, splenomegaly, hepatomegaly
- Autoimmune complications: AIHA (autoimmune hemolytic anemia), immune thrombocytopenia
- Hypogammaglobulinemia → recurrent infections
Staging (Rai System — Simplified)
| Stage | Features |
|---|---|
| 0 | Lymphocytosis only |
| I | + Lymphadenopathy |
| II | + Splenomegaly / hepatomegaly |
| III | + Anemia (Hb < 11 g/dL) |
| IV | + Thrombocytopenia (platelets < 100,000) |
Treatment (Pharmacology Relevance)
| Drug | Mechanism |
|---|---|
| Ibrutinib | BTK (Bruton tyrosine kinase) inhibitor — blocks B-cell receptor signaling |
| Venetoclax | BCL-2 inhibitor — restores apoptosis |
| Rituximab / Obinutuzumab | Anti-CD20 monoclonal antibodies |
| Fludarabine | Purine analog antimetabolite |
| Chlorambucil | Alkylating agent (older therapy) |
Comparison Table: The Four Main Leukemias
| Feature | AML | ALL | CML | CLL |
|---|---|---|---|---|
| Course | Acute | Acute | Chronic | Chronic |
| Lineage | Myeloid | Lymphoid | Myeloid | Lymphoid (B cell) |
| Most common in | Adults | Children | Adults (40–60 yrs) | Elderly (>60 yrs) |
| Hallmark finding | Auer rods | TdT+, no Auer rods | Philadelphia chr. t(9;22) | Smudge cells, CD5+ B cells |
| Key mutation | Various (FLT3, NPM1, PML-RARA) | t(12;21) in children; t(9;22) in adults | BCR-ABL fusion | del(13q), TP53 mutations |
| Bone marrow | ≥20% myeloblasts | ≥20% lymphoblasts | Hypercellular, all myeloid stages | Infiltrated by small lymphocytes |
| Key drug class | Antimetabolites, ATRA (APL) | Corticosteroids, L-asparaginase | TKIs (imatinib) | BTK inhibitors (ibrutinib), BCL-2 inhibitors (venetoclax) |
| Prognosis | Variable; poor without treatment | Children: excellent; Adults: moderate | Excellent with TKIs | Indolent; median survival 10+ years |
Pathology of the Bone Marrow in Leukemia
In all leukemias, the bone marrow is progressively replaced by malignant cells:
- Normal hematopoiesis is suppressed → anemia, neutropenia, thrombocytopenia
- Consequence of cytopenias:
- Anemia → fatigue, pallor, dyspnea
- Neutropenia → infections (bacterial, fungal)
- Thrombocytopenia → bleeding (petechiae, ecchymoses, mucosal bleeding)
- Extramedullary hematopoiesis — leukemic cells migrate to spleen, liver, and lymph nodes → organomegaly
Pharmacy-Relevant Summary: Drug Targets in Leukemia
BCR-ABL kinase (CML/Ph+ ALL) ──→ Imatinib, Dasatinib, Nilotinib, Ponatinib
BTK kinase (CLL, B-cell) ──→ Ibrutinib, Acalabrutinib
BCL-2 (CLL, AML) ──→ Venetoclax
FLT3 kinase (AML) ──→ Midostaurin, Gilteritinib
PML-RARA fusion (APL/AML-M3) ──→ ATRA (tretinoin), Arsenic trioxide
CD20 (B-cell leukemias) ──→ Rituximab, Obinutuzumab
DNA synthesis (ALL, AML) ──→ Methotrexate, Cytarabine, Fludarabine
Microtubules (ALL) ──→ Vincristine
Topoisomerase II (AML) ──→ Daunorubicin, Idarubicin
Review Questions
- What is the pathognomonic morphological finding in AML on peripheral blood smear?
- Which chromosomal translocation defines CML, and what protein does it encode?
- Why is ATRA effective specifically in acute promyelocytic leukemia (M3 AML)?
- What cell surface markers distinguish CLL from other B-cell neoplasms?
- A 7-year-old child presents with bone pain, lymphadenopathy, and 30% lymphoblasts in the marrow. What is the most likely diagnosis, and what is the most favorable cytogenetic finding?
- How does venetoclax work, and in which leukemias is it used?
- Compare the LAP score in CML versus a leukemoid reaction.
- What is the "blast crisis" in CML, and why does it occur?
- Which chromosomal deletion in CLL is associated with resistance to chemotherapy?
- Name two sanctuary sites in ALL and explain why they are important for treatment planning.
References
- Robbins, Cotran & Kumar — Pathologic Basis of Disease, 10th Ed. (Chapters: Myeloid Neoplasms, Lymphoid Neoplasms)
- Robbins & Kumar — Basic Pathology, 11th Ed.
- Goldman-Cecil Medicine, 27th Ed.
This assignment covers the pathology of leukemia at the level appropriate for pharmacy students — emphasizing disease mechanisms, diagnostic features, and pharmacological targets.
This is a shared conversation. Sign in to Orris to start your own chat.