Make assignment on leukemia (subject pathology) for pharmacy student
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Searching Images
leukemia bone marrow blast cells classification
I now have rich, comprehensive material from Robbins & Kumar Pathologic Basis of Disease to write a thorough pharmacy student assignment on leukemia. Let me compose it.
LEUKEMIA
Pathology Assignment — Pharmacy Students
1. Introduction
Leukemia is a clonal malignancy of hematopoietic stem or progenitor cells characterized by uncontrolled proliferation of abnormal white blood cells that accumulate in the bone marrow and peripheral blood, suppressing normal hematopoiesis. Unlike solid tumors, leukemia is primarily a disease of the blood and bone marrow, though it can infiltrate the spleen, liver, lymph nodes, and other organs.
The word leukemia derives from the Greek leukos (white) + haima (blood), reflecting the historic observation of whitened blood in affected patients.
2. Classification
Leukemia is broadly classified along two axes:
| Axis | Categories |
|---|---|
| Cell lineage | Myeloid vs. Lymphoid |
| Clinical course | Acute vs. Chronic |
This produces four major types:
| Type | Abbreviation | Cell of Origin | Predominant Age |
|---|---|---|---|
| Acute Myeloid Leukemia | AML | Myeloid progenitor (HSC) | Adults (median ~65 yrs) |
| Acute Lymphoblastic Leukemia | ALL | Immature B or T lymphoblast | Children (peak ~3 yrs) |
| Chronic Myeloid Leukemia | CML | Pluripotent HSC (BCR-ABL) | Adults (30–60 yrs) |
| Chronic Lymphocytic Leukemia | CLL | Mature B cell | Older adults (>60 yrs) |
The figure below illustrates the stages of B- and T-cell differentiation from which specific lymphoid neoplasms (including leukemias) arise:
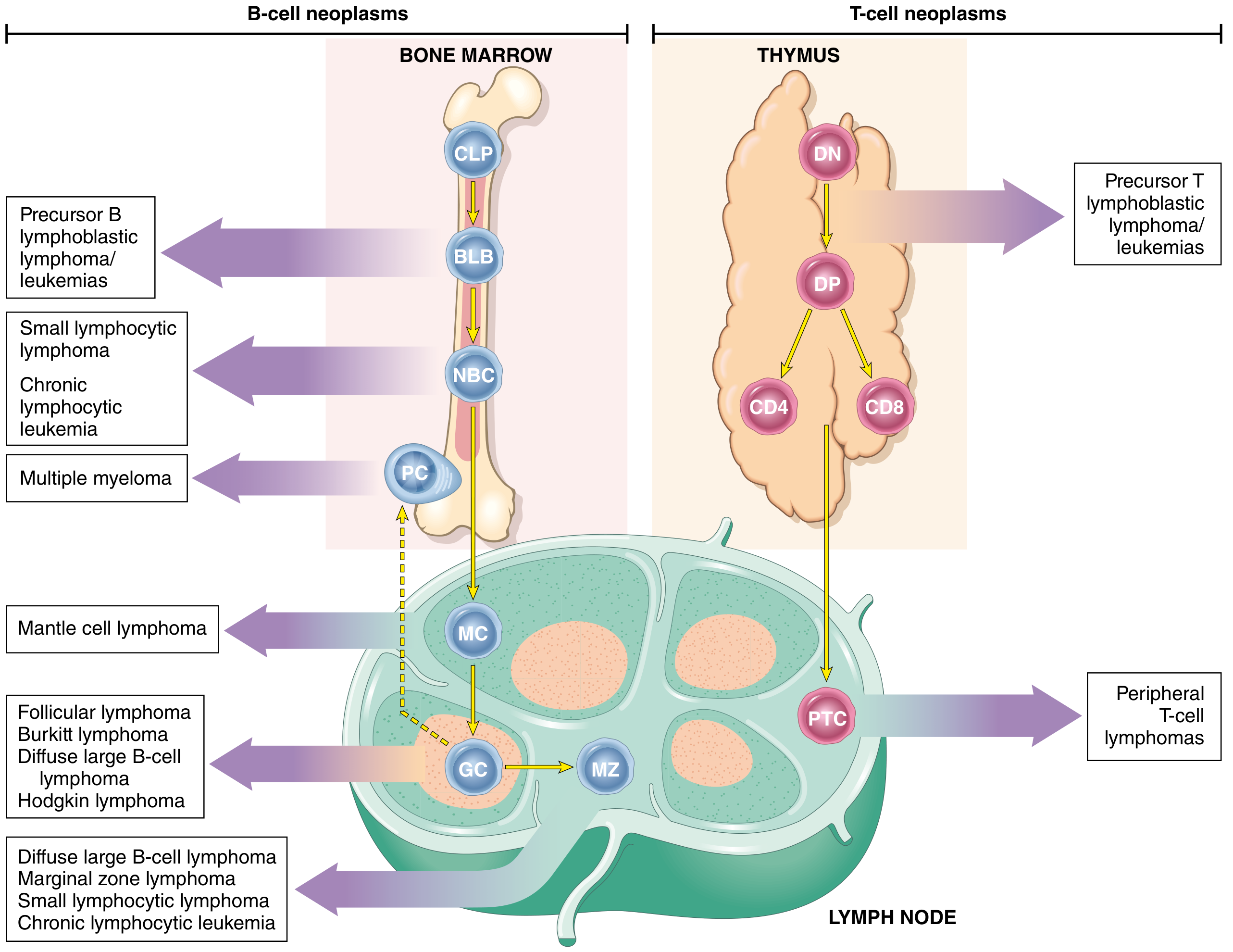
Fig. 1: Origin of lymphoid neoplasms. Stages of B- and T-cell differentiation from which specific lymphoid tumors emerge. BLB, Pre-B lymphoblast; CLP, common lymphoid precursor; GC, germinal center B cell; NBC, naïve B cell; PC, plasma cell; PTC, peripheral T cell. — Robbins & Kumar Pathologic Basis of Disease
3. Pathogenesis
3.1 General Mechanisms
Leukemogenesis is a multi-step process requiring accumulated genetic mutations that confer:
- Maturation arrest — cells are "frozen" at an immature stage and cannot differentiate
- Self-renewal — stem cell-like capacity for indefinite proliferation
- Resistance to apoptosis — escape from programmed cell death
Two broad categories of driver mutations cooperate in producing leukemia:
- Type I mutations — activate growth factor receptor signaling pathways (e.g., RAS, tyrosine kinases), promoting cell survival and proliferation
- Type II mutations — disrupt transcription factors required for normal differentiation (e.g., PAX5, RUNX1, NOTCH1), causing maturation arrest
3.2 Specific Molecular Mechanisms by Type
Acute Lymphoblastic Leukemia (ALL)
- B-ALL (~85% of ALL): mutations in transcription factor genes — PAX5, TCF3, ETV6, RUNX1, BCR::ABL1, KMT2A, and PBX1 — required for B-cell differentiation
- T-ALL (~15%): mutations in NOTCH1 (50–70% of cases), a gene essential for T-cell development
- Chromosomal aberrations in ~90% of ALL: hyperploidy (>50 chromosomes) is common and confers better prognosis; hypoploidy carries worse prognosis
- The BCR::ABL fusion (Philadelphia chromosome) occurs in a subset of B-ALL ("Ph+ ALL") and is especially important in adults — it defines a high-risk subgroup but is now targeted with TKIs
- Other recurrent translocations include t(12;21) involving RUNX1 and ETV6 (25% of childhood B-ALL, favorable)
Acute Myeloid Leukemia (AML)
- Arises from myeloid progenitors; associated with acquired mutations in transcription factor genes (RUNX1, CEBPA, PML::RARA in APL) that block myeloid differentiation
- Complementary mutations in growth-promoting genes: FLT3 (internal tandem duplication — most common, ~30%), NPM1, IDH1/2
- Can arise de novo or evolve from myelodysplastic neoplasms, myeloproliferative neoplasms, or following genotoxic exposures (therapy-related AML)
- The hallmark morphologic feature is >20% myeloid blasts in the bone marrow
Chronic Myeloid Leukemia (CML)
- Virtually defined by the Philadelphia chromosome — reciprocal t(9;22)(q34;q11) — present in >90% of cases
- This translocation creates the BCR::ABL chimeric gene, which encodes a constitutively active 210-kDa tyrosine kinase
- BCR-ABL activates multiple downstream proliferative pathways (RAS, PI3K, STAT5), causing massive clonal expansion of myeloid cells with retained ability to differentiate (hence chronic phase)
- The cell of origin is a pluripotent HSC, explaining why CML can transform to AML or ALL (blast crisis)
- CML follows a tri-phasic course: chronic phase → accelerated phase → blast crisis
Chronic Lymphocytic Leukemia (CLL)
- The most common adult leukemia in Western countries
- Clonal proliferation of mature B cells expressing CD5, CD19, CD20 (dim), CD23
- Key molecular features: deletion 13q14 (most common; favorable), trisomy 12, deletion 11q (ATM loss), deletion 17p (TP53 loss — worst prognosis), IGHV mutation status (mutated = better prognosis)
- Cells accumulate due to resistance to apoptosis (overexpression of BCL-2) rather than rapid proliferation
4. Morphology and Diagnosis
4.1 Acute Leukemias (AML & ALL)
Bone marrow biopsy shows replacement of normal marrow by blasts with:
- High nuclear-to-cytoplasm ratio
- Fine (euchromatic) chromatin
- Prominent nucleoli
- Scant cytoplasm
- Mitotic figures
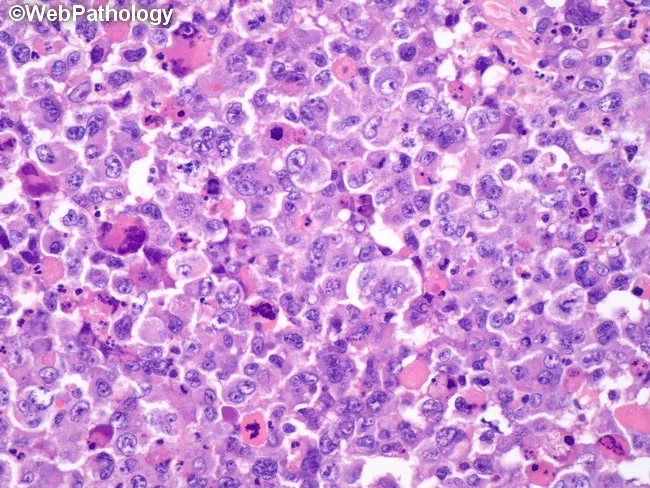
Fig. 2: High-magnification H&E of bone marrow showing sheets of blasts with high nuclear-to-cytoplasmic ratio and prominent nucleoli, consistent with acute leukemia.
AML-specific: Auer rods (pink cytoplasmic inclusions — crystallized azurophilic granules) are pathognomonic of AML when present. Myeloblasts may have granular cytoplasm.
ALL-specific: Lymphoblasts are slightly smaller, lack granules, and TdT (terminal deoxynucleotidyl transferase) is positive — a key immunophenotypic marker.
Diagnostic threshold: Acute leukemia is defined by ≥20% blasts in the marrow (WHO criteria).
4.2 CML
- Peripheral blood smear shows leukocytosis (often 50,000–200,000/μL) with the full spectrum of myeloid maturation: blasts, promyelocytes, myelocytes, metamyelocytes, bands, segmented neutrophils
- Basophilia and eosinophilia are characteristic
- Splenomegaly is often pronounced (extramedullary hematopoiesis)
- Marrow is hypercellular with near-complete replacement of fat
4.3 CLL
- Peripheral blood: absolute lymphocytosis (>5,000/μL) of small, mature-appearing lymphocytes with "smudge cells" (fragile lymphocytes crushed on the smear) — pathognomonic
- Bone marrow: small lymphocytes with condensed, clumped nuclear chromatin; scant cytoplasm
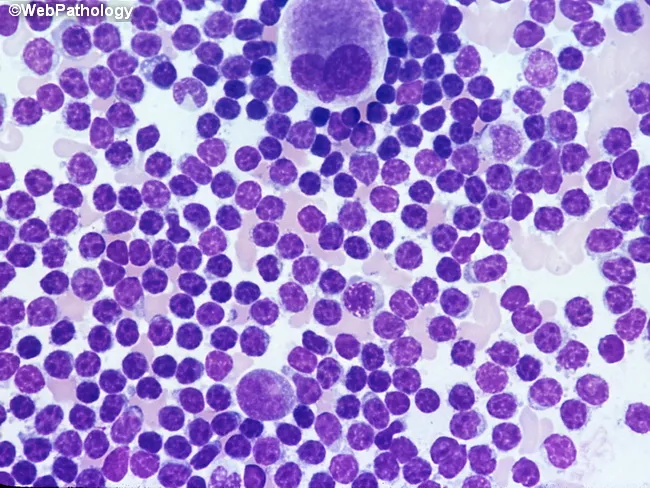
Fig. 3: Bone marrow aspirate in CLL showing predominant small lymphocytes with condensed chromatin and scant cytoplasm.
5. Clinical Features
5.1 Symptoms Resulting from Bone Marrow Failure
All acute leukemias share a triad of clinical presentations from marrow replacement:
| Cytopenia | Consequence | Symptoms |
|---|---|---|
| Anemia (↓ RBC) | Reduced oxygen delivery | Fatigue, pallor, dyspnea |
| Thrombocytopenia (↓ platelets) | Hemostatic failure | Petechiae, bruising, bleeding |
| Neutropenia (↓ WBC) | Immunosuppression | Recurrent/opportunistic infections, fever |
5.2 Infiltrative Features
| Site | Manifestation |
|---|---|
| Bone marrow | Pancytopenia, bone pain |
| Liver & spleen | Hepatosplenomegaly |
| Lymph nodes | Lymphadenopathy (ALL, CLL) |
| CNS | Headache, CN palsies (ALL) |
| Testes, skin, gums | Extramedullary leukemia (AML-M5) |
| Thymus | Anterior mediastinal mass (T-ALL) |
5.3 CML-Specific Course
| Phase | Features |
|---|---|
| Chronic phase (3–5 yrs) | Asymptomatic/mild; responds to treatment |
| Accelerated phase | ↑ blasts (10–19%), resistant cytopenias |
| Blast crisis | ≥20% blasts; behaves like AML or ALL |
5.4 CLL-Specific Features
- Often indolent; incidentally discovered on routine CBC
- Rai staging (0–IV) based on lymphocytosis, lymphadenopathy, organomegaly, anemia, thrombocytopenia
- Complications: hypogammaglobulinemia → infections; autoimmune hemolytic anemia; Richter transformation (→ aggressive DLBCL)
6. Laboratory Investigations
| Investigation | Purpose |
|---|---|
| CBC with differential | Establishes leukocytosis/cytopenia, WBC differential |
| Peripheral blood smear | Morphology of abnormal cells; smudge cells (CLL); Auer rods (AML) |
| Bone marrow aspirate & biopsy | Cellularity, blast percentage, architecture |
| Immunophenotyping (flow cytometry) | Cell surface markers for lineage determination |
| Cytogenetics (karyotype/FISH) | Chromosomal translocations — t(9;22), t(15;17), t(12;21) |
| Molecular tests (PCR, NGS) | BCR::ABL, FLT3-ITD, NPM1, IDH1/2, NOTCH1 mutations |
| LDH, uric acid | Tumor burden; risk of tumor lysis syndrome |
| Coagulation panel (PT, aPTT, fibrinogen) | Screen for DIC (especially AML-M3/APL) |
| Lumbar puncture | CNS involvement in ALL |
7. Treatment (Pharmacological Focus)
Pharmacological management is the cornerstone of leukemia treatment. The approach varies by subtype.
7.1 ALL
Pediatric ALL (most common childhood cancer)
- Highly curable (~85–90% in children) with multi-drug combination chemotherapy
- Standard phases: Induction → Consolidation → Maintenance
| Phase | Drugs Used |
|---|---|
| Induction | Vincristine + corticosteroids (prednisone/dexamethasone) + L-asparaginase ± anthracycline (daunorubicin) |
| Consolidation/Intensification | High-dose methotrexate, cytarabine, 6-mercaptopurine |
| Maintenance (2–3 years) | Oral 6-mercaptopurine daily + weekly methotrexate |
| CNS prophylaxis | Intrathecal methotrexate ± cytarabine (replaces cranial irradiation) |
Ph+ ALL (BCR-ABL+): Add a tyrosine kinase inhibitor (TKI):
- Imatinib (1st gen), dasatinib (2nd gen — preferred due to CNS penetration), or ponatinib (3rd gen)
Targeted therapy: Blinatumomab (anti-CD19 × CD3 bispecific T-cell engager) and inotuzumab ozogamicin (anti-CD22 antibody-drug conjugate) for relapsed/refractory B-ALL
7.2 AML
Induction ("7+3" regimen):
- Cytarabine (100–200 mg/m²) continuous IV infusion × 7 days
- Daunorubicin or idarubicin × 3 days
- Goal: achieve complete remission (marrow blasts <5%)
Acute Promyelocytic Leukemia (APL, AML-M3) — special case:
- Defined by t(15;17) → PML::RARA fusion
- Treated with All-Trans Retinoic Acid (ATRA) + Arsenic trioxide (ATO) — not conventional chemotherapy
- ATRA induces differentiation of the arrested promyelocytes; ATO promotes apoptosis
- Cure rate ~90% — the most curable form of AML
Targeted agents for AML:
- FLT3 inhibitors: Midostaurin (FLT3-ITD/TKD+), gilteritinib (R/R AML)
- IDH inhibitors: Ivosidenib (IDH1 mut), enasidenib (IDH2 mut)
- BCL-2 inhibitor: Venetoclax + azacitidine (for older/unfit patients)
- Gemtuzumab ozogamicin: Anti-CD33 antibody-drug conjugate (CD33+ AML)
7.3 CML — The Paradigm of Targeted Therapy
CML treatment is the landmark success story of molecular-targeted therapy in oncology.
| Generation | TKI | Target | Notes |
|---|---|---|---|
| 1st | Imatinib (Gleevec) | BCR-ABL, c-KIT, PDGFR | First approved TKI; revolutionized CML treatment |
| 2nd | Dasatinib, Nilotinib, Bosutinib | BCR-ABL + most resistant mutants | More potent; used for resistance/intolerance to imatinib |
| 3rd | Ponatinib | BCR-ABL including T315I mutant | Used for T315I "gatekeeper" mutation — resistant to 1st/2nd gen |
| 3rd | Asciminib | ABL myristoyl pocket (STAMP inhibitor) | Novel mechanism; specifically targets BCR-ABL |
Mechanism of imatinib: Competitively blocks the ATP-binding site of the BCR-ABL kinase, locking it in an inactive conformation → halts downstream proliferative signaling → apoptosis of CML cells.
Treatment goal: Deep molecular response (MR4.5 — BCR-ABL ≤0.0032% on international scale). With sustained deep molecular response, treatment-free remission (TFR) may be attempted.
7.4 CLL
- Many patients require no immediate treatment ("watch and wait")
- Treatment indicated for symptomatic or advanced disease
| Category | Regimen |
|---|---|
| Standard-risk | Ibrutinib (BTK inhibitor) or venetoclax + obinutuzumab |
| High-risk (del 17p, TP53 mut) | Ibrutinib or venetoclax (chemotherapy is ineffective) |
| Previously used (older) | FCR: Fludarabine + cyclophosphamide + rituximab |
Key drug mechanisms:
- Ibrutinib: Irreversible BTK (Bruton's tyrosine kinase) inhibitor → blocks B-cell receptor signaling → CLL cell death
- Venetoclax: BCL-2 inhibitor → restores apoptosis
- Rituximab/Obinutuzumab: Anti-CD20 monoclonal antibodies → ADCC + complement-mediated cytotoxicity
8. Complications
8.1 Tumor Lysis Syndrome (TLS)
- Occurs with rapid cell death (especially after chemotherapy in ALL/AML)
- Massive release of intracellular contents: hyperuricemia, hyperkalemia, hyperphosphatemia, hypocalcemia → AKI, arrhythmias, seizures
- Prevention: Allopurinol or rasburicase (recombinant urate oxidase) + vigorous IV hydration before therapy
8.2 Disseminated Intravascular Coagulation (DIC)
- Particularly associated with AML-M3 (APL)
- Leukemic promyelocytes release tissue factor and granule contents → activation of coagulation cascade → consumptive coagulopathy
- Can be fatal if ATRA/ATO therapy is delayed
8.3 Differentiation Syndrome (formerly ATRA syndrome)
- Complication of ATRA/ATO treatment in APL
- Symptoms: fever, respiratory distress, weight gain, effusions, hypotension (due to cytokine release from differentiating cells)
- Management: high-dose dexamethasone
8.4 Infections
- Major cause of morbidity and mortality in all leukemias
- Prolonged neutropenia from chemotherapy → bacterial, fungal (Aspergillus, Candida), and viral infections
- Prophylaxis: antibacterial (fluoroquinolones), antifungal (fluconazole/posaconazole), antiviral (acyclovir)
8.5 Drug-Specific Toxicities (Pharmacy-Relevant)
| Drug | Key Toxicities |
|---|---|
| Imatinib | Nausea, edema, hepatotoxicity, myelosuppression |
| Dasatinib | Pleural effusion, pulmonary hypertension, QT prolongation |
| Nilotinib | QT prolongation, hyperglycemia, peripheral arterial occlusion |
| Ibrutinib | Atrial fibrillation, bleeding, hypertension, diarrhea |
| Venetoclax | TLS (dose ramp-up required), neutropenia |
| ATRA | Differentiation syndrome, pseudotumor cerebri, teratogenicity |
| Arsenic trioxide | QT prolongation (monitor ECG), peripheral neuropathy |
| L-asparaginase | Pancreatitis, hepatotoxicity, coagulopathy, hypersensitivity |
| Methotrexate | Mucositis, nephrotoxicity, hepatotoxicity (requires leucovorin rescue at high doses) |
| Cytarabine | Cerebellar toxicity (high-dose), conjunctivitis (prophylactic eye drops required) |
9. Prognosis
| Type | Prognosis | Key Determinants |
|---|---|---|
| ALL (pediatric) | ~85–90% cure | Age, WBC count, cytogenetics, MRD status |
| ALL (adult) | ~40–50% cure | Ph-status, age; TKIs improve Ph+ ALL |
| AML | ~25–35% overall | Cytogenetics (ELN risk), FLT3/NPM1/IDH status, age |
| APL (AML-M3) | ~90% cure | ATRA + ATO era; early DIC management critical |
| CML | Near-normal life expectancy with TKIs | TKI adherence; TKI resistance; blast crisis transformation |
| CLL | Median survival 10+ years (low-risk) | del(17p)/TP53, IGHV mutation status, Rai stage |
10. Summary Table: Four Major Leukemias
| Feature | AML | ALL | CML | CLL |
|---|---|---|---|---|
| Cell origin | Myeloid progenitor | B/T lymphoblast | Pluripotent HSC | Mature B cell |
| Age | Adults | Children (ALL), adults (ALL) | 30–60 yrs | >60 yrs |
| Key mutation | PML::RARA, FLT3, NPM1 | BCR::ABL, NOTCH1, t(12;21) | BCR::ABL (Ph chrom) | del(13q), TP53, BCL-2 |
| Key morphology | Auer rods, myeloblasts | TdT+ lymphoblasts | Full myeloid spectrum in blood | Smudge cells, small lymphocytes |
| Cornerstone drug | Cytarabine + anthracycline; ATRA/ATO (APL) | Vincristine + steroids + L-asp; TKI (Ph+) | Imatinib/dasatinib (TKI) | Ibrutinib / venetoclax |
| Cure potential | Moderate (good risk) / Bone marrow transplant | High (pediatric) | No cure but durable remission | Indolent; not cured by current drugs |
11. Key Concepts for Pharmacy Students
-
Imatinib as a paradigm: The development of imatinib against BCR-ABL in CML transformed a fatal disease into a chronic, manageable condition and validated the concept of molecular targeted therapy. It demonstrates how understanding oncogenic kinases enables rational drug design.
-
ATRA in APL — differentiation therapy: Instead of killing cells, ATRA forces maturation arrest to resolve. This highlights that restoring differentiation, not just inducing cytotoxicity, is a valid pharmacological strategy.
-
TKI resistance: Mutations in the BCR-ABL kinase domain (e.g., T315I "gatekeeper mutation") confer resistance to 1st- and 2nd-generation TKIs → drives development of ponatinib and asciminib, illustrating pharmacological evolution against resistance.
-
Tumor Lysis Syndrome prophylaxis: Pharmacists play a critical role in risk-stratifying patients and ensuring allopurinol/rasburicase is prescribed before cytotoxic therapy is initiated.
-
Drug interactions with TKIs: Imatinib, dasatinib, nilotinib are substrates and inhibitors of CYP3A4 — significant drug interactions with azole antifungals, rifampin, and other CYP3A4 modulators. Pharmacist counseling is essential.
-
BCL-2 inhibition + TLS risk: Venetoclax carries a high risk of TLS, especially in CLL with high tumor burden. A mandatory dose ramp-up schedule (5 mg → 10 mg → 20 mg → 50 mg → 100 mg over 5 weeks) is required.
References
- Robbins & Kumar Pathologic Basis of Disease, 10th Edition — Chapter 13: Diseases of White Blood Cells, Lymph Nodes, Spleen, and Thymus, pp. 556–590
- Katzung's Basic and Clinical Pharmacology, 16th Edition — Chapter on Antineoplastic Agents
- WHO Classification of Haematopoietic and Lymphoid Tumours, 5th Edition (2022)
Assignment prepared for Pharmacy students, subject: Pathology | Topic: Leukemia
This is a shared conversation. Sign in to Orris to start your own chat.