Discuss congenital cholesteatoma in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
Finding Sources
Finding Sources
Finding Sources
Searching Images
congenital cholesteatoma middle ear diagram
Reading File
Reading File
Finding Sources
Reading File
Reading File
Loading Image
Loading Image
Loading Image
Now I have comprehensive material from the textbooks and imaging. Let me compose the detailed 50-mark answer.
CONGENITAL CHOLESTEATOMA
A Comprehensive Essay for 50 Marks
(RGUHS University Standard — ENT Postgraduate)
1. DEFINITION
Congenital cholesteatoma (CC) is a benign, cystic, expansile lesion composed of desquamating stratified squamous epithelium (keratinizing epidermoid cyst) located within the middle ear, mastoid, petrous apex, or other sites of the temporal bone, arising behind an intact tympanic membrane in a patient with no prior history of otitis media, otorrhoea, ear surgery, or tympanic membrane perforation.
The classic diagnostic criteria (Levenson, 1989):
- White, pearly mass medial to an intact tympanic membrane
- Normal pars flaccida and pars tensa
- No prior history of otorrhoea, perforation, or otological surgery
- No history of prior otitis media (debated — some authors now allow past OM)
— Cummings Otolaryngology Head and Neck Surgery
2. HISTORICAL BACKGROUND
| Year | Author | Contribution |
|---|---|---|
| 1838 | Müller | First described epidermoid cysts in temporal bone |
| 1850 | Virchow | Coined the term "Cholesteatoma" (misnomer — contains no cholesterol or fat) |
| 1922 | Aschoff | Better described as "Keratoma" |
| 1986 | Michaels | Demonstrated epidermoid formation (EF) in fetal temporal bones — basis of embryological theory |
| 1989 | Levenson | Established formal diagnostic criteria for CC |
| 2002 | Potsic et al. | Established the staging system |
The term "cholesteatoma" remains in use despite being a histological misnomer. The preferred pathological term is keratinising desquamative squamous epithelial cyst.
3. EPIDEMIOLOGY
- Congenital cholesteatoma accounts for 2–5% of all cholesteatomas in general otological practice
- In the pediatric population, CC represents approximately 20–30% of all pediatric cholesteatomas
- Annual incidence: approximately 3–15 per 100,000 children
- Sex: Male predominance (M:F ≈ 3:1)
- Age of presentation: Most commonly between 3–5 years (range: 6 months to teenage years)
- Side: Unilateral > bilateral (bilateral cases reported in twins — suggesting a genetic component)
- Site: Anterior-superior quadrant (most common, ~70%), posterior quadrant, or multiple quadrants
— Cummings Otolaryngology; Potsic et al., Arch Otolaryngol 2002
4. EMBRYOLOGY & THEORIES OF PATHOGENESIS
4.1 Michaels' Epidermoid Formation (EF) Theory (Most Widely Accepted)
Michaels (1986) examined 68 fetal temporal bones and found a small focus of keratinizing squamous epithelium — the Epidermoid Formation (EF) — in the anterior-superior quadrant of the middle ear between 10–33 weeks of gestation.
- This EF normally undergoes programmed apoptosis and disappears by the 33rd week of gestation
- Failure of involution of this EF leads to persistence and subsequent expansion, forming CC
- This explains the predominant location of CC in the anterior-superior quadrant
— Cummings Otolaryngology, Pathogenesis, block31; Dhingra Diseases of ENT
┌─────────────────────────────────────────────────────┐
│ MICHAELS' EPIDERMOID FORMATION THEORY │
│ │
│ 10-33 wks gestation │
│ Epidermoid Formation (EF) appears │
│ in anterior-superior middle ear cleft │
│ │ │
│ ┌──────┴──────┐ │
│ ↓ ↓ │
│ NORMAL FAILURE OF │
│ Involution Involution │
│ (apoptosis) (persistence) │
│ ↓ ↓ │
│ No disease CONGENITAL │
│ CHOLESTEATOMA │
└─────────────────────────────────────────────────────┘
4.2 Other Theories
| Theory | Description | Author |
|---|---|---|
| Implantation Theory | Squamous epithelium trapped during embryogenesis | — |
| Metaplasia Theory | Middle ear mucosa undergoes squamous metaplasia under chronic irritation | — |
| Amniotic Fluid Aspiration | Fetal aspiration of amniotic fluid with epithelial cells | — |
| Migration Theory | Epithelial cells migrate from EAC through a transient patent part of the developing TM | — |
| Neural Crest Cell Theory | Aberrant migration of neural crest cells that differentiate into squamous epithelium | Recent |
4.3 Genetic Basis
- Bilateral CC and CC in identical twins have been reported (Al Balushi et al.; Marchioni et al., IJPO 2017)
- A specific genetic mutation has not yet been identified
- Familial clustering suggests a heritable component — area of active research
5. SITES / LOCATION
LOCATIONS OF CONGENITAL CHOLESTEATOMA
(in order of frequency)
1. MIDDLE EAR (Most Common)
├── Anterior-superior quadrant (~70%)
├── Posterior quadrant
└── Entire middle ear
2. PETROUS APEX
3. MASTOID
4. CEREBELLOPONTINE ANGLE
5. EXTERNAL AUDITORY CANAL (rare)
6. INTRACRANIAL (very rare)
6. PATHOLOGY
6.1 Macroscopy
- Pearly-white, smooth, round, glistening cystic mass behind an intact TM
- Also called the "keratin pearl" or "white pearl"
- Cyst wall is thin and filled with laminated keratin flakes
6.2 Microscopy
The wall of the cyst consists of:
- Outer layer (Matrix): Stratified squamous epithelium — the "engine" of cholesteatoma
- Middle layer (Perimatrix): Fibrous connective tissue with inflammatory cells, fibrocytes
- Inner layer: Desquamated, laminated keratin debris (the "content")
6.3 Mechanism of Bone Erosion
A critical feature that makes cholesteatoma dangerous:
MECHANISM OF BONE EROSION IN CHOLESTEATOMA
Squamous Epithelium (Matrix)
│
↓ Produces
Cytokines: IL-1, IL-6, TNF-α
Enzymes: Collagenase, MMPs (Matrix Metalloproteinases)
Cathepsins, Acid Phosphatase
│
↓ Stimulate
Osteoclast Activation
│
↓
BONE EROSION
(Ossicles → Tegmen → Labyrinth → Facial Canal)
Key mediators identified in recent studies:
- MMPs (MMP-2, MMP-9) — most important
- RANKL/RANK/OPG pathway — regulates osteoclastogenesis
- EGF receptor signalling — promotes epithelial hyperproliferation
- TGF-β — promotes fibrosis in perimatrix
— Recent advances: Huisman et al.; Yung MW
7. CLINICAL FEATURES
7.1 Symptoms
The classic presentation of CC:
CLINICAL PRESENTATION FLOWCHART
Child (age 3-5 yrs), male predominance
No prior history of:
• Ear discharge (otorrhoea)
• Tympanic membrane perforation
• Ear surgery
• Chronic otitis media
│
↓
PRESENTING SYMPTOMS
┌────────────────┬────────────────────┬───────────────┐
│ Hearing loss │ Incidental finding │ Ear pain/ │
│ (Conductive) │ (on routine exam) │ fullness │
│ (Most common │ (common in early │ (uncommon) │
│ symptom) │ stage I) │ │
└────────────────┴────────────────────┴───────────────┘
│
Late/Complicated Cases:
• Facial nerve palsy
• Vertigo/sensorineural hearing loss
• Otorrhoea (if TM perforates)
• Meningitis / intracranial complications
7.2 Signs
On otoscopy:
- White, pearly mass behind an intact tympanic membrane — pathognomonic
- Typically in the anterior-superior quadrant
- Tympanic membrane is translucent; the mass shines through
- No perforation, no retraction pocket, no discharge
8. STAGING SYSTEM (Potsic et al., 2002)
The most widely used staging system — Cummings Otolaryngology, block31, p.2685:
╔══════════════════════════════════════════════════════════════╗
║ POTSIC STAGING SYSTEM FOR CONGENITAL ║
║ CHOLESTEATOMA (2002) ║
╠═══════╦══════════════════════════════════╦══════════════════╣
║ STAGE ║ EXTENT ║ RESIDUAL RISK ║
╠═══════╬══════════════════════════════════╬══════════════════╣
║ I ║ Single quadrant, no ossicular ║ Low ║
║ ║ involvement ║ (<5%) ║
╠═══════╬══════════════════════════════════╬══════════════════╣
║ II ║ Multiple quadrants, no ossicular ║ Moderate ║
║ ║ involvement ║ (~15%) ║
╠═══════╬══════════════════════════════════╬══════════════════╣
║ III ║ Ossicular involvement, no ║ High ║
║ ║ mastoid extension ║ (~33%) ║
╠═══════╬══════════════════════════════════╬══════════════════╣
║ IV ║ Mastoid extension ║ Very High ║
║ ║ ║ (~67%) ║
╚═══════╩══════════════════════════════════╩══════════════════╝
Stage IV carries a 67% risk of residual cholesteatoma after surgical resection, necessitating second-look surgery.
— Potsic WP et al., Arch Otolaryngol Head Neck Surg 128(9):1009–1012, 2002
9. INVESTIGATIONS
9.1 Audiological Assessment
- Pure Tone Audiometry (PTA): Conductive hearing loss — most common finding
- Air-Bone Gap (ABG): typically 20–40 dB
- Sensorineural component suggests labyrinthine involvement
- Tympanometry: Type A or As curve (if intact TM) — not Type B unless effusion coexists
- BERA (ABR): In young/uncooperative children
9.2 Radiology
CT Temporal Bone (High Resolution — HRCT):
- Investigation of choice for surgical planning
- Demonstrates:
- Soft tissue mass in middle ear (anterior-superior quadrant)
- Ossicular erosion — incus most commonly affected (lenticular process)
- Scutum erosion (Prussak's space involvement — more in acquired)
- Mastoid involvement (Stage IV)
- Tegmen or labyrinthine fistula (advanced disease)
MRI of Temporal Bone:
- Superior for soft tissue characterization and residual/recurrent disease
- Non-EPI DWI (Diffusion-Weighted Imaging): Investigation of choice for detecting residual cholesteatoma after surgery
- CC shows high signal on DWI + low signal on ADC map (restricted diffusion) — pathognomonic
- Sensitivity 90–100%, Specificity 90–100% for residual CC
- T1: Hypointense; T2: Hyperintense
IMAGING ALGORITHM FOR CONGENITAL CHOLESTEATOMA
Clinical suspicion
(White mass behind intact TM)
│
↓
HRCT Temporal Bone
(Extent, ossicular erosion, surgical planning)
│
↓
MRI (Non-EPI DWI)
(Soft tissue detail, pre-op characterization)
│
↓
POST-OPERATIVE SURVEILLANCE:
MRI Non-EPI DWI at 12–18 months
(Detect residual / recurrent disease)
│
┌───┴───┐
DWI +ve DWI -ve
│ │
Second-look Continue
surgery surveillance
The images below illustrate the radiological appearances:
Fig. 1 — Congenital Cholesteatoma: Otoscopic, CT, Intraoperative & Specimen
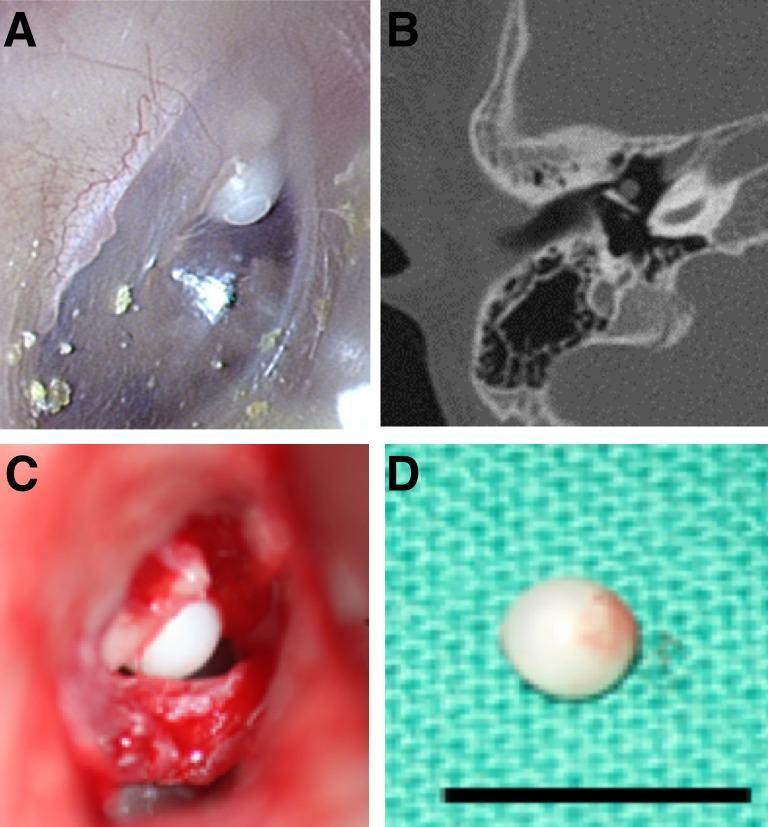
Panel A: Otoscopic view — white pearly mass in anterior-superior quadrant behind intact TM. Panel B: Axial CT showing soft tissue mass in middle ear. Panel C: Intraoperative view showing intact keratinous cyst. Panel D: Gross specimen — the classic keratin pearl.
Fig. 2 — Stage III CMEC: Endoscopy, CT, Intraoperative & Audiogram
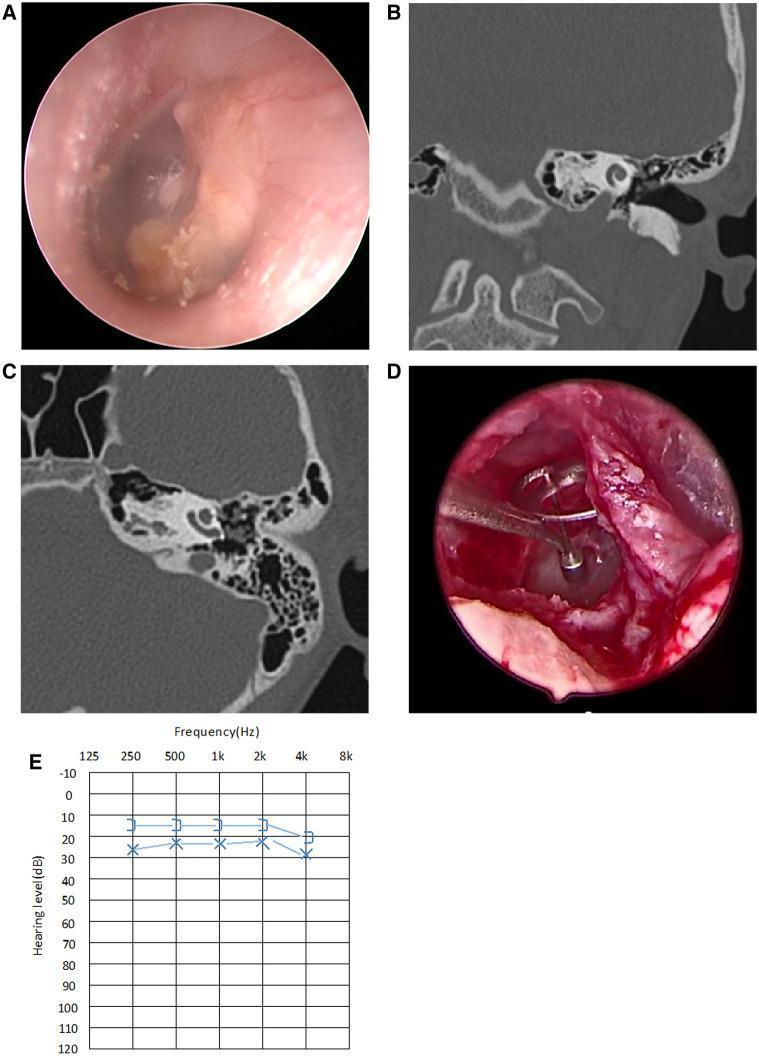
Panel A: Endoscopy showing whitish mass involving multiple quadrants. Panels B & C: CT (coronal/axial) showing middle ear soft tissue density with ossicular involvement. Panel D: TORP placement after complete excision. Panel E: Postoperative audiogram showing minimal residual CHL.
Fig. 3 — CBCT & MRI of Congenital Cholesteatoma
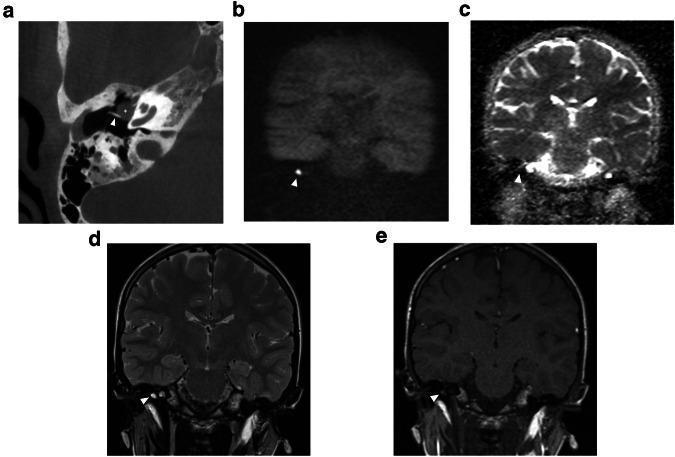
Panel a: Axial CBCT — well-defined soft tissue mass (asterisk) in anterior-superior middle ear. Panel b: High DWI signal; Panel c: Low ADC signal (restricted diffusion — confirms cholesteatoma). Panels d, e: T2 hyperintense, T1 hypointense. This MRI DWI pattern is pathognomonic.
10. DIFFERENTIAL DIAGNOSIS
DIFFERENTIAL DIAGNOSIS OF WHITE MASS BEHIND INTACT TM
┌─────────────────────┬───────────────────────────────────────────┐
│ CONDITION │ DISTINGUISHING FEATURES │
├─────────────────────┼───────────────────────────────────────────┤
│ Congenital │ Pearly, anterior-superior, DWI restricted │
│ Cholesteatoma │ No discharge, no perforation │
├─────────────────────┼───────────────────────────────────────────┤
│ Cholesterol │ Amber/blue TM; T1 bright on MRI │
│ Granuloma │ (no DWI restriction) │
├─────────────────────┼───────────────────────────────────────────┤
│ Middle Ear │ Fluid level, bilateral, resolves │
│ Effusion (OME) │ with decongestants │
├─────────────────────┼───────────────────────────────────────────┤
│ Glomus Tympanicum │ Pulsatile, red/pink, posterior-inferior │
├─────────────────────┼───────────────────────────────────────────┤
│ Aberrant ICA │ Pulsatile, do NOT biopsy, CT angiography │
├─────────────────────┼───────────────────────────────────────────┤
│ High Jugular Bulb │ Blue mass, inferior quadrant │
├─────────────────────┼───────────────────────────────────────────┤
│ Tympanosclerosis │ History of prior OM/surgery, chalky white │
├─────────────────────┼───────────────────────────────────────────┤
│ Facial Nerve │ Along CN VII course, enhances on MRI │
│ Schwannoma │ │
└─────────────────────┴───────────────────────────────────────────┘
11. MANAGEMENT
11.1 Overview
The only definitive treatment for congenital cholesteatoma is surgical excision. There is no medical treatment.
MANAGEMENT FLOWCHART — CONGENITAL CHOLESTEATOMA
Diagnosis Confirmed
(White mass + Intact TM + No prior OM/surgery)
│
↓
STAGING (Potsic system: HRCT + MRI)
│
├──────────────────┬─────────────────┬──────────────┐
│ │ │ │
Stage I Stage II Stage III Stage IV
│ │ │ │
Transcanal Transcanal/ Mastoid + Canal Wall
approach; Postauricular; Posterior Down (CWD) OR
Myringotomy + Tympanoplasty Tympanotomy; Canal Wall Up
Excision Ossiculoplasty (CWU) +
if needed Second-Look
│
↓
COMPLETE EXCISION (PRIMARY GOAL)
│
↓
HEARING RECONSTRUCTION
(Ossiculoplasty / TORP / PORP as required)
│
↓
SECOND-LOOK SURGERY (if Stage III/IV)
OR
MRI NON-EPI DWI at 12–18 months post-op
11.2 Surgical Approaches
A. Transcanal (Endaural/Endoscopic) Approach
- For early-stage CC (Stage I, small Stage II)
- Tympanomeatal flap raised; CC excised completely
- Increasingly performed endoscopically (TEES — Transcanal Endoscopic Ear Surgery)
B. Postauricular (Retroauricular) Approach
- For larger Stage II and Stage III lesions
- Better visualisation of the entire middle ear
C. Canal Wall Up (CWU) Mastoidectomy
- Stage III–IV; preserves anatomy
- Higher residual/recurrence rate — second-look surgery mandatory at 12–18 months
D. Canal Wall Down (CWD) / Modified Radical Mastoidectomy
- Extensive Stage IV with large-scale destruction
- Lower recurrence but meatoplasty needed; cavity care required
11.3 Ossiculoplasty
- Intact ossicular chain: Preserve during excision
- Incus erosion: PORP (Partial Ossicular Replacement Prosthesis) — connects stapes to malleus handle
- Stapes superstructure absent: TORP (Total Ossicular Replacement Prosthesis)
- Materials: Autologous cartilage/cortical bone or titanium/hydroxyapatite prostheses
11.4 Second-Look Surgery vs. MRI Surveillance
SECOND-LOOK / SURVEILLANCE ALGORITHM
Post-operative (Stage III/IV)
│
↓
MRI Non-EPI DWI at 12–18 months
│
┌────┴────┐
DWI +ve DWI -ve
│ │
Second-look Annual clinical
surgery review + repeat
(Revision MRI at 3 years
tympanomastoid)
This non-EPI DWI protocol has largely replaced routine second-look surgery in Stage I–II CC in many centres.
12. COMPLICATIONS
COMPLICATIONS OF UNTREATED / ADVANCED CONGENITAL CHOLESTEATOMA
INTRATEMPORAL INTRACRANIAL
───────────────────── ──────────────────────────
• Conductive hearing loss • Extradural abscess
• Sensorineural hearing loss • Subdural abscess
(labyrinthine fistula) • Meningitis
• Facial nerve palsy • Brain abscess
(canal dehiscence) • Lateral sinus thrombophlebitis
• Labyrinthitis • Otitic hydrocephalus
• Mastoiditis
• Petrous apicitis
(Gradenigo's syndrome)
13. COMPARISON WITH ACQUIRED CHOLESTEATOMA
┌────────────────────┬──────────────────────────┬──────────────────────────┐
│ FEATURE │ CONGENITAL │ ACQUIRED │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Age │ 3–5 years │ 10–15 years (pedi); │
│ │ │ any age (adult) │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ TM integrity │ INTACT (by definition) │ Perforated / retraction │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Prior OM/otorrhoea │ Absent │ Usually present │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Location │ Anterior-superior ME │ Pars flaccida (attic) or │
│ │ │ pars tensa (posterior) │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Pathogenesis │ Failure of EF involution │ Retraction, basal cell │
│ │ (Michaels' theory) │ hyperplasia, metaplasia │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Macroscopy │ Pearl-like, well-defined │ Irregular, often with │
│ │ cyst │ granulations / discharge │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Presentation │ Often incidental / CHL │ Foul-smelling discharge, │
│ │ │ CHL, pain │
├────────────────────┼──────────────────────────┼──────────────────────────┤
│ Prognosis │ Better (if early stage) │ Higher recurrence rate │
└────────────────────┴──────────────────────────┴──────────────────────────┘
14. RECENT ADVANCES
14.1 Endoscopic Ear Surgery (TEES)
- Transcanal endoscopic approach with zero-degree and 45-degree endoscopes
- Advantages: Single port, improved visualisation of anterior recesses (sinus tympani, facial recess), no postauricular scar
- Increasingly preferred for Stage I–II CC
- Allows complete excision without canal wall down surgery in early stages
- Recent literature: Badr-el-Dine M; Marchioni D, Presutti L
14.2 Non-Echo Planar DWI MRI (Non-EPI DWI)
- Has revolutionised post-operative surveillance
- High sensitivity (>90%) and specificity for detecting residual keratin as small as 3–5 mm
- PROPELLER/BLADE DWI sequences further improve accuracy
- May predict need for second-look surgery without surgical exploration
- Dubrulle F et al.; Lingam RK et al.
14.3 Molecular Pathogenesis
- RANKL/OPG ratio — elevated in CC; drives osteoclast activation
- MMP-2 and MMP-9 — major enzymes responsible for ossicular erosion
- Ki-67 index — CC has lower proliferative index than acquired cholesteatoma
- Notch signalling pathway — implicated in epithelial hyperproliferation
- Potential future target for medical/biological therapy to arrest growth
14.4 Genetic Research
- Reports of familial CC and bilateral CC in identical twins support a genetic susceptibility
- Genome-wide association studies (GWAS) ongoing — no confirmed mutation yet
14.5 Otoendoscopy (Office-based)
- High-definition otoscopy/otoendoscopy improves early detection of small CC
- Leukocoria of the ear analogy — white reflex behind TM as a screening finding
14.6 Robot-Assisted Ear Surgery
- Experimental robotic platforms (e.g., RobOtol) are being evaluated for minimally invasive middle ear surgery — potential future application for CC
14.7 PROPELLER/HASTE MRI Protocols
- Latest non-EPI DWI protocols with sub-millimetre resolution detect even tiny residual pearls
- Reduces the need for routine second-look surgery in carefully selected cases
15. PERSPECTIVES FROM MAJOR TEXTBOOKS
Scott Brown's Otorhinolaryngology Head & Neck Surgery
- Emphasises that CC arises from persistence of embryological epidermoid cell rests (Michaels' EF)
- Describes the diagnostic triad: white pearly mass + intact TM + no prior OM
- Recommends CWU approach for most cases with planned second-look at 12–18 months
- Stresses importance of complete matrix removal — leaving matrix behind is the most common cause of recurrence
Cummings Otolaryngology Head and Neck Surgery
- States: "Acquired cholesteatoma is much more common than congenital cholesteatoma, even in children"
- Documents Potsic staging and its correlation with residual disease risk
- Notes smaller mastoid volume in CC patients compared to controls — suggests a shared developmental predisposition
- Emphasises that non-EPI DWI MRI has transformed follow-up protocols
- References bilateral CC in twins as evidence for potential genetic basis
Stell & Maran's Head & Neck Surgery
- Describes CC as arising from sequestered ectodermal inclusions during embryological development
- Emphasises that surgical timing is important — early surgery prevents bone destruction and preserves hearing
- Documents the "inside-out" spread pattern — CC expands concentrically from its origin
Dhingra's Diseases of Ear, Nose & Throat
- Classic Indian ENT reference widely used in RGUHS curriculum
- Defines CC with Levenson's criteria
- Lists Michaels' EF theory as the most accepted
- Describes otoscopic appearance as a "white pearl" — distinguishing CC from OME
- Stages: Anterior-superior (most common) → posterior → ossicular involvement → mastoid
Hazarika's Textbook of Ear, Nose & Throat
- Emphasises CC in the Indian context — often presents late due to diagnostic delay
- Notes that CHL in a child with an intact TM should always raise suspicion of CC
- Discusses the role of HRCT temporal bone as the primary investigation
- Advocates CWU mastoidectomy with second-look for advanced cases
Zakir Hussain (ENT Reference)
- Documents CC as a keratoma with no cholesterol or fat content despite its name
- Stresses histological features: stratified squamous epithelium + fibrous perimatrix + keratinous content
- Includes CC in differential diagnosis of conductive hearing loss in children
- Advocates that all intact TM with CHL in children must be investigated with HRCT
KJ Lee's Essential Otolaryngology
- Lists CC under pediatric otology
- Notes anterior-superior quadrant predilection based on Michaels' fetal temporal bone studies
- Provides surgical decision-making guidance based on extent of disease
16. SUMMARY FLOWCHART — COMPLETE OVERVIEW
╔═══════════════════════════════════════════════════════════════════╗
║ CONGENITAL CHOLESTEATOMA — MASTER FLOWCHART ║
╠═══════════════════════════════════════════════════════════════════╣
║ ║
║ EMBRYOLOGY ║
║ Failure of involution of Michaels' Epidermoid Formation (EF) ║
║ at 10-33 weeks gestation (anterior-superior tympanum) ║
║ │ ║
║ ↓ ║
║ PATHOLOGY ║
║ Keratinizing squamous cyst → Bone erosion via MMPs/RANKL ║
║ │ ║
║ ↓ ║
║ CLINICAL ║
║ Child, male, age 3-5 yrs; white pearly mass, intact TM ║
║ CHL (most common symptom) | No otorrhoea | No prior OM ║
║ │ ║
║ ↓ ║
║ INVESTIGATIONS ║
║ PTA (CHL) → HRCT temporal bone → MRI Non-EPI DWI ║
║ │ ║
║ ↓ ║
║ STAGING (Potsic 2002) ║
║ Stage I → II → III → IV (67% residual risk) ║
║ │ ║
║ ↓ ║
║ SURGERY (ONLY TREATMENT) ║
║ Transcanal/Endoscopic (early) | CWU/CWD Mastoidectomy (late) ║
║ + Ossiculoplasty (if needed) ║
║ │ ║
║ ↓ ║
║ SURVEILLANCE ║
║ MRI Non-EPI DWI at 12-18 months ║
║ +/- Second-look surgery (Stage III/IV or DWI positive) ║
║ │ ║
║ ↓ ║
║ GOAL: Dry, hearing ear; prevent complications ║
╚═══════════════════════════════════════════════════════════════════╝
17. CONCLUSION
Congenital cholesteatoma is a benign but destructive lesion of the temporal bone arising from embryological epithelial cell rests (Michaels' EF) that fail to involute. It presents classically in young boys as a white pearly mass behind an intact tympanic membrane with conductive hearing loss and no prior otological history. The Potsic staging system (2002) guides surgical planning and predicts residual disease risk. Complete surgical excision remains the only treatment, with endoscopic ear surgery now being the preferred approach for early-stage disease. MRI Non-EPI DWI has transformed post-operative surveillance, largely replacing mandatory second-look surgery in early stages. Research into molecular mediators of bone erosion (MMPs, RANKL pathway) holds promise for future adjuvant biological therapies.
Key References
- Michaels L. An epidermoid formation in the developing middle ear: possible source of cholesteatoma. J Otolaryngol. 1986;15(3):169-174.
- Levenson MJ et al. Congenital cholesteatoma. Arch Otolaryngol. 1989;115(1):547-553.
- Potsic WP et al. A staging system for congenital cholesteatoma. Arch Otolaryngol Head Neck Surg. 2002;128(9):1009–1012.
- Cummings Otolaryngology Head and Neck Surgery, 7th ed. Cummings CW et al. Elsevier.
- Dhingra PL. Diseases of Ear, Nose and Throat. 7th ed. Elsevier.
- Hazarika P et al. Textbook of ENT & Head Neck Surgery.
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery, 8th ed. Gleeson M et al.
- Stell & Maran's Head and Neck Surgery.
- Marchioni D et al. Bilateral congenital cholesteatoma. IJPO. 2017;99:146–151.
- Huisman MA et al. MMP and RANKL in cholesteatoma. Otol Neurotol. 2008.
- Lingam RK et al. Non-echo-planar DWI MRI for cholesteatoma surveillance. Clin Radiol. 2013.
Note on image generation: Thegenerate_imagetool (for custom diagrams) has reached its monthly plan limit. The flowcharts above are provided as text-based ASCII diagrams. To access additional generated diagrams, please upgrade your subscription plan. The clinical photographs from the medical literature (Figs. 1–3 above) are sourced from peer-reviewed publications and illustrate all key features discussed.
This is a shared conversation. Sign in to Orris to start your own chat.