Fesoterodine in oAB
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Loading Image
Here is a comprehensive overview of fesoterodine in overactive bladder (OAB):
Fesoterodine in Overactive Bladder (OAB)
Mechanism of Action
Fesoterodine is a prodrug — after oral administration, it is rapidly and completely hydrolyzed by non-specific esterases to its active metabolite 5-hydroxymethyl tolterodine (5-HMT). The parent compound is undetectable in plasma after oral dosing. 5-HMT is a competitive muscarinic receptor antagonist that blocks M2 and M3 receptors in the detrusor muscle, reducing involuntary contractions. Unlike tolterodine (which is converted to 5-HMT via CYP2D6), fesoterodine's conversion is enzyme-independent, making it more predictable and consistent across patients regardless of CYP2D6 metabolizer status.
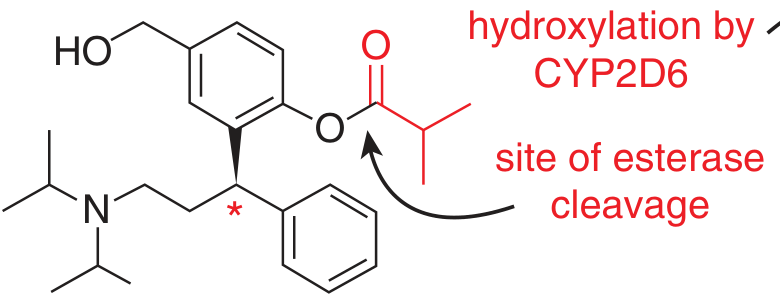
Fesoterodine (prodrug) — the ester bond is cleaved by non-specific plasma esterases to yield the active metabolite 5-HMT. CYP2D6 converts tolterodine to the same metabolite. (Goodman & Gilman, Fig. 11–4)
Classification
Fesoterodine belongs to the "pure" antimuscarinic category among OAB drugs (alongside darifenacin, solifenacin, and tolterodine), meaning it has no additional pharmacological actions beyond muscarinic antagonism.
Pharmacokinetics
| Parameter | Detail |
|---|---|
| Prodrug conversion | Non-specific plasma esterases → 5-HMT |
| CYP involvement | 5-HMT further metabolized by CYP2D6 and CYP3A4 in the liver |
| Dosing | 4 mg or 8 mg once daily (flexible dosing) |
| Half-life | ~7 hours (5-HMT) |
| Renal excretion | ~70% of dose excreted renally |
A significant portion of 5-HMT is excreted unchanged in urine, giving some direct effect at the bladder urothelium.
Clinical Efficacy
Phase III RCTs
Multiple RCTs have documented efficacy of fesoterodine 4 mg and 8 mg vs. placebo:
- Significant reductions in urgency urinary incontinence (UUI) episodes, micturition frequency, urgency episodes, and nocturia
- Improvements in Patient Perception of Bladder Condition (PPBC), Urgency Perception Scale (UPS), and OAB-q scores
Dose–Response
- Fesoterodine 8 mg > 4 mg for most efficacy endpoints; dose-titration from 4 mg to 8 mg is well-supported
- A pooled analysis of two Phase III trials confirmed superiority of fesoterodine 8 mg over tolterodine ER 4 mg across multiple OAB endpoints (Ginsberg et al., 2013)
- In women, fesoterodine 8 mg was superior to tolterodine 4 mg at 12 weeks in UUI episodes and other patient-reported outcomes
Special Populations
- Elderly (≥65 years): Confirmed efficacy and acceptable tolerability; fesoterodine is considered an option but anticholinergic burden in the elderly must be assessed
- Post-marketing (Korea, Kim et al., 2016): Large real-world study confirmed OAB efficacy; notably 63% of patients had symptom duration <1 week
Tolerability & Side Effects
| Side Effect | Frequency |
|---|---|
| Dry mouth | Most common; dose-dependent (higher at 8 mg) |
| Constipation | Common |
| Blurred vision | Less common |
| Urinary retention | Rare; risk higher in men with BOO |
| CNS effects | Low CNS penetration due to hydrophilicity of 5-HMT |
- Dry mouth and constipation are the principal reasons for discontinuation
- The peripheral selectivity of 5-HMT limits CNS adverse effects compared to older antimuscarinics (e.g., oxybutynin)
Comparison With Tolterodine
| Feature | Fesoterodine | Tolterodine ER |
|---|---|---|
| Conversion to 5-HMT | Non-specific esterases (predictable) | CYP2D6 (variable — poor vs. extensive metabolizers) |
| Bioavailability of active metabolite | More consistent across patients | Variable |
| Efficacy (8 mg vs. 4 mg ER) | Fesoterodine 8 mg superior | Reference |
| Tolerability | Similar dry mouth profile | Similar |
Because conversion to 5-HMT bypasses CYP2D6, fesoterodine avoids drug–drug interactions at that step and produces more uniform plasma levels of the active metabolite.
Place in Therapy
- First-line pharmacotherapy for OAB (alongside solifenacin, tolterodine, oxybutynin, mirabegron, vibegron)
- Preferred over older non-selective agents (oxybutynin) where cognitive or dry mouth side effects are a concern
- Flexible dosing (4 → 8 mg) allows dose escalation based on response and tolerability
- In combination approaches (e.g., with alpha-blockers in men with BPH+OAB), fesoterodine is listed among the approved antimuscarinic agents
Sources: Campbell Walsh Wein Urology (3-Volume Set) — Antimuscarinics With "Pure" Action; Goodman & Gilman's The Pharmacological Basis of Therapeutics, Fig. 11–4
This is a shared conversation. Sign in to Orris to start your own chat.