Proper Evidence based articles for collagen supplements and it’s uses and is it essential
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Here is a comprehensive, evidence-based review of collagen — what it is, how it works, and what the science actually says about supplementation.
Collagen: Biology, Supplementation & Clinical Evidence
1. What is Collagen?
Collagen is the most abundant protein in the human body, constituting approximately 30% of its dry weight. It is the major structural component of the extracellular matrix (ECM) and is found in:
- Skin, tendons, and ligaments
- Bone and cartilage
- Blood vessel walls
- Cornea, fascia, organ capsules
A family of 28 distinct collagen types exists in vertebrates. The most clinically relevant are:
| Type | Location | Function |
|---|---|---|
| Type I | Skin, tendon, bone, dentin | Resistance to tension (most abundant) |
| Type II | Cartilage, vitreous body | Resistance to pressure |
| Type III | Blood vessels, skin, GI tract | Structural support, wound healing |
— Junqueira's Basic Histology, 17e & Basic Medical Biochemistry, 6e
2. Collagen Biochemistry: The Triple Helix
Collagen polypeptide chains consist of repeating (Gly-X-Y) sequences. Every third residue must be glycine (the smallest amino acid) to fit the tight center of the triple helix. The X and Y positions are frequently proline and hydroxyproline, respectively.
Key biosynthesis steps:
- Pro-α chains synthesized on ribosomes → enter the endoplasmic reticulum
- Proline and lysine residues are hydroxylated → forming hydroxyproline & hydroxylysine (essential for helix stability)
- Three pro-α chains coil → procollagen (triple helix)
- Secreted into ECM → propeptides cleaved → collagen monomers self-assemble into fibrils
- Lysyl oxidase cross-links lysine residues → tensile strength
— Sabiston Textbook of Surgery, 21e; Basic Medical Biochemistry, 6e
3. The Critical Role of Vitamin C in Collagen Synthesis
This is the most well-established nutritional cofactor in collagen biology:
"Vitamin C (ascorbic acid) is required for the conversion of proline and lysine to hydroxyproline and hydroxylysine, respectively. Vitamin C deficiency [scurvy] leads to a defect in wound healing, particularly via a failure in collagen synthesis and cross-linking." — Schwartz's Principles of Surgery, 11e
Without vitamin C:
- Prolyl and lysyl hydroxylase enzymes cannot function
- Collagen melting temperature drops from 42°C → 24°C (structurally unstable at body temperature)
- Blood vessels become fragile, teeth loosen, wounds fail to heal
Recommended intake: 60 mg/day (RDA) for healthy adults. In severely injured or burned patients, requirements may increase to 2 g/day. However, there is no evidence that supratherapeutic doses beyond what corrects deficiency are beneficial. — Schwartz's Principles of Surgery, 11e
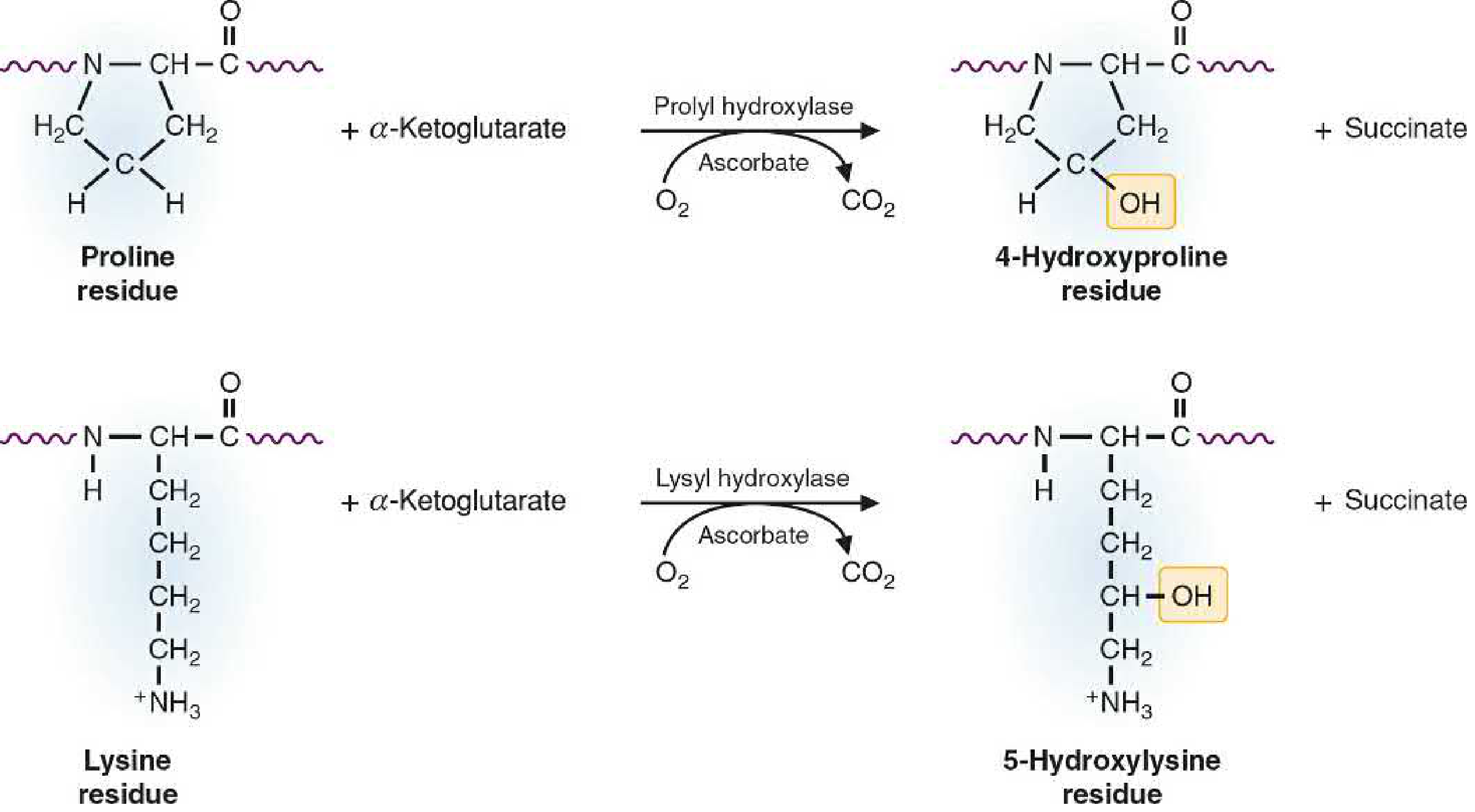
4. What the Evidence Actually Says About Oral Collagen Supplementation
This is where the evidence base diverges significantly from popular claims.
4a. The Absorption Problem
Textbooks do not support the notion that oral collagen peptides are absorbed intact and directed to specific tissues. Dietary collagen — like all dietary proteins — is digested into individual amino acids and di/tripeptides in the GI tract before absorption. There is no established physiologic mechanism by which ingested collagen preferentially "rebuilds" cartilage, skin, or joints.
The key bioavailability concern is noted in clinical nutrition literature: hydroxyproline urinary excretion increases with ingestion of meat or gelatin — meaning collagen-derived amino acids are systemically detectable — but this does not confirm targeted tissue deposition. — Tietz Textbook of Laboratory Medicine, 7th ed
4b. Oral Type II Collagen in Autoimmune/Arthritis Research
One specific application with immunological plausibility is oral tolerance induction using type II collagen in autoimmune arthritis:
"Oral administration of MBP [myelin basic protein] or type II collagen inhibits the development of these diseases in animal models." — Janeway's Immunobiology, 10e
This mechanism involves the gut-associated lymphoid tissue inducing regulatory T-cell tolerance to collagen antigens — a distinct mechanism from tissue "building." This approach has been studied in rheumatoid arthritis clinical trials with modest results, but it is not the same as the mechanism marketed by commercial supplements.
4c. Where Collagen Nutrition IS Evidence-Based: Deficiency Correction
The evidence firmly supports that collagen synthesis is impaired under deficiency states, and correcting those deficiencies restores function:
| Deficiency | Effect on Collagen | Evidence Level |
|---|---|---|
| Vitamin C | Impaired proline/lysine hydroxylation → scurvy | Very high (established) |
| Zinc | Decreased fibroblast proliferation, decreased collagen synthesis, impaired wound strength — reversed by zinc supplementation in zinc-deficient patients only | High |
| Copper | Required for lysyl oxidase (collagen cross-linking) — deficiency causes abnormal collagen cross-linking and muscle weakness | High |
| Arginine | Supplementation (17–25 g/day) shown to significantly increase wound collagen deposition in both young and older healthy volunteers | Moderate (RCTs in wound healing) |
"No study has shown improved wound healing with zinc supplementation in patients who are not zinc deficient." — Schwartz's Principles of Surgery, 11e
"Daily supplements of 30 g of arginine aspartate for 14 days resulted in significantly enhanced collagen and total protein deposition at the wound site when compared to controls." — Schwartz's Principles of Surgery, 11e
4d. Collagen Injectable Fillers (Established Clinical Use)
In dermatology, bovine and human collagen are used as injectable fillers for soft tissue augmentation — this is direct tissue placement, not oral supplementation, and has an established evidence base. — Andrews' Diseases of the Skin, Clinical Dermatology
5. Collagen and Aging/Skin
From the physiological standpoint:
- With aging, collagen cross-linking changes and the skin thins
- Articular cartilage loses proteoglycan and collagen content, contributing to osteoarthritis
- Joint flexibility falls due to aging of articular cartilage — Medical Physiology, Boron & Boulpaep
Commercial "anti-aging collagen supplements" target these changes, but the textbook literature does not contain evidence-based support for oral collagen supplementation reversing skin aging or cartilage loss. Topical retinoids and sun protection have stronger evidence for skin collagen maintenance.
6. Genetic Conditions Affecting Collagen (Not Supplement-Responsive)
Several serious conditions arise from defective collagen genes — oral supplementation cannot correct these:
| Condition | Mechanism |
|---|---|
| Osteogenesis Imperfecta | Deletion of procollagen α1 allele → brittle bones |
| Ehlers-Danlos Syndrome | Mutations in type III collagen → fragile skin, blood vessels, hypermobile joints |
| Scurvy | Nutritional vitamin C deficiency → unstable collagen triple helix |
— Sabiston Textbook of Surgery, 21e
Summary: Is Collagen Supplementation "Essential"?
| Question | Evidence-Based Answer |
|---|---|
| Is collagen essential for the body? | Yes — it is the most abundant structural protein |
| Is dietary collagen essential? | No — the body synthesizes collagen from amino acids (glycine, proline, hydroxyproline) and vitamin C. It is a non-essential nutrient |
| Does vitamin C supplementation support collagen synthesis? | Yes, when deficient. Well-established biochemistry |
| Do oral collagen supplements build tissue? | Unproven — not supported by major medical textbooks; plausible mechanistically in some contexts (e.g., oral tolerance) |
| Does arginine supplementation improve collagen deposition? | Yes, in wound healing contexts — RCT evidence in healthy volunteers |
| Are collagen supplements harmful? | Generally considered safe; zinc excess can paradoxically cause copper deficiency and impair cross-linking |
Bottom line: Collagen is biochemically essential, but your body synthesizes it endogenously. The strongest nutritional interventions for collagen support are adequate vitamin C, zinc, and copper — correcting deficiencies has proven effects. Oral collagen hydrolysate supplements have a plausible absorption pathway but lack robust textbook-level clinical evidence comparable to these established cofactors. The marketing of collagen supplements far outpaces the published evidence base.
This is a shared conversation. Sign in to Orris to start your own chat.