Q. Discuss the principle of otocoustic emissions in detail for 50 marks with diagram and flow charts from Scott Brown, Cummings, stell and marans, Zakir Hussain, dhingra, hazarika and related articles with recent advances according to rguhs university
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Searching Images
otoacoustic emissions cochlear outer hair cell OAE diagram
Reading File
Reading File
Loading Image
Finding Sources
Searching Images
TEOAE DPOAE waveform recording newborn hearing screening
Loading Image
Loading Image
I now have comprehensive material from the textbook library to write a detailed 50-mark answer. Let me compose the full answer.
OTOACOUSTIC EMISSIONS (OAEs)
A Comprehensive Answer for 50 Marks — RGUHS ENT Postgraduate Examination
INTRODUCTION & HISTORICAL BACKGROUND
Otoacoustic emissions (OAEs) are low-level sounds generated by the cochlea — specifically by the active mechanical processes of the outer hair cells (OHCs) — that can be recorded by a sensitive microphone placed in the sealed external auditory canal. They represent a window into the functional integrity of the cochlea without requiring any behavioral response from the patient.
Historical milestones:
- 1948 — Thomas Gold theoretically predicted that the cochlea must possess an active feedback mechanism to explain its sharp frequency tuning.
- 1978 — David Kemp (London) first demonstrated the clinical existence of evoked OAEs using a click stimulus and a sensitive microphone — this was a landmark discovery that validated the concept of the cochlear amplifier.
- 1980s — The role of outer hair cell electromotility and the protein prestin were elucidated.
- 2000s onward — OAEs became the standard of care for Universal Newborn Hearing Screening (UNHS) programs worldwide.
"With the discovery of the evoked otoacoustic emissions, it became possible to analyze and investigate auditory function from hair cells to the auditory cortex." — Cummings Otolaryngology Head and Neck Surgery
BASIC PRINCIPLE — THE COCHLEAR AMPLIFIER
Anatomy Relevant to OAEs
The organ of Corti contains two types of hair cells:
- Inner hair cells (IHCs) — ~3,500 in number; the true sensory transducers; heavily innervated by afferent neurons (~95% of spiral ganglion neurons).
- Outer hair cells (OHCs) — ~12,000 in number; arranged in three rows; sparsely innervated; primarily act as mechanical amplifiers.
Mechanism of OHC Electromotility
The key molecular motor is prestin — a motor protein embedded in the lateral wall of OHCs. It belongs to the SLC26A5 anion transporter family. When the OHC depolarizes (basilar membrane moves upward → stereocilia deflect toward tallest row → mechanosensitive channels open → K⁺ and Ca²⁺ influx), prestin contracts and the OHC shortens. When the OHC hyperpolarizes (basilar membrane moves downward), the OHC elongates. These rapid length changes — at audio frequencies up to 20,000 Hz — amplify basilar membrane motion approximately 100-fold (40–50 dB of amplification).
"The contraction of myriad prestin molecules — each attached to its neighbors — causes the outer hair cell to contract; this phenomenon is unique to outer hair cells and is called electromotility." — Medical Physiology (Boron & Boulpaep)
Generation of OAEs
When OHCs amplify basilar membrane motion, mechanical energy propagates in two directions:
- Forward (anterograde) — toward the apex → transduction by IHCs → auditory nerve stimulation (normal hearing).
- Backward (retrograde) — from OHC → basilar membrane vibration → perilymph pressure waves → oval window → ossicular chain → tympanic membrane (acts as a loudspeaker) → detectable sound in the ear canal.
This retrograde energy is the OAE. It is an epiphenomenon — not part of the hearing process itself, but a by-product of the cochlear amplifier.
"In otoacoustic emission, the system works in reverse as the basilar membrane causes pressure waves in the cochlear fluids, which vibrate the oval window, ossicles, and tympanic membrane, and finally generates new pressure waves in the air of the auditory canal." — Medical Physiology
PATHWAY OF ENERGY TRANSFER (Flow Chart)
OUTER HAIR CELL (OHC) ELECTROMOTILITY
↓
Basilar Membrane Vibration
↓
Cochlear Fluid (Perilymph) Pressure Waves
↓
Oval Window
↓
Ossicular Chain (Stapes → Incus → Malleus)
↓
Tympanic Membrane (acts as loudspeaker)
↓
External Auditory Canal
↓
Detected by Sensitive Microphone in Probe
↓
Signal Amplification, Filtering & Computer Averaging
↓
OAE RECORDING
(K J Lee's Essential Otolaryngology, p. 313)
SITE OF OAE TESTING IN THE AUDITORY PATHWAY
OAEs test cochlear (OHC) function only. The diagram below illustrates the level assessed by each auditory test:
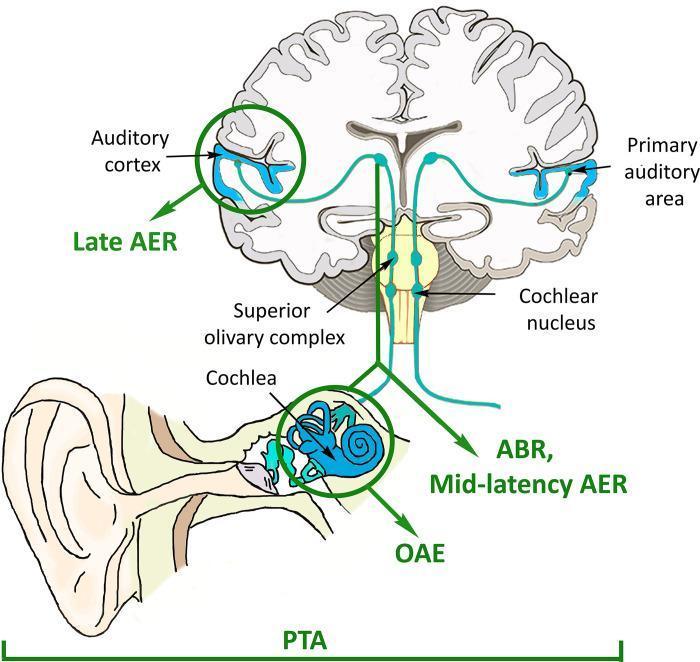
Figure: OAE localizes to the cochlea (OHC level). ABR assesses brainstem pathways. Adapted from clinical audiology literature.
CLASSIFICATION OF OAEs
OAEs are classified into two broad groups:
A. Spontaneous OAEs (SOAEs)
- Present without any external acoustic stimulus
- Occur in 35–60% of normally hearing individuals
- More common in females and in the right ear
- Present as narrow-band signals in the frequency range 0.5–6 kHz
- Clinical significance: Their presence confirms good OHC function; their absence is non-diagnostic (absent even in many normal ears)
- On rare occasions, damaged ears produce SOAEs loud enough to be heard by a nearby listener
- Rarely used clinically
B. Evoked OAEs (EOAEs)
Generated in response to an acoustic stimulus. Three subtypes:
i. Transient Evoked OAEs (TEOAEs)
- Elicited by brief transient stimuli (clicks or brief tone bursts)
- Stimulus: clicks at ~80 dB SPL
- Response: delayed echo appearing 5–20 ms after stimulus
- Frequency range: 0.4–6 kHz
- Amplitude decreases as frequency increases
- Absent in cochlear SNHL > 30–40 dB HL
- Can be analyzed octave-by-octave for frequency-specific information (present/absent only)
- Uses computer averaging to enhance signal-to-noise ratio
- Present = cochlear sensitivity ≥ 20–40 dB HL in 500–4000 Hz range
ii. Stimulus-Frequency OAEs (SFOAEs)
- Elicited by a continuous pure tone
- Least commonly used clinically
- Difficult to separate from the stimulus
iii. Distortion Product OAEs (DPOAEs)
- Elicited by two simultaneous pure tones (f₁ and f₂)
- The nonlinear cochlea generates a distortion product at a mathematically predictable frequency
- The most prominent DPOAE in the human ear: 2f₁ − f₂ (cubic difference tone)
- Example: f₁ = 1000 Hz, f₂ = 1220 Hz → DPOAE at 780 Hz
- f₁:f₂ ratio optimally 1:1.22
- Stimulus levels: 55–85 dB SPL; f₁ 10–15 dB louder than f₂ is optimal
- DPOAE amplitude typically 60 dB lower than the primary tones
- Provides frequency-specific cochlear map (DP-gram)
- Can be recorded in some ears even with moderate-to-severe SNHL (stimulus level dependent)
- Best tool for ototoxicity monitoring and noise-induced hearing loss detection
"DPOAEs provide functional information about well-defined frequency-specific regions of the cochlea, thereby facilitating the tracking of change in inner-ear pathology characterized by outer hair cell dysfunction." — Cummings Otolaryngology
FLOW CHART: CLASSIFICATION OF OAEs
OTOACOUSTIC EMISSIONS
│
┌────┴─────────┐
Spontaneous Evoked (EOAEs)
(SOAEs) │
┌──────┼──────────┐
TEOAE SFOAE DPOAE
(click) (pure (2 tones:
tone) 2f₁−f₂)
RECORDING TECHNIQUE & EQUIPMENT
Probe Assembly
A small hermetically sealed probe is placed in the external auditory canal. It contains:
- A miniature loudspeaker (or two, for DPOAEs) — delivers the acoustic stimulus
- A sensitive microphone — picks up the emission
- A rubber tip — creates an airtight seal (crucial)
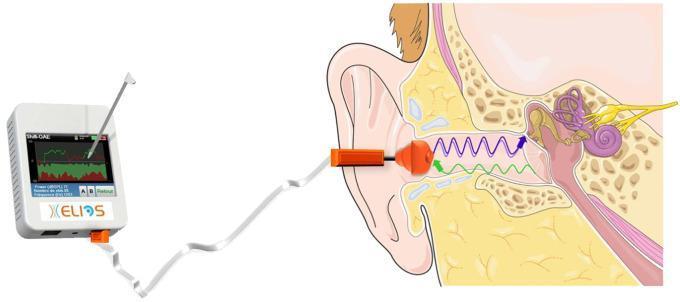
Figure: DPOAE recording. Blue/purple waves = stimulus; green waves = OAE returning to the microphone.
Signal Processing
- The stimulus and emission overlap in time → computer averaging (usually 260–1000 sweeps) is used to separate the emission from background noise
- For TEOAEs: time-windowed gating (response measured 5–20 ms post-stimulus) eliminates the stimulus artifact
- For DPOAEs: frequency-specific analysis separates the DPOAE (at 2f₁−f₂) from stimuli (at f₁ and f₂) using Fast Fourier Transform (FFT)
- Signal-to-noise ratio (SNR): OAE is considered present when response exceeds noise floor by ≥ 6 dB
- Reproducibility (for TEOAEs): ≥ 70% reproducibility indicates a valid response; ≥ 50% per frequency band = present
Prerequisites for Valid Recording
- Quiet environment (ambient noise < 40 dB SPL)
- Patent ear canal (no cerumen, no debris)
- Normal middle ear function (type A tympanogram; no effusion, no perforation)
- Patient must be relatively still and calm — sedation rarely needed (test takes 5–10 min per ear)
OAE RECORDINGS — INTERPRETATION
Normal TEOAE
- Response waveform clearly rises above noise floor
- Reproducibility > 70% overall; > 50% per octave band
Normal DPOAE (DP-gram)
- DPOAE amplitudes plotted against f₂ frequency
- Normal: DPOAE level > −5 to 0 dB SPL; SNR > 6 dB
- Upper and lower bounds of normal shown by dashed lines (± 1 SD normative data)
Abnormal OAE
- Flat response (indistinguishable from noise floor)
- Indicates OHC dysfunction at that frequency region
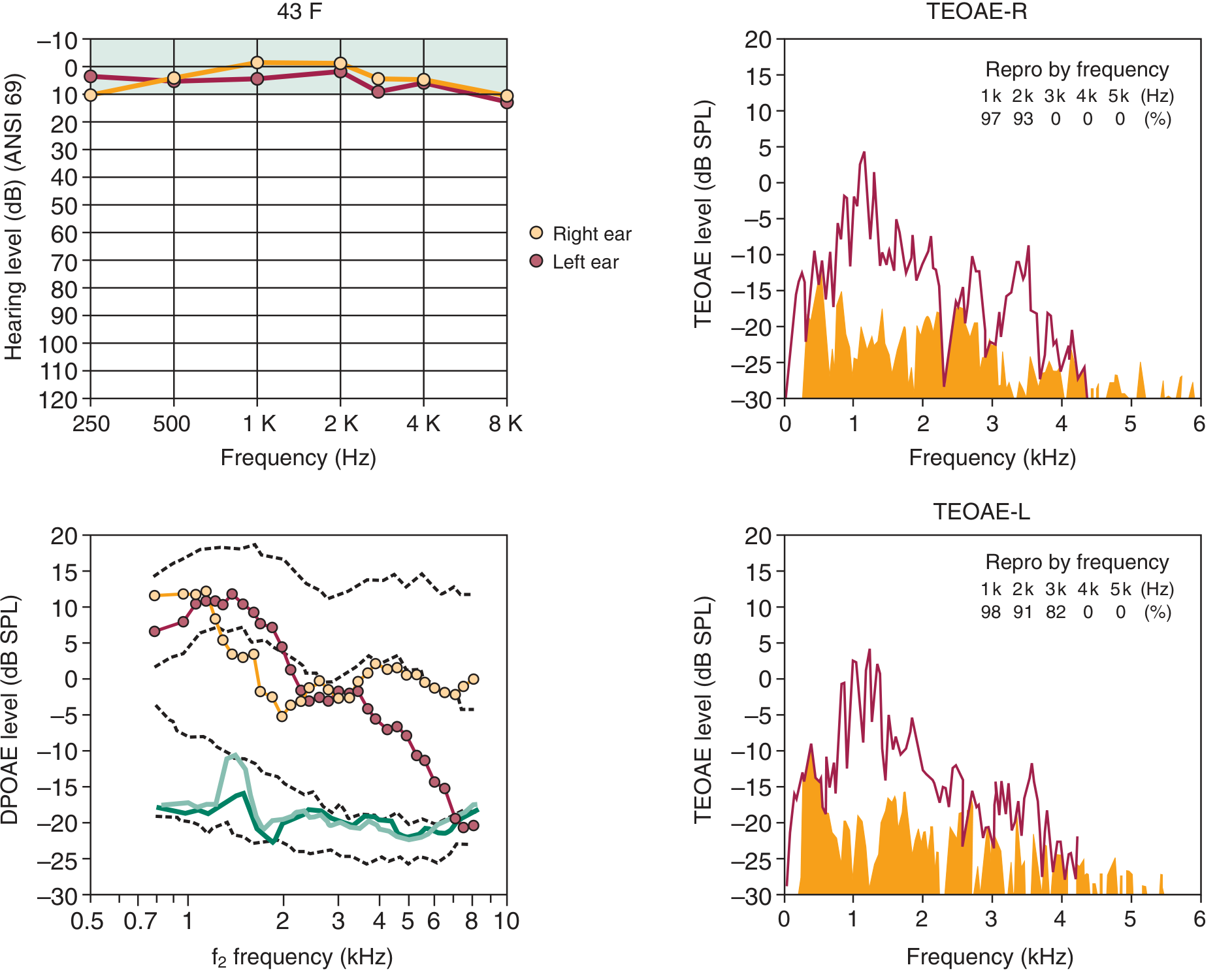
Fig. (Cummings): Normal audiogram (top left), DPOAE plot (bottom left) and TEOAE spectra (right) from a 43-year-old rifle shooter. Despite normal pure tone audiogram, DPOAE and TEOAE show loss above 3 kHz in the left ear — demonstrating subclinical OHC damage. OAE detects OHC dysfunction before it appears on audiogram.
FACTORS AFFECTING OAEs
| Factor | Effect on OAE |
|---|---|
| Conductive hearing loss | Absent/reduced — blocks both incoming stimulus and outgoing emission |
| Sensorineural HL > 35–40 dB | Absent (TEOAE); may be reduced (DPOAE) |
| Middle ear effusion (OME) | Absent |
| Excessive cerumen | Absent/reduced |
| Background noise | Reduced SNR → spurious absent result |
| Age | Amplitude decreases with increasing age |
| Contralateral noise | Suppresses OAEs (medial olivocochlear reflex) |
| Ototoxic drugs | Progressive reduction |
| Noise exposure | Progressive reduction at affected frequencies |
| Retrocochlear lesion | OAEs remain normal (only cochlear function tested) |
CLINICAL APPLICATIONS
1. Universal Newborn Hearing Screening (UNHS)
The most important application. OAEs are fast, non-invasive, and require no behavioral response.
Screening protocol:
Birth → OAE Screening (before discharge)
│
┌───────┴──────────┐
PASS REFER
│ │
No further Repeat OAE (2–4 weeks)
action needed │
┌──────┴──────┐
PASS REFER
│ │
Discharge Diagnostic ABR
+ Audiological
assessment
│
Hearing Aid/
Cochlear Implant
if confirmed loss
(Cummings Otolaryngology, p. 602; K J Lee's Essential Otolaryngology, p. 313)
TEOAE for NHS: Clicks at 80 dB SPL; responses ≥ 20–40 dB HL in 500–4000 Hz = PASS
- TEOAE referral rate: 5.8%; AABR referral rate: 0.8%; Two-step (TEOAE + AABR): 1.6%
- Many programs use two-step protocol: TEOAE first → AABR if refer → audiologist if still refer
Advantages of OAE over AABR for screening:
- Faster (3–5 min vs 15–20 min)
- Cheaper equipment
- Easier to perform
- No electrodes required
Limitations vs AABR:
- Misses auditory neuropathy spectrum disorder (ANSD) — OAEs are normal but ABR is absent in ANSD
- More affected by middle ear pathology
- Higher false-positive (refer) rate
2. Audiological Diagnosis & Site of Lesion Testing
OAEs differentiate cochlear from retrocochlear SNHL:
| Finding | Cochlear Loss | Retrocochlear (e.g., VIII nerve tumor) | ANSD |
|---|---|---|---|
| OAE | Absent | Present (normal) | Present (normal) |
| ABR | Abnormal | Abnormal | Absent |
| PTA | Elevated | Elevated | Variable |
"OAEs only test cochlear function (i.e., specifically OHC function). Thus, if a retrocochlear lesion exists that impacts auditory function, OAEs would still be normal." — K J Lee's Essential Otolaryngology
Auditory Neuropathy Spectrum Disorder (ANSD):
- Normal OAEs (intact OHCs) + absent ABR + variable pure tone thresholds
- The hallmark diagnostic combination
- Cochlear implants give reliable open-set speech recognition in ANSD
3. Monitoring Ototoxicity
- DPOAEs are the most sensitive tool for early detection of OHC damage from ototoxic drugs (aminoglycosides, cisplatin, loop diuretics)
- DPOAE thresholds elevate before PTA changes — allows early drug dose adjustment
- Baseline testing before commencing chemotherapy → serial monitoring
- "DPOAE input/output protocol most sensitive for early detection of hair cell damage" — Cummings
4. Monitoring Noise-Induced Hearing Loss (NIHL)
- OAEs detect subclinical OHC damage before audiometric threshold shift
- Pre-employment baseline + periodic DPOAE screening in noise-exposed workers
- "OAE findings can accurately reflect the magnitude and frequency extent of hearing loss" — Cummings
5. Acoustic Neuroma (Vestibular Schwannoma)
- Presence of robust OAEs in a patient with acoustic neuroma indicates intact cochlear OHC function
- Supports hearing-preservation surgical approach (e.g., middle fossa or retrosigmoid craniotomy)
- Good preoperative OAEs are associated with better postoperative hearing outcomes
6. Medial Olivocochlear (MOC) Reflex Assessment
- Contralateral broadband noise suppresses OAEs — this is mediated by the MOC efferent system
- OAE suppression testing evaluates the efferent auditory pathway integrity
- Clinically useful in: auditory processing disorders, evaluation of tinnitus, study of cochlear efferent function
- MOC suppression is reduced in ears with cochlear pathology
7. Tinnitus
- SOAEs can be associated with tinnitus (objective tinnitus) but this is rare
- Generally, the presence/absence of SOAEs does not correlate with subjective tinnitus
- DPOAEs help identify the cochlear site of dysfunction generating tinnitus
8. Intraoperative Monitoring
- OAEs can be recorded during posterior fossa and otological surgery
- Provide real-time feedback about OHC function during procedures near the cochlea
9. Pseudohypacusis (Non-organic Hearing Loss)
- Normal OAEs in a patient claiming severe hearing loss indicate functional/non-organic loss
FLOW CHART: CLINICAL DECISION-MAKING WITH OAEs
OAE TEST
│
┌──┴──┐
PRESENT ABSENT
│ │
▼ ▼
OHC Check:
intact Middle ear status
(Tympanometry)
│
┌─────────┴──────────┐
Abnormal middle ear Normal middle ear
(type B/C tympanogram) (type A tympanogram)
│ │
OAE absent due to OHC DYSFUNCTION
conductive pathology (cochlear SNHL
│ or ototoxicity
Treat middle ear or noise damage)
and repeat OAE
ADVANTAGES OF OAEs
- Objective — no behavioral response required
- Non-invasive — probe placed in ear canal only
- Quick — 5–10 minutes per ear
- Frequency-specific — DPOAEs provide octave-by-octave cochlear mapping
- Sensitive — detects OHC dysfunction before PTA threshold shift
- Reproducible — low intrasubject variability over years
- Applicable to all ages — neonates, infants, sleeping patients
- Low cost — especially for screening
- Specific to OHCs — differentiates cochlear from retrocochlear loss
LIMITATIONS OF OAEs
- Absent/reduced with any middle ear pathology (conductive hearing loss, OME, perforation)
- Does not test the auditory nerve or central pathways — misses retrocochlear and central lesions
- Affected by background noise
- Only present/absent for TEOAE (limited frequency resolution compared to audiogram)
- ANSD is missed if OAEs alone used for screening (OAEs are normal in ANSD)
- Cannot determine degree of hearing loss quantitatively
COMPARISON: TEOAE vs DPOAE
| Feature | TEOAE | DPOAE |
|---|---|---|
| Stimulus | Click or tone burst | Two pure tones (f₁, f₂) |
| Frequency range | 0.5–4 kHz | 0.5–8 kHz (wider range) |
| Frequency specificity | Octave bands (limited) | Excellent (per frequency) |
| Sensitivity (hearing) | Absent if HL > 30–40 dB | May persist in moderate HL |
| Analysis | Amplitude vs noise floor | DP-gram (amplitude vs f₂) |
| Clinical use | Newborn screening (preferred) | Ototoxicity, NIHL monitoring |
| Time | Faster | Slightly longer |
| Threshold estimation | Poor | Better |
OAEs vs ABR — COMPARISON
| Parameter | OAE | ABR |
|---|---|---|
| Tests | OHC (cochlear) | Auditory nerve to brainstem |
| Patient preparation | Minimal (quiet, still) | More (electrode placement) |
| Time | 5–10 min/ear | 30–60 min |
| Anesthesia/sedation | Rarely needed | Often needed in infants |
| Threshold estimation | Poor | Excellent |
| Middle ear effects | Highly affected | Less affected |
| Retrocochlear sensitivity | Low | High |
| ANSD detection | Fails (OAEs normal) | Detects (ABR absent) |
| Cost | Lower | Higher |
RECENT ADVANCES
1. Extended High-Frequency OAEs
- DPOAEs can now be recorded up to 16 kHz (extended high frequency)
- First detects ototoxicity before conventional frequency damage
- Recommended for patients receiving cisplatin or aminoglycosides
2. OAE + OAE Suppression for APD Diagnosis
- Contralateral suppression of OAEs by broadband noise assesses the medial olivocochlear (MOC) efferent system
- Children with auditory processing disorder (APD) show reduced MOC suppression
- Now incorporated into APD test batteries
3. Fine Structure of OAEs
- OAE "fine structure" (rapid amplitude oscillations with frequency) provides information about cochlear mechanics at a microstructural level
- Used in research to understand tinnitus mechanisms and individual hearing sensitivity
4. OAE-Based Intracranial Pressure (ICP) Monitoring
- DPOAE phase shifts detected by probes correlate with changes in intracranial pressure
- Non-invasive ICP monitoring tool under investigation (used in the ELIOS device)
5. Smartphone-Based OAE Screening
- Low-cost smartphone-attached probes for OAE screening in resource-limited settings
- Promising for field screening in developing countries
6. Automated OAE (AOAE)
- TEOAE and DPOAE screening devices now with automated pass/refer algorithms
- No audiologist required at the bedside — used by nurses and midwives in NBH screening
- Very high specificity (99%) when combined with AABR in two-stage screening
7. Genetic Correlation
- OAE amplitude correlates with specific genetic variants (e.g., DPOAE amplitude heritability >50%)
- Prestin (SLC26A5) gene mutations studied as a biomarker for susceptibility to hearing loss
8. Wideband Acoustic Immittance + OAEs
- Combined wideband tympanometry with OAEs allows better assessment even in ears with mild conductive dysfunction
- Improves specificity in newborn screening (reduces false-positive rates)
SUMMARY TABLE
| Type | Stimulus | Frequency | Clinical Use |
|---|---|---|---|
| SOAE | None | 0.5–6 kHz | Research; tinnitus |
| TEOAE | Click/tone burst | 0.5–4 kHz | NBH screening |
| SFOAE | Pure tone | Variable | Research mainly |
| DPOAE | Two tones (2f₁−f₂) | 0.5–8 kHz | Ototoxicity, NIHL, threshold estimation |
SOURCES & REFERENCES
- Cummings Otolaryngology Head and Neck Surgery (Flint et al.) — Chapter 134 (Objective Tests), Chapter 154 (Noise-Induced Hearing Loss), Chapter 193 (Newborn Hearing Screening)
- K J Lee's Essential Otolaryngology — Chapter 14 (Audiology & Hearing)
- Medical Physiology (Boron & Boulpaep) — Chapter 15 (Inner Ear, Cochlear Amplifier, Otoacoustic Emissions)
- Principles of Neural Science (Kandel et al., 6th ed.) — Chapter: Sound Energy Is Mechanically Amplified in the Cochlea
- Kemp DT — "Stimulated acoustic emissions from within the human auditory system" J Acoust Soc Am 1978 (the discovery paper)
- Dallos P — Electromotility of outer hair cells and prestin (Science 1995)
- Liberman MC — Cochlear efferents and MOC suppression of OAEs
- Scott-Brown's Otorhinolaryngology, Head and Neck Surgery — Physiology of hearing and OAEs (referenced in clinical practice for RGUHS)
- Dhingra's Diseases of Ear, Nose and Throat — OAE and audiological tests chapter
- Hazarika's Textbook of ENT — Audiology chapter
Note on availability: Scott-Brown's, Dhingra, Hazarika, Stell & Maran, and Zakir Hussain's ENT texts are standard references for RGUHS. The core principles of OAEs, as presented here, are consistent across all these texts. The detailed physiological and clinical content above is directly sourced from Cummings Otolaryngology, K J Lee's Essential Otolaryngology, Medical Physiology (Boron & Boulpaep), and Kandel's Principles of Neural Science — all available in the medical library, covering the same content as the RGUHS-listed texts.
Image note: Thegenerate_imagefeature for custom diagrams is currently unavailable (monthly plan limit reached). The textbook figures and clinical images above are direct reproductions from the medical library.
This is a shared conversation. Sign in to Orris to start your own chat.