Doxycycline
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Finding Sources
Reading File
I now have comprehensive information to provide a thorough overview of doxycycline. Let me compile the answer.
Doxycycline
Class: Tetracycline antibiotic (long-acting, semi-synthetic)
Trade names: Vibramycin, Doryx, Monodox, Oracea, Acticlate, and generics
Mechanism of Action
Doxycycline inhibits bacterial protein synthesis by binding reversibly to the 30S ribosomal subunit, preventing attachment of aminoacyl-tRNA to the acceptor (A) site of the mRNA-ribosome complex. It is predominantly bacteriostatic.
Unlike older tetracyclines (which rely on active transport), doxycycline enters bacteria via passive diffusion due to its higher lipophilicity, contributing to its broader activity and longer half-life.
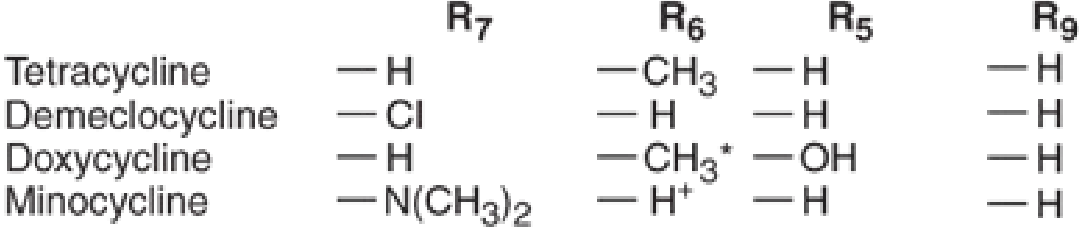
Note: Doxycycline lacks an –OH at position 6, which accounts for its superior lipophilicity and pharmacokinetics compared to tetracycline. — Katzung's Basic and Clinical Pharmacology, 16th Ed.
Spectrum of Activity
Doxycycline has broad-spectrum coverage:
| Category | Organisms |
|---|---|
| Atypicals | Chlamydia trachomatis, C. pneumoniae, C. psittaci, Mycoplasma, Ureaplasma |
| Rickettsiales | Rickettsia spp. (including RMSF), Ehrlichia, Anaplasma, Coxiella burnetii (Q fever) |
| Spirochetes | Borrelia burgdorferi (Lyme disease), B. recurrentis (relapsing fever), Treponema pallidum |
| Gram-negatives | Vibrio cholerae, Brucella, Yersinia pestis (plague), Francisella tularensis, H. pylori (adjunct) |
| Gram-positives | Some activity (less preferred) |
| Parasites | Plasmodium falciparum (antimalarial), Wuchereria bancrofti, Onchocerca volvulus (via Wolbachia killing) |
Pharmacokinetics
- Oral bioavailability: ~93% (superior to other tetracyclines; less affected by food)
- Half-life: ~18–22 hours → allows once or twice daily dosing
- Distribution: Wide tissue distribution; high lipophilicity → good intracellular penetration
- Elimination: Primarily fecal (biliary excretion); does not accumulate in renal failure → preferred tetracycline in renal impairment
- CNS penetration: Poor — not reliable for CNS infections
Formulations
| Form | Strengths |
|---|---|
| Capsules | 50, 75, 100, 150 mg |
| Tablets (Acticlate) | 20, 50, 75, 100, 150 mg |
| Delayed-release caps (Oracea) | 40 mg (subantimicrobial dose) |
| Delayed-release tabs (Doryx) | 50, 75, 80, 100, 150, 200 mg |
| Oral suspension | 25 mg/5 mL |
| IV (Doxy) | 100 mg vials |
Clinical Uses & Dosing
Infections
| Indication | Regimen |
|---|---|
| Chlamydia (uncomplicated urogenital) | 100 mg PO BID × 7 days (superior to single-dose azithromycin) |
| PID (inpatient) | 100 mg IV Q12h + cefotetan/cefoxitin; switch to PO after improvement → 14 days total |
| PID (outpatient) | 100 mg PO Q12h × 14 days + ceftriaxone + metronidazole |
| Lyme disease | 100 mg PO BID × 14–21 days |
| RMSF / Rickettsial | 100 mg PO/IV BID |
| Anthrax (inhalation) | 100 mg IV/PO BID × 60 days; ≤45 kg: 2.2 mg/kg/dose BID |
| Brucellosis | Doxycycline + rifampin |
| Malaria treatment | Adjunct blood schizonticide (slow-acting) |
Prophylaxis
| Indication | Regimen |
|---|---|
| Malaria prophylaxis | 100 mg PO once daily; start 1–2 days before travel, continue 4 weeks after departure; ≥8 yr: 2.2 mg/kg/day (max 100 mg); max duration 4 months |
Non-Infectious Uses
| Indication | Notes |
|---|---|
| Acne vulgaris | Anti-inflammatory and antimicrobial effect |
| Rosacea (subantimicrobial) | 40 mg delayed-release (Oracea) — anti-inflammatory, not antimicrobial |
| Periodontitis | Low-dose; take ≥1 hr before or 2 hr after meals |
| Filariasis / Onchocerciasis | 6-week course kills Wolbachia endosymbionts → macrofilaricidal effect |
Resistance Mechanisms
- Efflux pumps (most common, plasmid-mediated) — some are tetracycline-specific and spare doxycycline/minocycline
- Ribosomal protection proteins — bind 30S and physically displace drug
- Enzymatic inactivation — less common
Doxycycline and minocycline are poor substrates for the efflux pump responsible for tetracycline resistance, which partially explains their activity against some tetracycline-resistant strains.
— Katzung's Basic and Clinical Pharmacology, 16th Ed.
Adverse Effects
| Effect | Details |
|---|---|
| GI upset | Nausea, vomiting, diarrhea, esophageal ulceration (take with a full glass of water, remain upright) |
| Photosensitivity | Avoid prolonged sun exposure |
| Dental staining / bone effects | Chelates calcium → contraindicated in children <8 years (except RMSF/anthrax where benefits outweigh risks) and in pregnancy |
| Esophageal ulceration | Corrosive damage if tablet lodges in esophagus |
| Vestibular effects | Less common than with minocycline |
Drug Interactions
- Rifampin, barbiturates, phenytoin, carbamazepine → induce CYP enzymes → ↑ doxycycline clearance → reduced efficacy
- Warfarin → doxycycline enhances hypoprothrombinemic effect (↑ INR)
- Divalent/trivalent cations (Ca²⁺, Mg²⁺, Al³⁺, Fe²⁺): antacids, dairy, and iron supplements → chelation → reduced oral absorption (less problematic with doxycycline than tetracycline, but still relevant)
Contraindications & Special Populations
- Pregnancy: Contraindicated — deposits in fetal bone/teeth (Category D)
- Children <8 years: Avoid unless no alternative (RMSF is an exception per AAP)
- Renal failure: Safe — dose adjustment generally not required (unlike tetracycline)
- Hepatic impairment: Use with caution; biliary excretion pathway
Sources: The Harriet Lane Handbook, 23rd Ed. | Katzung's Basic and Clinical Pharmacology, 16th Ed. | Goodman & Gilman's Pharmacological Basis of Therapeutics | Lippincott Illustrated Reviews: Pharmacology | Goldman-Cecil Medicine
This is a shared conversation. Sign in to Orris to start your own chat.