Paraneoplastic syndrome
Reading File
Finding Sources
Searching PubMed
"paraneoplastic syndrome" AND diagnosis
Finding Sources
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Finding Sources
Reading File
I now have comprehensive material from Harrison's 22E (2025). Let me compile the full, authoritative answer:
Paraneoplastic Syndromes
Paraneoplastic syndromes (PNS) refer to disorders that accompany benign or malignant tumors but are not directly related to mass effects or invasion — they arise from tumor-secreted substances (hormones, cytokines, growth factors) or tumor-triggered immune responses. They affect up to 15% of cancer patients and, critically, precede the cancer diagnosis in ~60–65% of cases, making recognition essential for early tumor detection.
Pathogenesis
Two major mechanisms operate:
1. Ectopic Hormone/Substance Production
Tumor cells (often poorly differentiated or dedifferentiated) express peptide hormones normally produced only by specific endocrine tissues. The resulting hormone excess is:
- Unregulated — defective feedback control (e.g., ACTH from SCLC is not suppressed by dexamethasone in standard doses)
- Aberrantly processed — large, unprocessed precursors (e.g., POMC instead of mature ACTH)
- Driven by epigenetic derepression, translocation, or cellular dedifferentiation
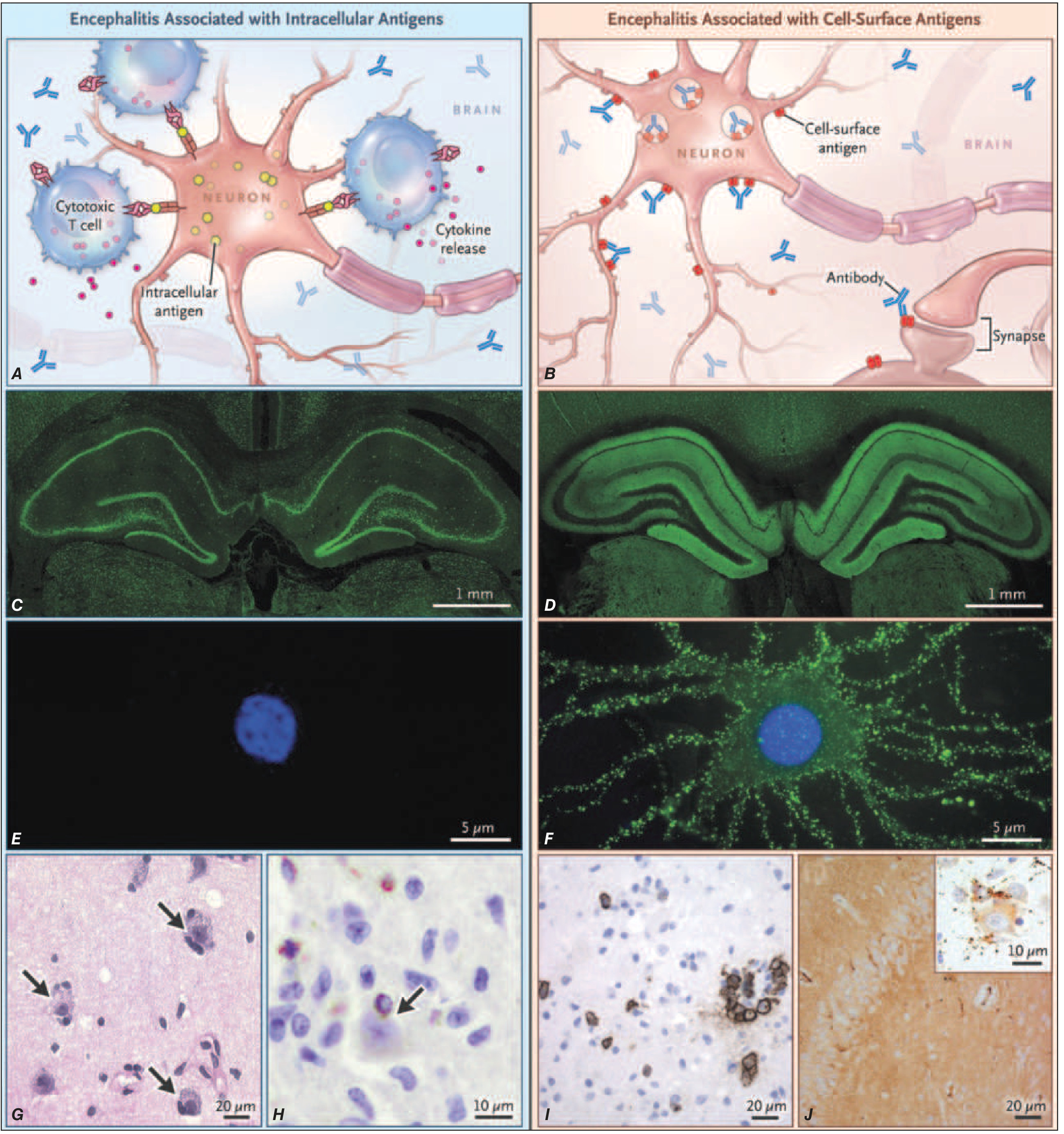
2. Autoimmune/Immune-Mediated (Paraneoplastic Neurologic Disorders)
Tumors ectopically express neural antigens → immune response generated against the tumor cross-reacts with the nervous system. Two mechanistic subtypes:
| Target Antigen Location | Mechanism | Antibody Examples | Response to Treatment |
|---|---|---|---|
| Intracellular | CD4+/CD8+ T-cell cytotoxicity predominates; antibodies cannot access the epitope | Anti-Hu, Anti-Yo, Anti-Ri | Poor — irreversible neuronal loss |
| Cell-surface / synaptic | Antibodies directly alter receptor function/structure | Anti-NMDAR, Anti-AMPAR, Anti-LGI1, Anti-CASPR2 | Better — often reversible |
I. Endocrine Paraneoplastic Syndromes
The most common and clinically significant:
Hypercalcemia of Malignancy
- Most common paraneoplastic endocrine syndrome
- Mediator: PTHrP (parathyroid hormone-related protein) → activates PTH receptor
- Tumors: squamous cell carcinoma (lung, head/neck, esophagus), renal cell, breast, bladder
- Also caused by: ectopic 1,25-(OH)₂D (lymphoma), direct bone resorption via cytokines (myeloma), rarely ectopic PTH
Ectopic ACTH Syndrome (Cushing's Syndrome)
- Mediator: ectopic ACTH or CRH production
- Tumors: SCLC (#1), carcinoid tumors, medullary thyroid cancer, pheochromocytoma, pancreatic islet cell tumors
- Key features: rapid onset, profound hypokalemia, muscle wasting, hyperglycemia; classic cushingoid habitus often absent because tumor progression is too fast
- Biochemistry: very high cortisol, high ACTH, no suppression with high-dose dexamethasone
SIADH (Syndrome of Inappropriate ADH)
- Mediator: ectopic vasopressin secretion
- Tumors: SCLC (#1), head/neck squamous cell, primary brain tumors
- Presents: euvolemic hyponatremia, urine osmolality inappropriately high relative to serum
- Treatment: fluid restriction; demeclocycline for long-term control; tumor treatment is definitive
Other Endocrine Syndromes
| Syndrome | Mediator | Tumors |
|---|---|---|
| Non-islet cell hypoglycemia | IGF-2 (large retroperitoneal sarcomas, hepatocellular Ca) or insulin (islet cell) | Sarcomas, HCC, islet cell |
| Gynaecomastia / hCG excess | hCG, estrogens | Testicular germ cell, lung |
| Acromegaly | Ectopic GH or GHRH | Carcinoid, pancreatic islet cell |
| Hyperthyroidism | hCG (TSH-like activity) | Choriocarcinoma, gestational trophoblastic disease |
| Erythrocytosis | Ectopic EPO | RCC, cerebellar hemangioblastoma, uterine fibroids, HCC |
II. Neurologic Paraneoplastic Disorders (PNDs)
Clinically disabling PNDs occur in 0.5–1% of all cancer patients but 2–3% of SCLC/neuroblastoma patients and 30–50% of thymoma patients.
High-Risk Antibodies (>70% probability of underlying cancer)
| Antibody | Syndrome | Tumor |
|---|---|---|
| Anti-Hu (ANNA-1) | Encephalomyelitis, sensory neuronopathy | SCLC |
| Anti-Yo (PCA-1) | Rapidly progressive cerebellar degeneration | Ovary, breast |
| Anti-Ri (ANNA-2) | Cerebellar degeneration, opsoclonus, brainstem encephalitis | Breast, gynecologic, SCLC |
| Anti-CRMP5 (CV2) | Encephalomyelitis, chorea, optic neuritis, peripheral neuropathy | SCLC, thymoma |
| Anti-Tr (DNER) | Rapidly progressive cerebellar degeneration | Hodgkin's lymphoma |
| Anti-Ma2 | Limbic/hypothalamic/brainstem encephalitis | Testicular (young men) |
| Anti-Kelch-like protein 11 | Brainstem encephalitis, ataxia, sensorineural hearing loss | Seminoma, germ cell |
Intermediate-Risk Antibodies (30–70% probability of cancer)
Anti-NMDAR, Anti-AMPAR, Anti-GABA-B-R, Anti-LGI1, Anti-CASPR2
Specific PND Syndromes
Paraneoplastic Cerebellar Degeneration (PCD)
- Subacute onset of dysarthria, limb and gait ataxia, oculomotor dysfunction
- Pathology: severe Purkinje cell loss
- Antibodies: Anti-Yo (breast/ovarian), Anti-Tr (Hodgkin's), Anti-Hu (SCLC)
Paraneoplastic Encephalomyelitis / Limbic Encephalitis
- Limbic: subacute amnesia, personality change, temporal lobe seizures, psychiatric symptoms
- MRI: bilateral medial temporal lobe FLAIR hyperintensity
- Antibodies: Anti-Hu, Anti-Ma2, Anti-LGI1 (SCLC, testicular)
Paraneoplastic Sensory Neuronopathy
- Asymmetric numbness/burning paresthesias, loss of proprioception and vibration (posterior column)
- Anti-Hu (SCLC)
Lambert-Eaton Myasthenic Syndrome (LEMS)
- Proximal lower limb weakness; strength improves with sustained contraction (opposite to myasthenia gravis)
- Antibody: Anti-VGCC (voltage-gated calcium channels); >80% with SCLC also have Anti-SOX1
- EMG: incremental response at high-frequency (>10 Hz) stimulation
- Autonomic features common (dry mouth, erectile dysfunction)
Paraneoplastic Opsoclonus-Myoclonus
- Chaotic conjugate eye movements + myoclonus; in adults usually SCLC or breast (Anti-Ri)
- In children: neuroblastoma (most common cause)
Paraneoplastic Stiff-Person Syndrome
- Progressive rigidity, painful spasms of axial muscles; Anti-amphiphysin or Anti-GAD65
- Tumors: breast cancer (Anti-amphiphysin), SCLC
III. Hematologic Paraneoplastic Syndromes
| Syndrome | Mechanism | Associated Tumors |
|---|---|---|
| Erythrocytosis | Ectopic EPO | RCC, cerebellar hemangioblastoma, HCC |
| Granulocytosis / leukocytosis | Tumor production of G-CSF, GM-CSF, IL-6 | Lung, GI, GU cancers |
| Thrombocytosis | Ectopic thrombopoietin or IL-6 | Various carcinomas |
| Pure red cell aplasia | Autoimmune | Thymoma (#1) |
| Trousseau's syndrome | Hypercoagulability (mucin-secreting adenocarcinomas activate clotting) | Pancreatic, GI, lung adenocarcinoma |
IV. Dermatologic Paraneoplastic Syndromes
| Sign | Features | Tumor |
|---|---|---|
| Acanthosis nigricans | Velvety hyperpigmentation in skin folds | Gastric, GI adenocarcinoma |
| Dermatomyositis | Heliotrope rash, Gottron's papules, proximal myopathy | Lung, ovarian, breast, GI |
| Leser-Trélat sign | Sudden eruption of multiple seborrheic keratoses | GI, lymphoma |
| Paraneoplastic pemphigus | Severe mucocutaneous blistering | Lymphoma (NHL, CLL), thymoma |
| Necrolytic migratory erythema | Migratory, blistering rash in skin folds | Glucagonoma |
| Sweet's syndrome | Tender red plaques + fever + neutrophilia | AML, myelodysplastic syndrome |
V. Rheumatologic & Other
- Hypertrophic osteoarthropathy — periosteal new bone formation, clubbing; lung carcinoma
- Palmar fasciitis/polyarthritis syndrome — rapidly progressive joint contractures; ovarian cancer
- Paraneoplastic glomerulonephritis — membranous nephropathy (solid tumors, especially lung/GI), minimal change disease (lymphoma, NSAID-like)
Diagnosis
The Goldman-Cecil approach to evaluating a suspected PNS:
- Characterize the abnormality; obtain labs and biopsy as needed
- Eliminate common causes first
- If no obvious etiology → consider PNS
- If consistent with a known PNS → screen for occult malignancy (CT chest/abdomen/pelvis, PET-CT, tumor markers, testicular US in young men)
- Antibody panel (serum and CSF for neurologic PNDs)
- Course of the syndrome should parallel tumor course — a hallmark of PNS
Treatment
| Principle | Details |
|---|---|
| Treat the underlying tumor | Definitive treatment; resolution of PNS often follows tumor response |
| Immunosuppression (neurologic PNDs) | High-dose corticosteroids, IVIG, plasmapheresis, rituximab |
| Prognosis | Surface-antigen PNDs (anti-NMDAR, LGI1) respond better than intracellular-antigen PNDs (anti-Hu, anti-Yo) |
| Endocrine syndromes | Symptomatic management (bisphosphonates for hypercalcemia, fluid restriction + tolvaptan for SIADH, metyrapone/ketoconazole for ectopic ACTH) while treating the primary cancer |
Sources: Harrison's Principles of Internal Medicine 22E (2025), Ch. 98–99; Goldman-Cecil Medicine, Ch. 164; Bradley and Daroff's Neurology in Clinical Practice
This is a shared conversation. Sign in to Orris to start your own chat.