Does all this cause bilateral hand wasting
Reading File
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Finding Sources
Now I have enough to give a comprehensive answer. Let me analyze each item on the diagram:
BL Hand Muscle Wasting — Does Everything on This Diagram Cause It?
Yes — but with important nuances for several entries. Here's a systematic review of each item:
LEFT BRANCH: UMN Signs (+) → Mixed UMN + LMN
These represent conditions where both UMN and LMN are involved, with wasting coming from the LMN component:
| # | Condition | Causes Bilateral Hand Wasting? |
|---|---|---|
| ① | Syringomyelia | ✅ Yes — cavity destroys anterior horn cells at C8–T1, causing bilateral, suspended/cape distribution wasting with dissociated sensory loss |
| ② | Cervical Spondylitic Myelopathy | ✅ Yes — cord compression at C5–C6 or C3–C4 can cause bilateral intrinsic hand wasting (C8–T1 myotomes) + long tract signs. Harrison's confirms: "The diagnosis should always be considered in cases of progressive cervical myelopathy… or wasting of the hands" |
| ③ | Intramedullary Tumours | ✅ Yes — same mechanism as syringomyelia (central cord expansion destroys anterior horn cells bilaterally) |
| ④ | MND → ALS | ✅ Yes — classic presentation. ALS causes split-hand pattern (preferential thenar > hypothenar wasting). "As the weeks and months pass, the other hand and arm become similarly affected" — Adams & Victor's Neurology |
RIGHT BRANCH: Pure LMN Type
Anterior Horn Cell
| Condition | Bilateral Hand Wasting? |
|---|---|
| PMA (Progressive Muscular Atrophy) | ✅ Yes — pure LMN MND, bilateral wasting |
| Kennedy's Disease | ✅ Yes — X-linked bulbospinal muscular atrophy, bilateral proximal > distal wasting |
| Hirayama Disease | ⚠️ Typically unilateral or asymmetric — juvenile muscular atrophy of the distal upper limb. Characteristically unilateral (dominant side), though bilateral cases exist. This is a notable exception |
| Polio / Enterovirus / Coxsackievirus | ⚠️ Can be bilateral but typically asymmetric/patchy, not symmetrically bilateral |
Root (C8–T1 Radiculopathy)
| Condition | Bilateral Hand Wasting? |
|---|---|
| Cervical Spondylosis (radiculopathy) | ⚠️ Usually unilateral — C8/T1 root compression is typically on one side. Bilateral only if multilevel stenosis affects both sides |
| Cervical Rib / TOS | ⚠️ Usually unilateral |
Plexus
| Condition | Bilateral Hand Wasting? |
|---|---|
| Pancoast Tumour | ⚠️ Unilateral — involves lower trunk (C8/T1) of one brachial plexus only |
| Lower Trunk Brachial Plexopathy | ⚠️ Usually unilateral |
| Thoracic Outlet Syndrome | ⚠️ Usually unilateral |
Peripheral Nerve
| Condition | Bilateral Hand Wasting? |
|---|---|
| Median Neuropathy (bilateral CTS) | ✅ Yes — bilateral carpal tunnel syndrome is common; causes thenar wasting bilaterally |
| Ulnar Neuropathy | ⚠️ Usually unilateral; bilateral possible but less common |
| Combined Neuropathy | ✅ Yes |
| CMT (Charcot–Marie–Tooth) | ✅ Yes — hereditary motor and sensory neuropathy, classically bilateral and symmetric |
| mmNCB (likely MMN — Multifocal Motor Neuropathy with Conduction Block) | ⚠️ Typically asymmetric/multifocal, not symmetrically bilateral |
| Leprosy | ✅ Yes — bilateral ulnar + median nerve involvement causes bilateral hand wasting in endemic regions |
Muscle
| Condition | Bilateral Hand Wasting? |
|---|---|
| Distal Myopathy / Miyoshi | ✅ Yes — Miyoshi myopathy affects distal limbs bilaterally (though primarily lower limb in Miyoshi) |
| LGCOE (likely LGMD — Limb Girdle CMD/Myopathy?) | ⚠️ Primarily proximal; distal hand wasting is not a major feature |
| IBM (Inclusion Body Myositis) | ✅ Yes — IBM is unique among myopathies in causing finger flexor and quadriceps wasting bilaterally |
| Myotonic Dystrophy | ✅ Yes — distal wasting, bilateral hand/forearm involvement is characteristic |
Bottom Left: Disuse Atrophy → RA
| Condition | Bilateral Hand Wasting? |
|---|---|
| RA + Disuse Atrophy | ✅ Yes — rheumatoid arthritis causes interosseous wasting from a combination of disuse atrophy, vasculitis, and entrapment neuropathy (median/ulnar). Classically bilateral and symmetric |
Summary
Most causes on this diagram do produce bilateral hand wasting, but three groups deserve qualification:
- Typically unilateral (unless bilateral disease): Pancoast tumour, lower trunk plexopathy, TOS, C8–T1 radiculopathy, ulnar neuropathy
- Characteristically unilateral/asymmetric: Hirayama disease, MMN (multifocal motor neuropathy)
- Bilateral but primarily proximal (hand wasting not a main feature): LGMD
The most reliably bilateral causes are: ALS/MND, PMA, Kennedy's disease, syringomyelia, intramedullary tumour, cervical myelopathy, CMT, bilateral CTS, IBM, myotonic dystrophy, and RA.
60 Yr with female with pancerebellar symp with wt loss 10 kg what other history to ask
Finding Sources
Finding Sources
Reading File
Finding Sources
Reading File
Reading File
This is a high-yield clinical scenario. The combination of 60-year-old female + pancerebellar syndrome + 10 kg weight loss is a classic red-flag presentation for Paraneoplastic Cerebellar Degeneration (PCD). Here's a structured history to take:
History to Ask — Structured Approach
🔴 The Working Diagnosis: Paraneoplastic Cerebellar Degeneration (PCD)
In a 60F with subacute pancerebellar syndrome + significant weight loss, PCD must be the first diagnosis to exclude. The associated cancers are:
| Cancer | Antibody |
|---|---|
| Ovarian / Uterine | Anti-Yo (PCA-1) — most common in women |
| Breast | Anti-Yo, Anti-Ri |
| Small Cell Lung (SCLC) | Anti-Hu, Anti-Zic4, Anti-VGCC |
| Hodgkin Lymphoma | Anti-Tr (mGluR1) |
"Paraneoplastic cerebellar degeneration associated with breast and gynecologic (ovaries, uterus) carcinomas — anti-Yo, anti-Ri antibodies" — Localization in Clinical Neurology, 8e
1. History of Presenting Illness — Cerebellar Symptoms
- Onset and progression — subacute onset over weeks to months is a red flag for PCD (vs. slow years-long course of hereditary ataxia)
- Sequence — which came first: gait unsteadiness, limb ataxia, slurred speech, double vision, oscillopsia?
- Any nystagmus / oscillopsia noticed?
- Dysphagia or dysphonia — brainstem involvement (paraneoplastic encephalomyelitis overlap)
- Cognitive changes / memory problems — limbic encephalitis component?
- Sensory symptoms in limbs — sensory neuronopathy (anti-Hu)
- Autonomic symptoms — postural dizziness, constipation, gastroparesis (anti-Hu PEM)
2. Gynecological / Oncological History ← Most Critical in a 60F
- Last Pap smear / cervical screening — when was it done?
- Postmenopausal bleeding — endometrial/uterine carcinoma
- Breast lump, nipple discharge, last mammogram — breast cancer (anti-Yo)
- Ovarian symptoms — bloating, early satiety, pelvic fullness (ovarian carcinoma)
- Family history of breast, ovarian, or colorectal cancer (BRCA risk)
3. Weight Loss Characterisation
- Duration — how long for 10 kg loss?
- Associated anorexia vs. normal appetite (anorexia + weight loss = malignancy)
- Night sweats — lymphoma (Hodgkin's → anti-Tr cerebellar degeneration)
- Dysphagia / odynophagia — oesophageal/gastric malignancy
- Haemoptysis / chronic cough / smoker — SCLC (anti-Hu)
- Change in bowel habits, rectal bleeding — colorectal cancer
4. Smoking & Substance History
- Smoking pack-years — SCLC most associated with PCD via anti-Hu; also risk for paraneoplastic encephalomyelitis
- Alcohol — chronic alcohol use is a major non-paraneoplastic cause of cerebellar degeneration (vermis predominant, but can be pancerebellar)
- Alcohol quantity and duration — vermis atrophy, thiamine deficiency (Wernicke's)
5. Medication & Toxic History
- Phenytoin — classic cause of cerebellar atrophy/toxicity
- Chemotherapy (prior cancer treatment) — cytarabine, 5-FU, methotrexate can cause cerebellar syndrome
- Lithium toxicity
- Heavy metals — mercury, lead
6. Family History
- Hereditary ataxias (SCA — spinocerebellar ataxia types): onset at 60 is late but possible for SCA3/SCA6
- Family history of ataxia, Parkinson's, tremor
7. Past Medical History
- Prior malignancy (even if "cured" — recurrence can present with PCD, including Hodgkin's)
- Hypothyroidism — cause of cerebellar syndrome (ask about fatigue, cold intolerance, constipation)
- Coeliac disease — ask about chronic diarrhoea, bloating (gluten ataxia)
- Diabetes — non-ketotic hyperglycaemia can cause cerebellar signs; also peripheral neuropathy
- Autoimmune disease — SLE, Sjögren's (anti-Ro antibodies linked to cerebellar ataxia)
8. Systems Review
- Rash — dermatitis herpetiformis (gluten ataxia), or paraneoplastic
- Uveitis / vision changes
- Myoclonus / jerks — opsoclonus-myoclonus (anti-Ri, breast/gynaecological)
- Psychiatric symptoms — depression, anxiety, personality change (limbic involvement)
Bottom Line for Exam
The key message: 60F + subacute pancerebellar + weight loss = paraneoplastic until proven otherwise. The history must specifically screen for:
- Gynaecological cancer (ovary/uterus → anti-Yo) — most likely in this demographic
- Breast cancer screening history
- Smoking/SCLC (anti-Hu)
- Lymphoma (night sweats, nodes → anti-Tr)
- Alcohol (non-paraneoplastic cause to exclude)
- Subacute onset (weeks–months) — confirms paraneoplastic pattern vs. hereditary (years)
Differential for temporal hyperintensity and approch
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Finding Sources
Finding Sources
Excellent. Here is a comprehensive, structured answer:
Temporal Lobe Hyperintensity on MRI — Differentials & Approach
(FLAIR/T2 signal; medial > lateral depending on cause)
DIFFERENTIALS
🔴 Category 1: Infectious / Inflammatory
| Diagnosis | Key Features |
|---|---|
| Herpes Simplex Encephalitis (HSE-1) | Most important to exclude — haemorrhagic necrosis of medial temporal lobe + insula + cingulate. Unilateral → bilateral. Fever, personality change, seizures, aphasia. Spares basal ganglia |
| HHV-6 Encephalitis | Symmetric bilateral medial temporal hyperintensity; post-bone marrow / stem cell transplant setting |
| Autoimmune / Limbic Encephalitis | Unilateral or bilateral mesial temporal FLAIR/T2 signal, best seen on coronal sequences; subacute amnesia, seizures, psychiatric features |
| Tuberculous/Fungal encephalitis | Often with leptomeningeal enhancement; basal ganglia involvement also |
🟠 Category 2: Autoimmune / Paraneoplastic Limbic Encephalitis
This is the most important differential after HSE and deserves its own breakdown:
| Antibody | Clinical Clue | Associated Tumour |
|---|---|---|
| Anti-Hu | Sensory neuronopathy + limbic encephalitis | SCLC |
| Anti-Ma2 | Young man + upper brainstem + limbic | Testicular germ cell |
| Anti-LGI1 | Faciobrachial dystonic seizures (FBDS), hyponatraemia | Thymoma (minor) |
| Anti-CASPR2 | Morvan syndrome, neuromyotonia | Thymoma |
| Anti-AMPA-R | Relapsing course | Lung, breast, thymoma |
| Anti-GABA-B | Prominent seizures | SCLC |
| Anti-NMDAR | Young female, psychiatric → movement disorder → coma | Ovarian teratoma |
"Typical MRI findings include unilateral or bilateral mesial temporal lobe abnormalities best seen on T2-weighted and FLAIR images. The temporal-limbic regions may be hypointense on T1 and rarely enhance with contrast." — Bradley & Daroff's Neurology
🟡 Category 3: Vascular
| Diagnosis | Key Features |
|---|---|
| PCA territory infarct | Acute DWI restriction + FLAIR; ipsilateral field defect; look for contralateral peduncle (Wernicke haemianopia) |
| CADASIL | Temporal pole white matter involvement is pathognomonic (distinguishes from other small vessel disease). NOTCH3 mutation, migraine, lacunar strokes, family history |
| Cerebral venous sinus thrombosis | FLAIR hyperintensity ± haemorrhage; does not respect arterial territories |
| Vasculitis (CNS) | Multifocal, bilateral; headache, CSF pleocytosis |
🟢 Category 4: Tumour
| Diagnosis | Key Features |
|---|---|
| Low-grade glioma | FLAIR hyperintensity without enhancement; slow expansion; often incidental or seizures |
| DNET / Ganglioglioma | Young patient with chronic temporal lobe epilepsy; bubbly T2, cortical-based |
| GBM | Ring enhancement, mass effect, oedema; older adult |
| Lymphoma | Periventricular/deep; may involve temporal lobe; enhancing |
| Metastasis | Usually cortical/grey-white junction; multiple; known primary |
🔵 Category 5: Metabolic / Toxic / Genetic
| Diagnosis | Key Features |
|---|---|
| MELAS | Stroke-like episodes; posterior > anterior; crosses vascular territories; maternal inheritance, young age, lactic acidosis |
| Wernicke Encephalopathy | Medial thalami + mammillary bodies + periaqueductal grey; temporal involvement less typical |
| Hypoglycaemia | Can mimic stroke; posterior temporal/parietal; DWI changes |
| Status epilepticus | Post-ictal/peri-ictal FLAIR hyperintensity in the hippocampus; transient; resolves |
| Hippocampal Sclerosis | Unilateral hippocampal FLAIR hyperintensity + volume loss; longstanding TLE history |
APPROACH
Step 1 — Characterise the MRI Signal
| Feature | Implication |
|---|---|
| Medial temporal (hippocampus, amygdala, parahippocampus) | HSE, limbic encephalitis, hippocampal sclerosis |
| Medial + insula + cingulate | HSE (classic triad) |
| Temporal pole white matter | CADASIL |
| Bilateral symmetric mesial | Autoimmune limbic encephalitis, HHV-6 |
| Unilateral + cortical | Infarct (PCA), HSE, low-grade glioma |
| Swelling + mass effect | HSE (haemorrhagic), high-grade glioma, abscess |
| DWI restriction | Acute infarct, HSE, status epilepticus |
| Enhancement | Tumour, abscess, HSE (late), autoimmune (rare) |
Step 2 — Clinical Context (History + Exam)
| Feature | Points Toward |
|---|---|
| Fever + rapid onset | HSE |
| Subacute amnesia + psychiatric symptoms | Limbic encephalitis |
| Faciobrachial dystonic seizures | Anti-LGI1 |
| Hyponatraemia | Anti-LGI1 |
| Known malignancy | Paraneoplastic |
| Young woman + psych features | Anti-NMDAR |
| Migraine + strokes + family history | CADASIL |
| Immunocompromised | HHV-6, opportunistic infection |
| Post-ictal state | Peri-ictal changes |
| Chronic seizures + atrophy | Hippocampal sclerosis |
Step 3 — Investigations
Bloods:
- FBC, CRP, ESR
- Electrolytes (hyponatraemia → LGI1)
- Serum autoimmune encephalitis panel: Anti-NMDAR, LGI1, CASPR2, AMPAR, GABA-B, Hu, Ma2
- Paraneoplastic screen (Anti-Hu, Yo, Ri)
- Lactic acid (MELAS)
- Thyroid (Hashimoto encephalopathy)
CSF:
- Opening pressure, cells, protein, glucose
- HSV PCR ← start empirical aciclovir BEFORE result
- VZV, EBV, HHV-6, CMV PCR
- Oligoclonal bands
- CSF autoimmune antibodies (more sensitive than serum for some)
- Cytology (if malignancy suspected)
EEG:
- Temporal lobe epileptiform activity supports limbic encephalitis or TLE
- Periodic lateralising discharges (PLEDs) → HSE
Further Imaging:
- MRI with contrast — enhancement pattern
- MR spectroscopy — if tumour vs. inflammation unclear
- PET/CT whole body — if autoimmune/paraneoplastic (find hidden primary)
- CT chest/abdomen/pelvis — tumour screen
Step 4 — Management Principle
Temporal lobe hyperintensity → fever/acute onset?
↓ YES → Start IV Aciclovir immediately (HSE treatment)
↓ NO → Subacute? → Autoimmune workup + paraneoplastic screen
→ Seizures only? → Consider status/postictal change
→ Structural/chronic? → Tumour / hippocampal sclerosis workup
Never delay aciclovir while awaiting HSV PCR — HSE is fatal if untreated, and the CSF PCR can be negative in the first 72 hours.
What CSF findings differentiate HSE from limbic encephalitis?
Finding Sources
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Here is a focused, exam-ready comparison:
CSF Findings: HSE vs Limbic Encephalitis
| Parameter | HSE | Autoimmune Limbic Encephalitis |
|---|---|---|
| Opening pressure | Elevated (180–400 mmH₂O) | Usually normal |
| Appearance | Clear or xanthochromic (if haemorrhagic) | Clear |
| White cells | 10–1,000 cells/µL, predominantly lymphocytes | Mild lymphocytic pleocytosis or normal (up to 50% normal) |
| Red blood cells | ✅ Present — up to 500 RBC/µL; reflects haemorrhagic necrosis | ❌ Absent |
| Protein | Elevated — typically 50–200 mg/dL (up to 870 mg/dL reported) | Mildly elevated or normal (>50 mg/dL) |
| Glucose | Usually normal; low in ~5% | Normal |
| HSV PCR | ✅ Positive (sens ~96%, spec ~99%) — diagnostic | Negative |
| Specific antibodies | Negative | Positive — Anti-LGI1, NMDAR, CASPR2, GABA-B, Hu, Ma2, etc. |
| Oligoclonal bands | May be present (non-specific) | May be present (non-specific) |
Key Differentiating Points
1. RBCs in CSF — the single most useful bedside discriminator
HSE causes haemorrhagic temporal lobe necrosis. RBCs in the CSF (in the absence of a traumatic tap) strongly favour HSE. Autoimmune limbic encephalitis does not cause haemorrhage.
"Up to 500 red cells/mm³ are common [in HSE], and the CSF protein content is usually elevated." — Plum & Posner's Diagnosis & Treatment of Stupor and Coma
2. Pleocytosis is variable in both — but more marked in HSE
- HSE: typically 50–200 WBC/µL, can be up to 1,000
- Limbic encephalitis: mild or absent — the CSF can be entirely normal in up to 50% of autoimmune cases
"CSF often demonstrates findings consistent with inflammation (protein >50 mg/dL or lymphocytic pleocytosis >5 cells/µL) but can be normal." — Washington Manual of Medical Therapeutics
3. HSV PCR — definitive test, but timing matters
- Sensitivity 96%, specificity 99% when tested at 48–72 hrs after symptom onset
- Can be falsely negative in the first 24–48 hours — a negative early PCR does NOT rule out HSE
- If strong clinical suspicion + negative PCR < 72 hrs → repeat LP at 72–96 hrs
- Never stop aciclovir based on a single negative early PCR
4. Antibody testing in CSF vs serum
- For autoimmune encephalitis, CSF antibodies are more sensitive than serum for some (e.g., anti-NMDAR)
- Must be sent specifically; not part of routine CSF biochemistry
- Some antibodies (Anti-LGI1, CASPR2) are more reliably detected in serum than CSF
Practical Algorithm
CSF Result
│
├─ RBCs present + lymphocytic pleocytosis + elevated protein
│ → HSE most likely → CONTINUE aciclovir, await PCR
│
├─ Lymphocytic pleocytosis, NO RBCs, normal glucose
│ → HSE still possible (early) OR autoimmune
│ → Send HSV PCR + autoimmune antibody panel
│
└─ Completely NORMAL CSF
→ Does NOT exclude autoimmune limbic encephalitis
→ Does make HSE less likely (but not impossible early)
→ Send serum + CSF antibody panel
Important Caveat — Post-HSE Autoimmune Encephalitis
A clinically important overlap: 1–7 weeks after HSE, patients can develop a secondary anti-NMDAR encephalitis as the viral necrosis exposes previously hidden NMDAR epitopes. This presents as relapse after initial improvement. The CSF in this phase may show a new or recurrent pleocytosis with positive anti-NMDAR antibodies — treatment is immunotherapy, not more antivirals.
"Late autoimmune encephalitis can occur 1 to 7 weeks after herpes simplex encephalitis, associated with NMDA-receptor antibodies in the spinal fluid." — Goldman-Cecil Medicine
Images of both and Differential
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Loading Image
Loading Image
Loading Image
MRI Images: HSE vs Limbic Encephalitis
🔴 Herpes Simplex Encephalitis (HSE)
Image 1 — Two cases of HSE (Grainger & Allison's Diagnostic Radiology):
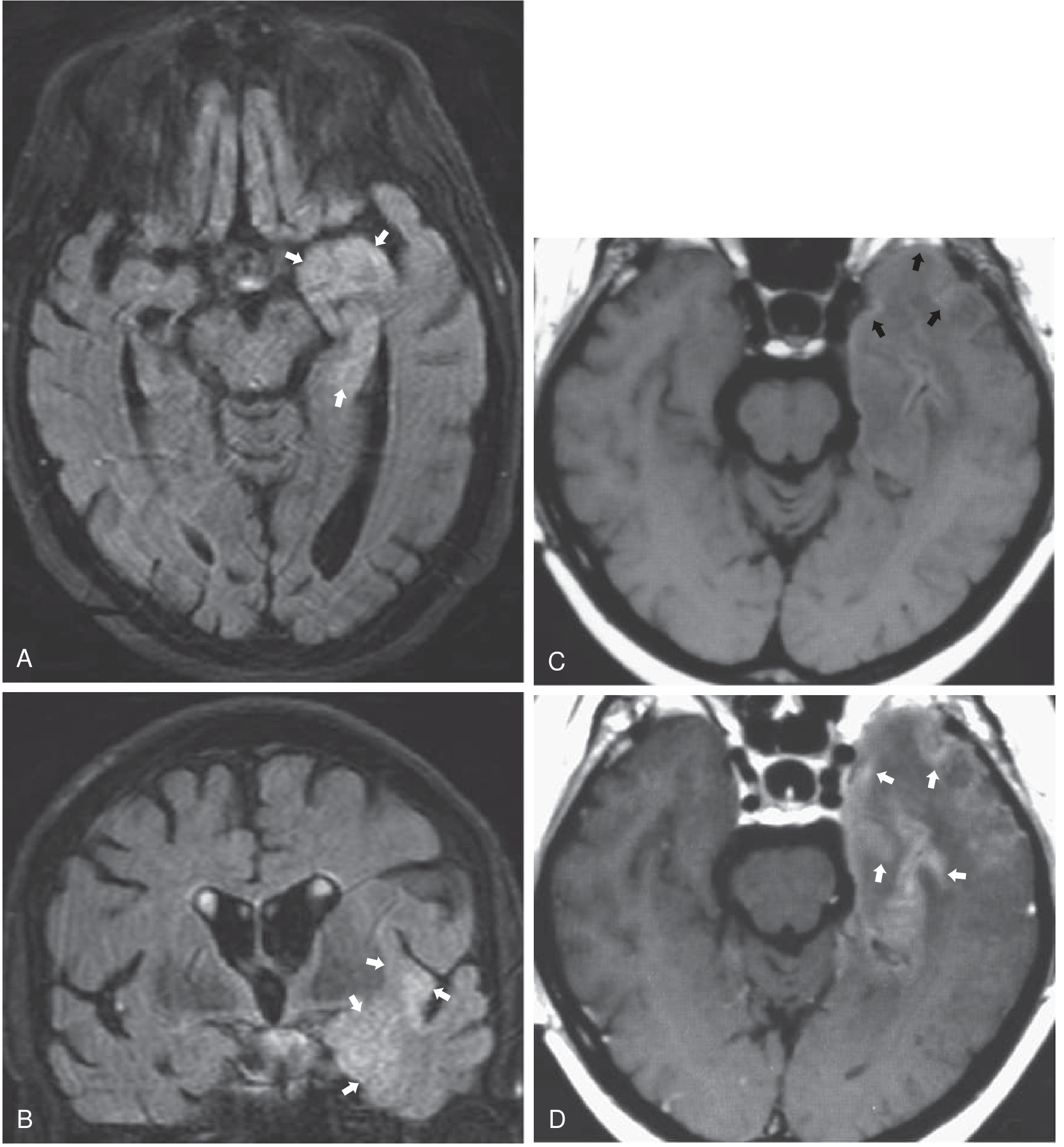
- A & B (Case 1): Axial + coronal FLAIR — hyperintense signal in the left medial temporal lobe and insula (arrows), with cortical swelling. Classic HSE territory.
- C (Case 2): T1 non-contrast — swollen left temporal lobe with faint cortical hyperintensity indicating haemorrhage (black arrows).
- D: T1 post-contrast — gyriform enhancement (white arrows) — a pattern highly suggestive of HSE.
Image 2 — Acute bilateral HSE, FLAIR + contrast (Plum & Posner):
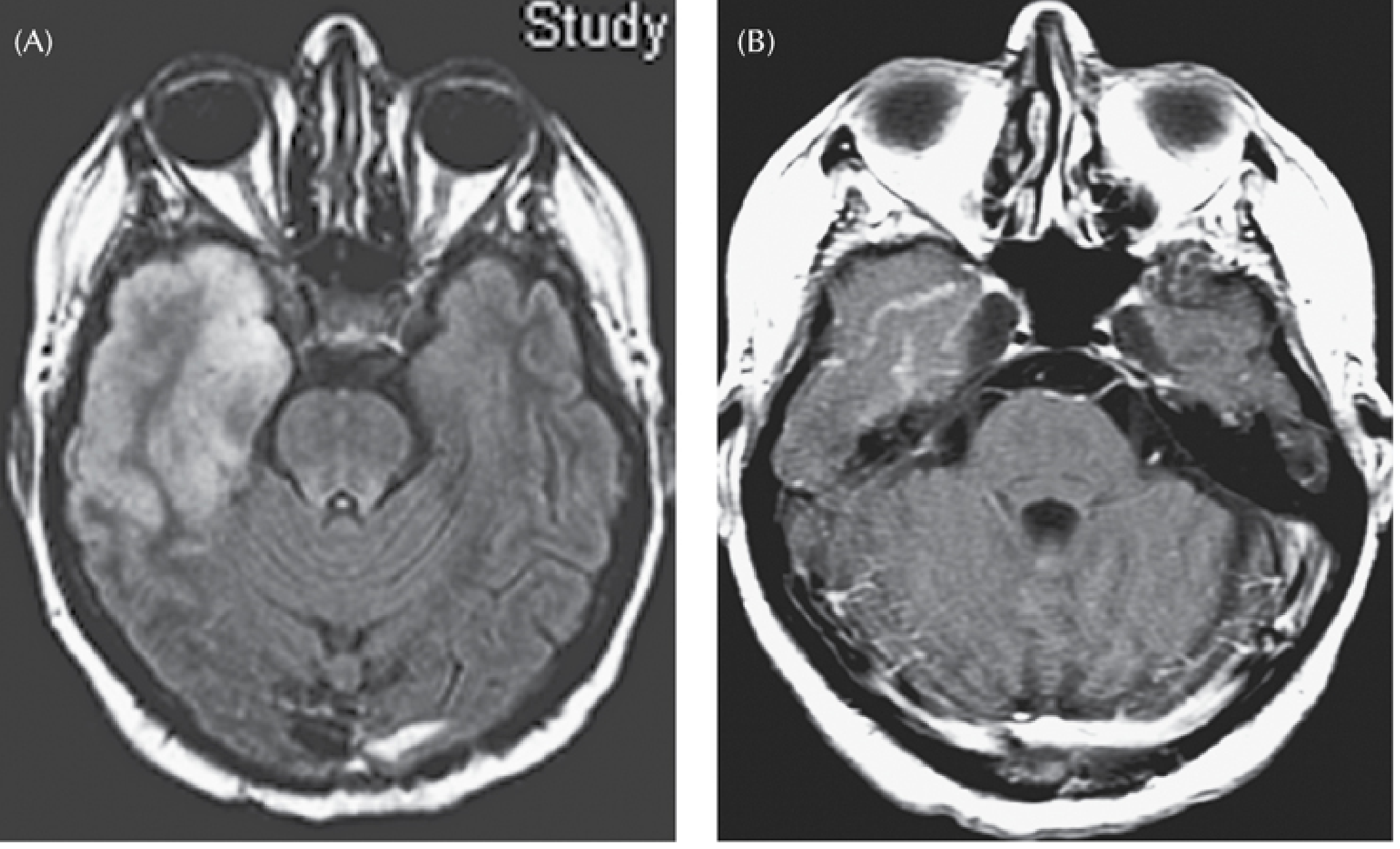
- (A) FLAIR: massive bilateral temporal lobe hyperintensity — confluent and asymmetric, consistent with fulminant HSE. Note the left temporal lobe is more severely affected.
- (B) Post-contrast T1: bilateral enhancement pattern with associated swelling.
Key radiological hallmarks of HSE: medial temporal + insula + cingulate + orbitofrontal involvement; haemorrhagic component; gyriform enhancement; grey matter > white matter.
🟠 Autoimmune / Limbic Encephalitis
Image 3 — Spectrum of autoimmune encephalitides (Harrison's Principles of Internal Medicine 22e, Fig. 99-2):
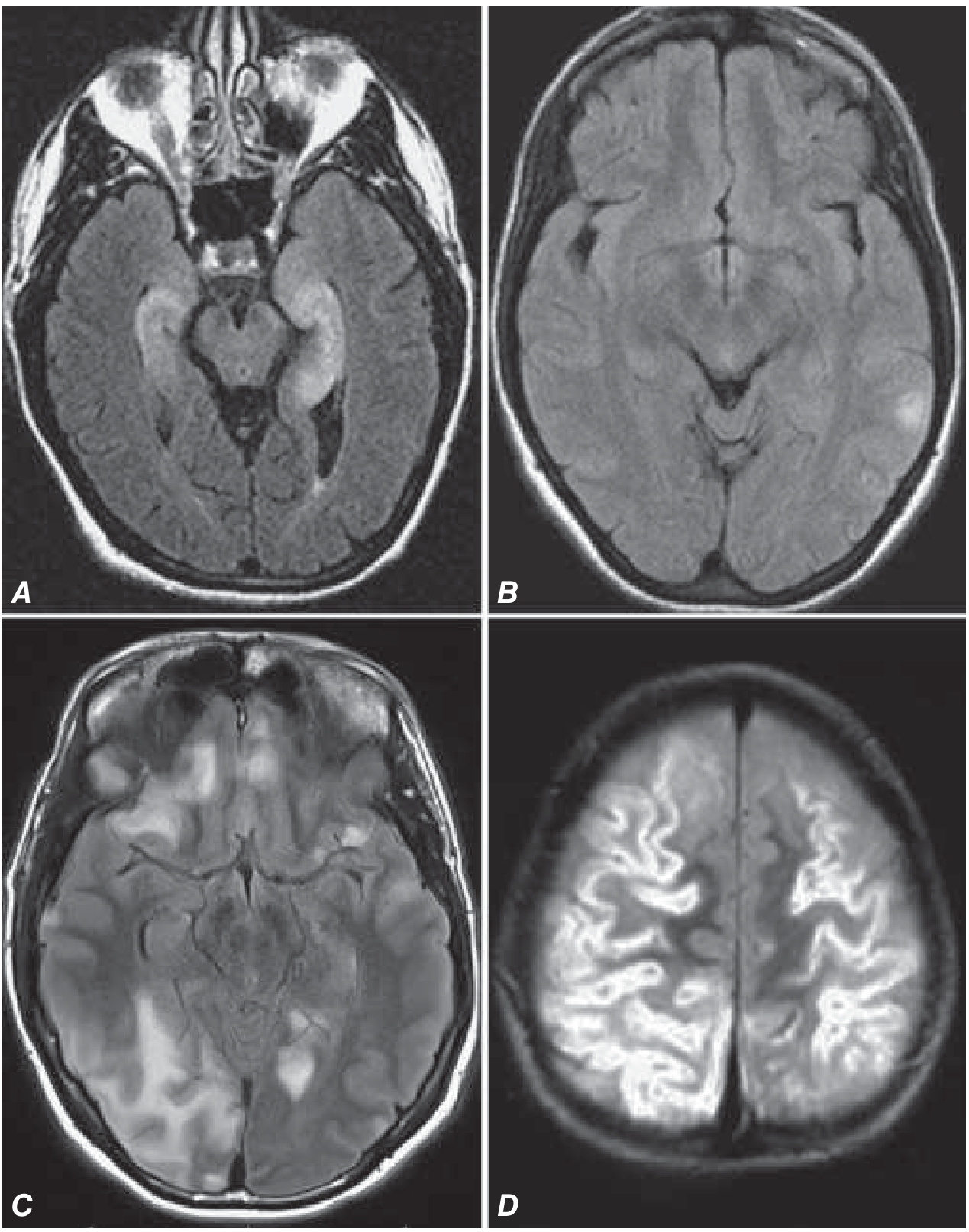
- A — Limbic Encephalitis (anti-Hu, Ma2, AMPAR, GABA-B, LGI1, Caspr2): Bilateral medial temporal lobe FLAIR hyperintensity — confined, symmetric, no haemorrhage, no gyriform enhancement. Clean signal restricted to hippocampus/amygdala.
- B — Anti-NMDAR encephalitis: Often normal MRI or only mild, subtle FLAIR changes — despite severe clinical syndrome.
- C — Anti-GABA-B encephalitis: Multiple cortical-subcortical FLAIR changes, more widespread than classic limbic encephalitis.
- D — MOG-antibody encephalitis (paediatric): Extensive cortical T2 signal, diffuse cortical involvement.
Side-by-Side Differentiating Features on MRI
| Feature | HSE | Autoimmune Limbic Encephalitis |
|---|---|---|
| Distribution | Medial temporal + insula + cingulate + orbitofrontal | Medial temporal only (hippocampus/amygdala) |
| Laterality | Unilateral → bilateral (asymmetric) | Often bilateral and symmetric |
| Haemorrhage | ✅ Frequent — T1 cortical bright spots, blooming on SWI | ❌ Absent |
| Swelling/mass effect | ✅ Marked temporal lobe swelling, can herniate | Mild or none |
| Enhancement | ✅ Gyriform / leptomeningeal (after day 3–5) | ❌ Rarely enhances |
| Grey vs white matter | Grey matter predominant (cortex + hippocampus) | Hippocampus (grey matter, but less cortical spread) |
| DWI restriction | ✅ Common (cortical restricted diffusion early) | Mild or absent |
| Insula involvement | ✅ Characteristic | ❌ Typically spared |
| Progression | Rapid (hours to days) | Subacute (days to weeks) |
| MRI may be normal | ❌ Rarely (very early) | ✅ Up to 50% initially normal |
Memory Hook
HSE = Haemorrhage + Swelling + Enhancement (gyriform) + Insula involved
Limbic Encephalitis = Limited to mesial temporal, Enhancement absent, Bilateral and symmetric
This is a shared conversation. Sign in to Orris to start your own chat.