Neural tube defect
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have comprehensive content across definition, epidemiology, pathogenesis, risk factors, classification, diagnosis, associated anomalies, screening, prognosis, and management. Let me compose a thorough answer.
Neural Tube Defects (NTDs)
Definition
An open neural tube defect (ONTD) is an embryologic defect in formation of the posterior vertebral arches of the spine, exposing the neural elements. It is also referred to as spina bifida or spinal dysraphism.
— Creasy & Resnik's Maternal-Fetal Medicine, p. 585
Embryology & Pathogenesis
The embryonic neural tube forms via neurulation — shaping, folding, and midline fusion of the neural plate — normally complete by day 25 after conception. ONTD results from failed caudal fusion of the neural tube (defect in primary neurulation), leaving neural tissue exposed.
Epidemiology
- Occurs in approximately 1:1,000 live births
- One of the most common congenital CNS malformations
Risk Factors
| Risk Factor | Odds Ratio / Risk |
|---|---|
| Prior affected pregnancy | 3–5% recurrence |
| Folate antagonists (valproic acid, carbamazepine) | ~1–2% risk |
| Folic acid deficiency (<400 mcg/day) | OR 3.72 (95% CI 1.77–7.81) |
| Pregestational diabetes (T1 or T2 DM) | OR 2.88 (95% CI 1.79–4.65) |
| Periconceptional fever (≥101°F) | OR 2.4 (95% CI 1.5–4.0) |
| Maternal obesity (BMI ≥30) | OR 1.79 (95% CI 1.51–2.13) |
| MTHFR mutation (677C>T) | OR 1.34 (95% CI 1.17–1.54) |
— Creasy & Resnik's Maternal-Fetal Medicine, p. 585
Classification
ONTD is classified by what overlies the bony defect:
| Type | Description |
|---|---|
| Myelomeningocele | Sac containing spinal cord or other neural elements (most common) |
| Meningocele | Sac containing only meninges and CSF (no neural tissue) |
| Myeloschisis | Wide splaying of vertebral arch, no covering, neural tube completely exposed |
- Location: >80% are in the lumbosacral region, but can occur anywhere along the spinal column including the cervical spine.
Cranial Defects
- Anencephaly: Absent skull and brain. A lethal defect due to failure of the anterior neuropore to close.
- Encephalocele/Cephalocele: Herniation of brain/meninges through a calvarial defect (most commonly occipital).
Associated Anomalies
Chiari II Malformation (Arnold-Chiari)
Almost always associated with lumbosacral myelomeningocele. Features include:
- Small posterior fossa
- Downward displacement of the cerebellum, pons, medulla, and cervical cord through an enlarged foramen magnum
- Medullary kinking, elongated slit-like 4th ventricle, beaking of the tectum
- Hydrocephalus: Present in 25% at birth; 80% develop it following myelomeningocele repair
Other associated findings: hydrosyringomyelia, absent corpus callosum, cortical dysplasia.
— Grainger & Allison's Diagnostic Radiology, p. 1976
Diagnosis
Prenatal Ultrasound
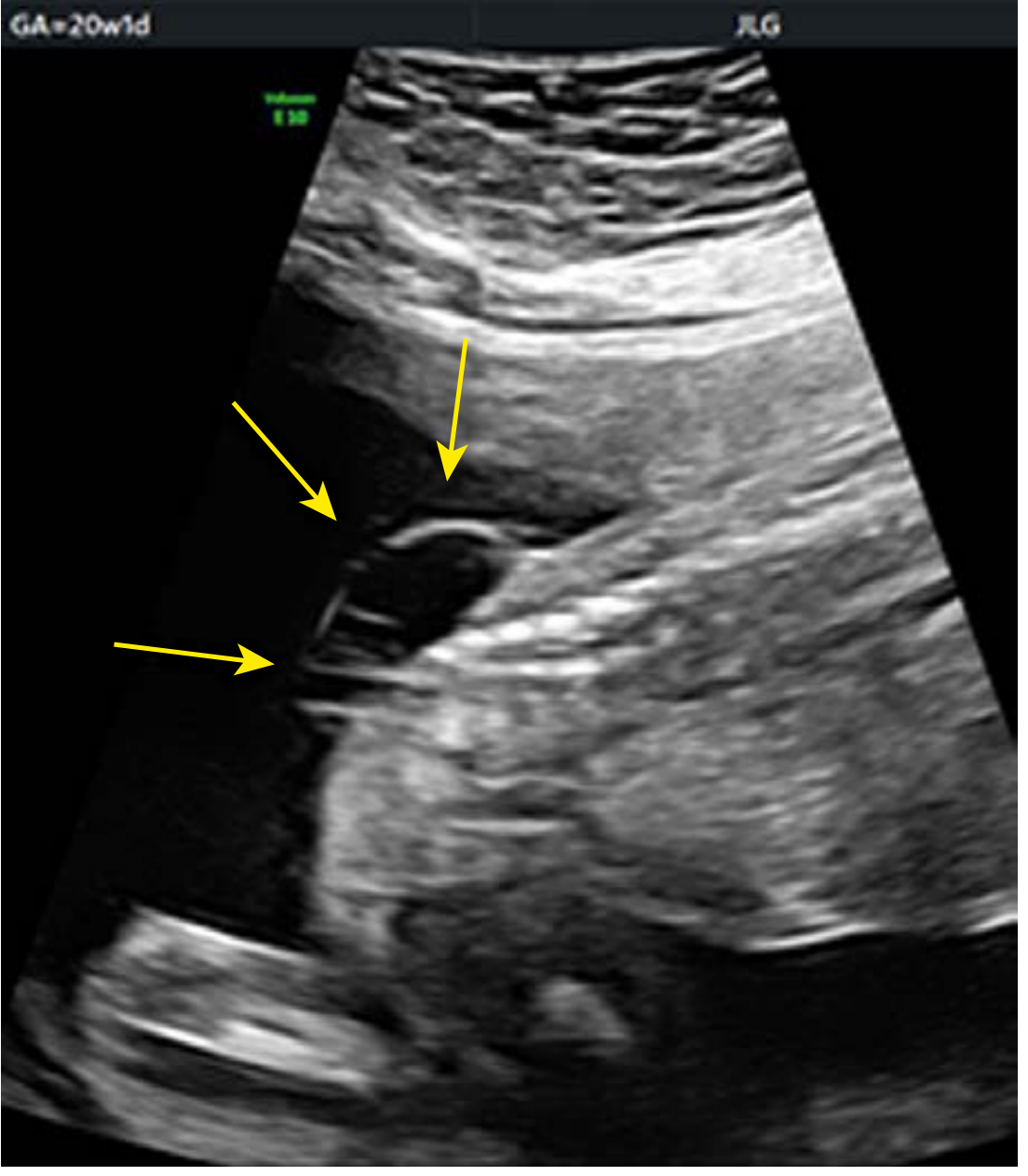
First trimester (11–14 weeks): Abnormal posterior brain on midsagittal view — non-visualization of the brainstem, 4th ventricle (intracranial translucency), or cisterna magna — indicates high risk of open spina bifida. Sensitivity ~76.5%, specificity ~99.6%.
Second trimester (standard): Spine imaged in axial, sagittal, and coronal planes.
- Sagittal: Defect in the dorsal spine with overlying cystic mass
- Axial: V- or U-shaped splaying of posterior vertebral elements
- Coronal: Splayed posterior elements with midline defect
Key intracranial signs (indirect signs of spina bifida):
- Lemon sign: Inward scalloping of frontal bones (due to low-lying cerebellum)
- Banana sign: Cerebellum wrapped around the brainstem (Chiari II)
- Ventriculomegaly / hydrocephalus
- Absent or compressed cisterna magna
Maternal Serum AFP (MSAFP)
- Elevated MSAFP (typically >2.0–2.5 MoM) at 15–20 weeks is the primary biochemical screen for ONTD
- Amniotic fluid AFP and acetylcholinesterase confirm the diagnosis
Fetal MRI
Useful for determining lesion level, size of bony defect, and associated intracranial anomalies.
Prognosis
Prognosis depends primarily on:
- Lesion level (higher = worse motor/sensory deficit)
- Lesion type (myeloschisis worse than meningocele)
- Associated anomalies (hydrocephalus, Chiari II severity)
Typical outcomes with myelomeningocele:
- Lower limb paralysis and sensory loss below the lesion level
- Neurogenic bladder and bowel (nearly universal)
- Hydrocephalus requiring VP shunt (~80% after repair)
- Cognitive outcomes range from normal to moderate delay depending on shunt complications and degree of Chiari II malformation
Management
Prevention
- Folic acid supplementation: 400 mcg/day periconceptionally for all women; 4 mg/day for women with a prior NTD-affected pregnancy (prescription-strength dose)
— Harriet Lane Handbook, p. 1959
Prenatal
- Detailed ultrasound and fetal MRI for lesion characterization
- Referral to multidisciplinary fetal center
- Fetal surgery (in utero myelomeningocele repair): The MOMS trial showed that prenatal repair (at 19–25 weeks) reduces the need for VP shunting and improves motor outcomes compared to postnatal repair. Performed at specialized centers.
- Delivery typically by cesarean section to minimize trauma to the exposed neural sac
Postnatal
- Neonatal neurosurgery: Back closure within 24–48 hours of birth to prevent infection
- Neurosurgical management of hydrocephalus (VP shunt)
- Urologic evaluation and management of neurogenic bladder
- Orthopedic and rehabilitation involvement
- Long-term multidisciplinary follow-up
Screening Summary
| Test | Timing | Marker |
|---|---|---|
| Nuchal translucency + posterior brain | 11–14 weeks | Absent IT/cisterna magna |
| MSAFP | 15–20 weeks | Elevated AFP |
| Anatomy ultrasound | 18–22 weeks | Lemon/banana signs, spinal defect |
| Amniotic fluid AFP + AChE | When needed | Confirms open defect |
Grading of power
Reading File
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
I now have all the content needed for a comprehensive answer.
Grading of Muscle Power — MRC Scale
The Medical Research Council (MRC) Scale is the universally accepted system for grading muscle power. It is a 6-point scale (0–5) with good interobserver reliability.
— Localization in Clinical Neurology, 8e, p. 32 | Bradley and Daroff's Neurology in Clinical Practice | Rockwood and Green's Fractures in Adults, 10e, p. 769
MRC Scale for Muscle Power
| Grade | Description |
|---|---|
| 0 | No muscle contraction visible |
| 1 | Flicker or trace of contraction, but no movement |
| 2 | Active joint movement when the effect of gravity is eliminated |
| 3 | Active movement against gravity (but not against resistance) |
| 4 | Active movement against gravity and resistance, but weaker than normal |
| 5 | Normal power |
Subdivision of Grade 4
Because Grade 4 covers a wide range of strength, it is often subdivided in clinical practice:
| Subgrade | Meaning |
|---|---|
| 4− | Movement against slight resistance |
| 4 | Movement against moderate resistance |
| 4+ | Movement against strong resistance (but still less than normal) |
In peripheral nerve injury contexts, M4+ is sometimes used separately to denote "strong movement, but not normal" — distinct from full M5.
Clinical Correlations
- Grade 0–1: Complete or near-complete paralysis (lower motor neuron lesion, severe neuropathy, or myopathy)
- Grade 2: Severe weakness — useful plane to test is horizontal (gravity eliminated), e.g., shoulder abduction in the supine position
- Grade 3: A clinically important threshold — the muscle can move the joint through full range against gravity; the patient can be functional for some tasks
- Grade 4: The most clinically heterogeneous grade — further subdivision (4−/4/4+) helps track recovery
- Grade 5: Normal power; defined as the ability to hold the test position against strong examiner pressure
Key Points on Testing
- Muscle testing should be standardized — the examiner applies force in the opposite direction of the movement being tested
- The patient must be positioned correctly; gravity-eliminated positions are required for Grade 2 assessment
- Grades should be recorded sequentially to monitor recovery (especially important in peripheral nerve injuries and spinal cord injuries)
- Normal grading presumes the muscle is capable of holding the test position against strong pressure from the examiner
MRC Sensory Grading (for reference)
In peripheral nerve injuries, the MRC also provides a parallel sensory grading scale:
| Grade | Description |
|---|---|
| S0 | No sensation |
| S1 | Deep pain sensation only |
| S2 | Touch, pain, and thermal sensation (protective sensation) |
| S3 | S2 + accurate localization; cold sensitivity/hypersensitivity may be present |
| S3+ | Object and texture recognition; good but not normal two-point discrimination |
| S4 | Normal sensation |
— Rockwood and Green's Fractures in Adults, 10e, p. 770–771
If paresis present below knee!
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
I have comprehensive content to answer this well. The question is about localizing weakness (paresis) below the knee — which structures can be responsible and how to differentiate them.
Paresis Below the Knee — Localization
Weakness confined to muscles below the knee points to one of four anatomical levels. Careful examination narrows the localization.
Structures Supplying Muscles Below the Knee
All muscles below the knee are supplied by branches of the sciatic nerve via two divisions that diverge in the popliteal fossa:
| Division | Muscles | Action |
|---|---|---|
| Common Peroneal (Fibular) Nerve | Tibialis anterior, EDL, EHL, peroneus longus/brevis | Dorsiflexion, eversion, toe extension |
| Tibial Nerve | Gastrocnemius, soleus, tibialis posterior, FHL, FDL, intrinsic foot muscles | Plantar flexion, inversion, toe flexion |
Differential Localization
1. Common Peroneal (Fibular) Nerve Lesion
Most common cause of foot drop
Weakness:
- Ankle dorsiflexion (foot drop)
- Toe extension
- Ankle eversion (superficial peroneal branch)
Spared:
- Ankle inversion (tibial nerve + L5 root) — key differentiating point
- Plantar flexion (tibial nerve)
- Hamstrings
Sensory loss: Dorsum of foot and lateral lower leg
Reflex: Ankle jerk preserved (tibial nerve)
Common causes: Compression at fibular head (leg crossing, plaster cast, prolonged lithotomy position), fracture of fibula head, Baker's cyst, diabetes
— Adams and Victor's Principles of Neurology, 12e, p. 1358–1359
2. Tibial Nerve Lesion
Weakness:
- Ankle plantar flexion
- Ankle inversion
- Toe flexion
- Intrinsic foot muscles
Spared:
- Dorsiflexion and eversion (peroneal)
Sensory loss: Plantar aspect of foot
Reflex: Ankle jerk absent
Complete tibial nerve lesion → calcaneovalgus deformity (foot cannot be plantar-flexed or inverted)
Tarsal tunnel syndrome (posterior tibial nerve compressed under flexor retinaculum): burning/tingling over sole, usually no motor deficit; pain worsens with standing/walking
3. Sciatic Nerve Lesion (above popliteal fossa)
Affects both peroneal and tibial distributions:
- All muscles below the knee paralyzed
- Hamstrings weakened (knee flexion impaired) — key differentiator from peroneal/tibial lesions
- Ankle jerk absent
- Sensory loss: posterior thigh, posterior/lateral leg, entire sole
Note: Partial sciatic lesions tend to preferentially involve the peroneal division (giving the false impression of a peroneal palsy)
Causes: Disc herniation (L4–5 or L5–S1), hip fracture/dislocation, gluteal injection injury, total hip arthroplasty
4. Nerve Root (Radiculopathy)
| Root | Weakness | Reflex Lost | Sensory Loss |
|---|---|---|---|
| L4 | Ankle dorsiflexion, knee extension (quadriceps) | Knee jerk | Medial leg/foot |
| L5 | Dorsiflexion + eversion + hip abduction; tibialis posterior (inversion) also affected | Medial hamstring (variable) | Lateral leg, dorsum of foot, great toe |
| S1 | Plantar flexion (gastrocnemius-soleus), toe flexion | Ankle jerk | Lateral foot, small toe |
L5 vs. peroneal nerve: Both cause foot drop. Key differentiators:
- L5 radiculopathy also weakens tibialis posterior (inversion) and hip abductors — both spared in peroneal neuropathy
- L5 may have back/buttock pain with straight leg raise
Quick Differentiating Exam
| Finding | Peroneal N. | Tibial N. | Sciatic N. | L5 Root | S1 Root |
|---|---|---|---|---|---|
| Foot drop | ✓ | — | ✓ | ✓ | — |
| Plantar flexion weak | — | ✓ | ✓ | — | ✓ |
| Inversion weak | — | ✓ | ✓ | ✓ | — |
| Eversion weak | ✓ | — | ✓ | ✓ | — |
| Hamstring weak | — | — | ✓ | ± | — |
| Ankle jerk absent | — | ✓ | ✓ | — | ✓ |
| Knee jerk absent | — | — | — | ✓ | — |
Key Clinical Pearl
Inversion of the foot is the single most useful test to distinguish peroneal nerve palsy from an L5 radiculopathy or sciatic nerve lesion:
- Inversion is preserved in peroneal palsy (tibial nerve intact)
- Inversion is impaired in L5 radiculopathy (tibialis posterior is L5-innervated via tibial nerve) and sciatic lesions
— Adams and Victor's Principles of Neurology, 12e, p. 1359 | Localization in Clinical Neurology, 8e, p. 153
Heart rate in pedia
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Excellent — I now have two authoritative tables with detailed age-based breakdowns. Here's the full answer:
Normal Heart Rate in Pediatrics (by Age)
Detailed Age-Based Table
(Adapted from Nelson Textbook of Pediatrics, via Barash Clinical Anesthesia)
| Age | Heart Rate (beats/min) | Blood Pressure (mmHg) |
|---|---|---|
| 0–3 months | 100–150 | 65–85 / 45–55 |
| 3–6 months | 90–120 | 70–90 / 50–65 |
| 6–12 months | 80–120 | 80–100 / 55–65 |
| 1–3 years | 70–110 | 90–105 / 55–70 |
| 3–6 years | 65–110 | 95–110 / 60–75 |
| 6–12 years | 60–95 | 100–120 / 60–75 |
| >12 years | 55–85 | 110–135 / 65–85 |
— Barash Clinical Anesthesia, 9e, p. 3703 (adapted from Nelson Textbook of Pediatrics)
Simplified Rosen's Emergency Medicine Table
| Age | Heart Rate (beats/min) | Respiratory Rate (breaths/min) |
|---|---|---|
| <1 year | 100–160 | 30–60 |
| 1–2 years | 90–150 | 24–40 |
| 2–5 years | 80–140 | 22–34 |
| 6–12 years | 70–120 | 18–30 |
| >12 years | 60–100 | 12–16 |
— Rosen's Emergency Medicine, p. 3066
Key Physiological Points
- Cardiac output in infants is rate-dependent: Unlike adults, neonates and infants have limited capacity to increase stroke volume (due to the high ratio of noncontractile to contractile myocardial fibers). Therefore, heart rate is the primary determinant of cardiac output in early life.
- Bradycardia is poorly tolerated in young children — it directly causes a drop in cardiac output.
- Heart rate decreases progressively with age, reaching adult values (60–100 bpm) by adolescence.
- Tachycardia in a child with hypotension most often signals hypovolemia — managed with fluids, not vasopressors (unless congenital heart disease is present).
Tachycardia / Bradycardia Thresholds (general rule)
| Definition | |
|---|---|
| Tachycardia | HR above the upper limit for age |
| Bradycardia | HR below the lower limit for age |
The younger the child, the higher the "normal" threshold — a heart rate of 110 bpm is normal in a toddler but tachycardic in an adolescent.
Lights criteriaa is seen in?
Finding Sources
Reading File
Reading File
Light's Criteria
Light's criteria are used to differentiate a pleural exudate from a transudate, applied to pleural effusion analysis. Published by Richard Light et al. in 1972, they remain the standard initial method of categorizing pleural effusions.
— Roberts and Hedges' Clinical Procedures in Emergency Medicine | Fishman's Pulmonary Diseases and Disorders
The Criteria
The pleural fluid is an EXUDATE if at least ONE of the following is present:
| Criterion | Threshold |
|---|---|
| Pleural fluid : serum protein ratio | > 0.5 |
| Pleural fluid : serum LDH ratio | > 0.6 |
| Pleural fluid LDH | > 2/3 the upper limit of normal serum LDH |
If none of the criteria are met → the fluid is a transudate.
Performance
| Feature | Value |
|---|---|
| Sensitivity | Very high (~98%) — correctly classifies nearly all exudates |
| Specificity | Lower — misclassifies up to 25% of transudates as exudates |
Important Exception — Diuretic-Treated Heart Failure
Diuretics concentrate pleural fluid protein and LDH, causing a true transudate (CHF effusion) to appear exudative by Light's criteria. In this setting, use alternative tests:
| Alternative Test | Transudate if… |
|---|---|
| Serum protein − pleural protein | > 3.1 g/dL |
| Serum albumin − pleural albumin (albumin gradient) | > 1.2 g/dL |
| Pleural fluid NT-proBNP | > 1500 pg/mL |
Causes of Transudate vs. Exudate
| Transudate | Exudate |
|---|---|
| Congestive heart failure | Pneumonia / parapneumonic effusion |
| Cirrhosis (hepatic hydrothorax) | Malignancy |
| Nephrotic syndrome | Tuberculosis |
| Peritoneal dialysis | Pulmonary embolism |
| Hypoalbuminemia | Connective tissue disease (SLE, RA) |
| Pancreatitis | |
| Chylothorax |
Bottom Line
Light's criteria = pleural effusion (transudate vs. exudate) Use all 3 criteria together; exudate = any ONE positive. Be cautious in diuretic-treated CHF — use the albumin gradient instead.
This is a shared conversation. Sign in to Orris to start your own chat.