Breathlessness u/e For 6months, housewife , using traditional cooking method with blower
Reading File
search_medical_text
biomass smoke indoor air pollution COPD housewife breathlessness
search_medical_text
chronic breathlessness exertional dyspnea diagnosis evaluation approach
search_medical_image
biomass fuel COPD chest X-ray findings
GradeResponse
GradeResponse
GradeResponse
Here is a structured clinical approach to this case:
Breathlessness on Exertion (6 Months) — Housewife Using Biomass/Blower for Cooking
Clinical Context
This is a classic presentation of biomass fuel-associated lung disease — a housewife using a traditional cooking method with a blower (chulha/wood stove) is exposed daily to high concentrations of smoke, particulates, and combustion byproducts. According to the GOLD 2025 Report (p. 21), wood, animal dung, crop residues, and coal burned in open fires or poorly functioning stoves lead to very high levels of household air pollution, and this exposure is significantly associated with COPD development, particularly in low- and middle-income countries.
Most Likely Diagnoses
| Diagnosis | Key Clue |
|---|---|
| Biomass-associated COPD | Chronic smoke exposure, progressive dyspnea |
| Biomass-associated ILD | Housewife, prolonged exposure, fine crackles |
| Pulmonary hypertension (secondary) | Long-standing hypoxia from above |
| Anaemia | Common in housewives, additive breathlessness |
| Tuberculosis | Endemic context, cough + dyspnea |
| Asthma | Variable wheeze, smoke as trigger |
History to Elicit
- Dyspnea characterization: exertional vs. rest, MRC grade, progression
- Associated symptoms: chronic cough, expectoration (mucoid/purulent), wheeze, chest tightness
- Systemic symptoms: weight loss, fever, night sweats (TB), ankle swelling (cor pulmonale)
- Exposure details: type of fuel (wood, dung cake, coal), years of use, ventilation of kitchen, hours/day of cooking
- Past history: recurrent chest infections, childhood asthma, TB treatment
- Menstrual/gynaecologic history: heavy periods (anaemia)
- Smoking: even passive exposure
Examination Findings to Look For
- General: pallor, central cyanosis, clubbing, pedal oedema
- Chest:
- Barrel-shaped chest, increased AP diameter (COPD)
- Hyperresonance on percussion
- Decreased air entry bilaterally
- Wheeze (obstructive) or fine end-inspiratory crackles (ILD)
- Loud P2, right parasternal heave (pulmonary hypertension / cor pulmonale)
- Vitals: SpO₂ on room air (may be reduced)
Investigations
First Line
| Investigation | Purpose |
|---|---|
| Spirometry (pre & post bronchodilator) | Confirm obstructive (COPD/asthma) or restrictive (ILD) pattern |
| Chest X-ray (PA view) | Hyperinflation, ILD pattern, cardiomegaly, TB |
| CBC | Anaemia, polycythaemia (chronic hypoxia) |
| SpO₂ / ABG | Degree of hypoxaemia, type II respiratory failure |
| Sputum AFB + culture | Rule out TB |
Second Line (based on findings)
| Investigation | Purpose |
|---|---|
| HRCT chest | ILD pattern (ground glass, reticulation, honeycombing) |
| ECG + Echocardiography | Pulmonary hypertension, cor pulmonale, RVH |
| 6-Minute Walk Test | Functional capacity, desaturation on exertion |
| DLCO (diffusion capacity) | Reduced in emphysema and ILD |
| Thyroid function | Hypothyroid-associated respiratory muscle weakness |
Chest X-Ray Findings in Biomass-COPD
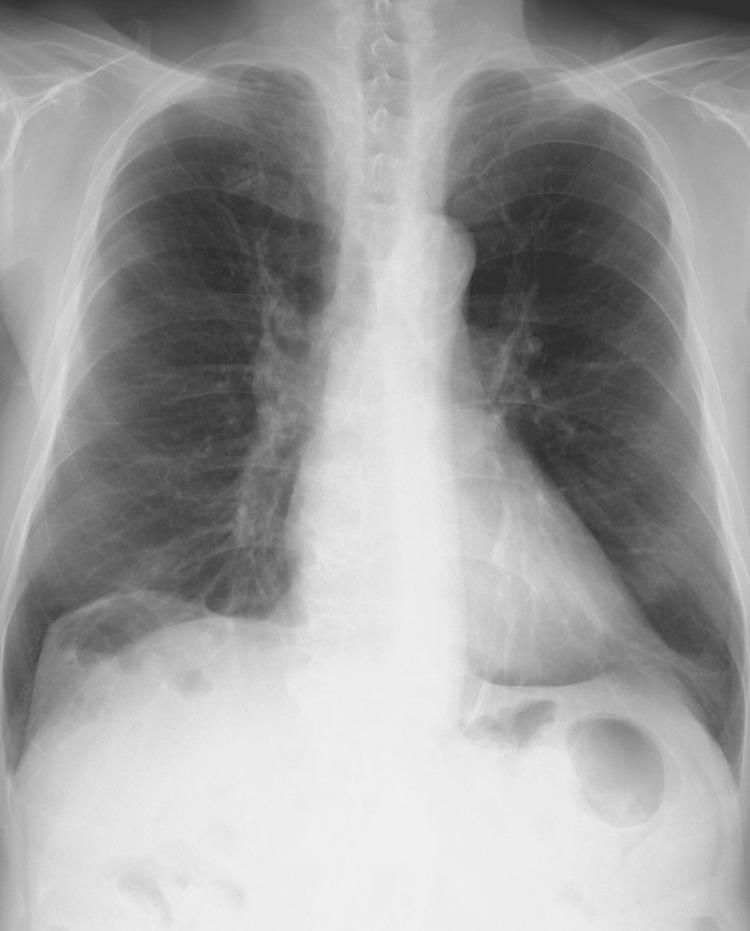
PA CXR showing hyperinflated lungs, bilaterally flattened diaphragm, vertical heart, and pruned peripheral vascular markings — classic obstructive pattern as seen in biomass COPD.
Management
Non-Pharmacological (Critical)
- Remove from exposure: switch to LPG/electric stove — GOLD 2025 (p. 21) specifically recommends gas as a transitional fuel to reduce COPD risk
- Improve kitchen ventilation: exhaust fans, chimney
- Pulmonary rehabilitation: breathing exercises, graded exercise
Pharmacological
- If COPD confirmed: SABA (salbutamol MDI) for relief; LABA + ICS or LAMA for maintenance (per GOLD ABCD grouping)
- If ILD: referral to pulmonologist; consider antifibrotics or steroids depending on subtype
- If anaemia: iron supplementation
- If TB: initiate RNTCP/DOTS regimen
Supportive
- Long-term oxygen therapy (LTOT) if PaO₂ < 55 mmHg or SpO₂ < 88% consistently
- Vaccination: influenza, pneumococcal
Key Teaching Point
Biomass-COPD (also called non-smoking COPD or "housewives' COPD") predominantly affects women in rural and semi-urban settings in South Asia, Africa, and Latin America. It is mechanistically distinct from tobacco COPD — it tends to present with a more mixed obstructive-restrictive pattern, greater small airway involvement, and higher rates of pulmonary hypertension. The blower (forced-air bellows) dramatically increases the rate of combustion and particulate generation, worsening the exposure.
This patient's 6-month progressive breathlessness warrants spirometry and HRCT at minimum before attributing it solely to COPD — ILD secondary to biomass exposure (also called biomass smoke-induced ILD or "cooking-related ILD") is an important and under-recognized entity in this demographic.
Breathlessness on walking up at morning
search_medical_text
orthopnea nocturnal dyspnea waking up breathless causes cardiac pulmonary
search_medical_text
paroxysmal nocturnal dyspnea heart failure differential diagnosis
GradeResponse
GradeResponse
This adds a critical new symptom to the clinical picture. "Waking up breathless in the morning" points toward Paroxysmal Nocturnal Dyspnea (PND) or orthopnea — both cardinal features of left heart failure / pulmonary venous congestion.
New Symptom: Waking Up Breathless at Night/Morning
What This Means Clinically
According to Harrison's Principles of Internal Medicine (p. 7144):
-
Orthopnea: Dyspnea that occurs within 1–2 minutes of lying down, relieved by sitting up/propping pillows. Caused by redistribution of fluid from lower body into the chest + decreased lung compliance in recumbency.
-
Paroxysmal Nocturnal Dyspnea (PND): Sudden awakening from sleep with breathlessness, anxiety, and suffocation — relieved by sitting upright or walking to a window. Unlike orthopnea, it occurs after prolonged recumbency (typically 2–4 hours into sleep), and is less predictable.
This Changes the Diagnostic Priority
Given this patient — a housewife with 6 months of exertional breathlessness + waking up breathless at night — the differential must now prominently include cardiac causes, particularly:
| Diagnosis | Mechanism | Key Clues |
|---|---|---|
| Left Heart Failure (HFrEF/HFpEF) | Pulmonary venous congestion | PND, orthopnea, basal crepitations, S3 gallop |
| Mitral Stenosis | Elevated LA pressure → pulmonary venous HTN | Common in women, rheumatic history, mid-diastolic murmur, PND precipitated by AF/exertion — Harrison's p. 7344 |
| Mitral Regurgitation | Volume overload → LV failure | Pansystolic murmur at apex |
| Hypertensive Heart Disease | LVH → diastolic dysfunction | BP history, S4 gallop |
| Dilated Cardiomyopathy | Global LV dysfunction | Cardiomegaly, displaced apex |
| Biomass-associated Pulmonary HTN | Chronic hypoxia → RV failure | Loud P2, RV heave |
| Nocturnal Asthma | Airway hyperreactivity peaks at 4 AM | Wheeze, diurnal variation, responds to bronchodilator |
| OSA with nocturnal hypoxaemia | Arousal from hypoxia | Snoring, obesity, daytime sleepiness |
Revised History Priorities
Ask specifically:
- How many pillows does she sleep on? (orthopnea grading — 1, 2, 3 pillow orthopnea)
- Does she wake up gasping, needing to sit/stand for relief? (PND)
- Any ankle swelling by evening? (right heart failure)
- Any palpitations? (AF — common precipitant of decompensation in mitral stenosis)
- Past history of rheumatic fever as a child? (rheumatic mitral stenosis is prevalent in this demographic)
- Wheeze on waking? (nocturnal asthma — resolves with bronchodilator)
- Snoring, waking unrefreshed? (OSA)
Revised Examination Focus
| System | Finding | Suggests |
|---|---|---|
| Pulse | Irregularly irregular | AF (mitral stenosis) |
| BP | Elevated | Hypertensive HF |
| JVP | Raised | Right/biventricular failure |
| Apex | Displaced, heaving/tapping | LV dilation / mitral stenosis |
| Heart sounds | S3 gallop, loud P2, mid-diastolic murmur | HF, pulmonary HTN, MS |
| Lungs | Bilateral basal fine crepitations | Pulmonary oedema |
| Legs | Pitting pedal oedema | Biventricular failure |
Revised Investigation Priority
| Investigation | Purpose |
|---|---|
| ECG | LVH, AF, P-mitrale, RVH |
| Chest X-ray (PA) | Cardiomegaly, pulmonary venous congestion, Kerley B lines, upper lobe diversion |
| Echocardiography (2D + Doppler) | Most important — EF, valve lesions, diastolic function, PAP |
| BNP / NT-proBNP | Elevated in heart failure; helps differentiate cardiac from pulmonary dyspnea |
| Spirometry | Still needed to quantify any obstructive/restrictive component |
| CBC | Anaemia as precipitant |
| RFT, LFT, electrolytes | Baseline before diuretics |
| Thyroid function | Thyrotoxicosis can precipitate or worsen HF |
Synthesis
This patient now has a two-hit clinical picture:
- Daytime exertional dyspnea (6 months) — biomass-COPD / ILD / anaemia
- Nocturnal/early-morning breathlessness on waking — strongly suggests pulmonary venous hypertension / left heart failure, possibly rheumatic mitral stenosis (given her demographic — young/middle-aged South Asian woman)
Echocardiography is now the single most important investigation to obtain early. Mitral stenosis in this demographic is frequently missed because the murmur is soft and requires deliberate auscultation in the left lateral decubitus position.
Clinical pearl: In a South Asian housewife with progressive dyspnea + PND/orthopnea + biomass exposure, always consider coexisting rheumatic heart disease AND biomass lung disease — both can and do occur together, and each worsens the other.
This is a shared conversation. Sign in to Orris to start your own chat.