Brudzinski sign is positive in
Reading File
Finding Sources
Reading File
Reading File
Brudzinski Sign — Positive in Meningeal Irritation
Definition of the sign:
Brudzinski's sign is elicited with the patient lying supine with limbs extended. The examiner passively flexes the neck. A positive sign = spontaneous flexion of the hips and knees in response to neck flexion.
This reflex occurs because meningeal inflammation makes stretching of the meninges painful, so the patient involuntarily draws the legs up to reduce tension on the inflamed meninges.
Conditions where Brudzinski sign is positive:
The sign is a classic indicator of meningeal irritation, seen in:
| Condition | Notes |
|---|---|
| Bacterial meningitis | Most common; sign present along with Kernig's sign and nuchal rigidity |
| Viral (aseptic) meningitis | Similar meningeal irritation |
| Tuberculous meningitis | Though classic signs may be less prominent |
| Fungal meningitis (e.g., Cryptococcus) | Especially in immunocompromised patients |
| Meningococcemia with meningitis | Kernig and Brudzinski signs may become positive |
| Subarachnoid hemorrhage (SAH) | Blood in subarachnoid space causes chemical meningeal irritation |
Classic Triad of Bacterial Meningitis (context):
Fever + Nuchal rigidity + Headache — with Kernig's and Brudzinski's signs as hallmark clinical signs of meningeal irritation.
Important caveats (from Harrison's):
- Both Kernig's and Brudzinski's signs have uncertain sensitivity and specificity
- May be absent in: very young or elderly patients, immunocompromised individuals, severely obtunded patients
- Cervical spine disease in older patients may cause false-positive nuchal rigidity
Bottom line: A positive Brudzinski sign = meningeal irritation, most classically seen in acute bacterial meningitis, but also in any condition causing subarachnoid inflammation (viral/TB/fungal meningitis, SAH).
- Harrison's Principles of Internal Medicine 22E, p. 1163
- Gray's Anatomy for Students, Case 8
- Neuroanatomy through Clinical Cases, 3rd Ed., Table 5.6
Lipoprotein metabolism
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
Reading File
Reading File
Reading File
Reading File
Reading File
Loading Image
I now have comprehensive content from multiple authoritative textbooks. Here is a complete, well-structured answer:
Lipoprotein Metabolism
Structure of Lipoproteins
Lipoproteins are macromolecular assemblies that transport hydrophobic lipids (triglycerides [TG] and cholesterol esters) through the aqueous plasma. Their general structure consists of:
- Hydrophobic core: TG + cholesteryl esters
- Hydrophilic shell: phospholipids + unesterified cholesterol + apolipoproteins
They are classified by density (inversely related to lipid content):
| Lipoprotein | Density | Major Lipid | Key Apolipoprotein | Origin |
|---|---|---|---|---|
| Chylomicron | Lowest | TG (~90%) | ApoB-48 | Intestine |
| VLDL | Very low | TG (~60%) | ApoB-100 | Liver |
| IDL | Intermediate | TG + Cholesterol | ApoB-100, ApoE | VLDL remnant |
| LDL | Low | Cholesterol (~70% of plasma cholesterol) | ApoB-100 | IDL remnant |
| HDL | Highest | Cholesterol ester | ApoA-I | Liver + Intestine |
Key Apolipoproteins and Their Functions
| Apolipoprotein | Source | Lipoprotein | Function |
|---|---|---|---|
| ApoB-48 | Intestine | Chylomicrons | Structural; does NOT bind LDL receptor |
| ApoB-100 | Liver | VLDL, IDL, LDL | Structural; ligand for LDL receptor |
| ApoC-II | Liver/HDL | Chylomicrons, VLDL | Activates LPL |
| ApoC-III | Liver/HDL | Chylomicrons, VLDL | Inhibits LPL |
| ApoE | Liver/HDL | Chylomicron remnants, IDL | Ligand for hepatic remnant receptor and LDL receptor |
| ApoA-I | Liver + Intestine | HDL | Structural; activates LCAT; promotes ABCA1-mediated cholesterol efflux |
| ApoA-V | Liver | VLDL, chylomicrons | Facilitates LPL activity |
The Three Metabolic Pathways
1. Exogenous Pathway (Dietary Lipids → Chylomicrons)
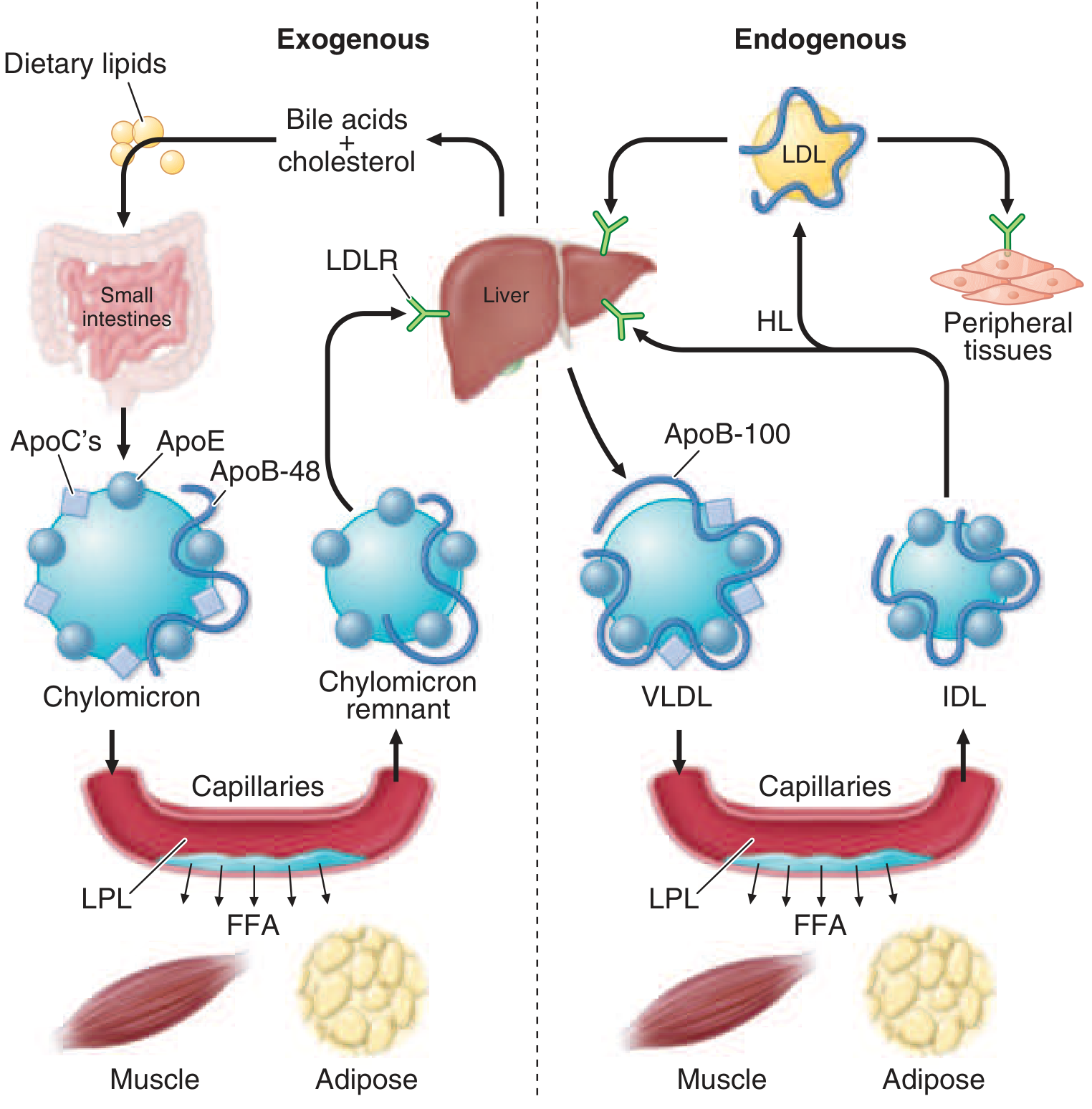
- Dietary fat is digested and absorbed in the proximal small intestine
- Fatty acids + cholesterol are re-esterified in enterocytes and loaded onto ApoB-48 by MTP (microsomal TG transfer protein) → forms nascent chylomicrons
- Chylomicrons enter the lymphatics → thoracic duct → left subclavian vein → systemic circulation
- In the blood, nascent chylomicrons receive ApoC-II and ApoE from circulating HDL
- LPL (on capillary endothelium in adipose, cardiac, skeletal muscle), activated by ApoC-II, hydrolyzes the core TGs → releases free fatty acids (FFAs)
- FFAs → taken up by adipocytes (storage) or myocytes (energy)
- Glycerol → taken up by liver
- As >90% of TG is removed, the shrunken particle becomes a chylomicron remnant (retains ApoB-48 and ApoE; loses ApoC apolipoproteins back to HDL)
- Chylomicron remnants are rapidly cleared from blood by the liver via ApoE binding to hepatic LDL receptors (remnant receptor)
- After a 12-hour fast, virtually no chylomicrons remain in blood
2. Endogenous Pathway (Hepatic Lipids → VLDL → IDL → LDL)
- VLDL is assembled in the liver: ApoB-100 + TG + phospholipids + cholesteryl esters, loaded by MTP → secreted directly into blood
- Nascent VLDL acquires ApoC-II and ApoE from HDL
- LPL (activated by ApoC-II) hydrolyzes VLDL-TG at capillaries → FFAs released to tissues
- As TG is depleted, VLDL → IDL (VLDL remnant); IDL has roughly equal TG and cholesterol
- ~40–60% of IDL is taken up by the liver via ApoE → LDL receptor (receptor-mediated endocytosis)
- Remaining IDL is remodeled by hepatic lipase (HL) → LDL
- During this remodeling, CETP (cholesteryl ester transfer protein) transfers cholesteryl esters from HDL → VLDL/IDL in exchange for TG
- LDL is the major cholesterol carrier in plasma (~70% of plasma cholesterol). It contains primarily ApoB-100
- LDL is cleared by LDL receptor-mediated endocytosis primarily in the liver:
- ApoB-100 binds LDL receptors (clustered in clathrin-coated pits)
- Endocytosis → endosome → lysosomal degradation → free cholesterol released
- Receptor is recycled; free cholesterol downregulates HMG-CoA reductase and LDL receptor synthesis (feedback)
- PCSK9 (proprotein convertase subtilisin/kexin type 9) promotes lysosomal degradation of the LDL receptor → reduces receptor recycling → raises plasma LDL
3. Reverse Cholesterol Transport (RCT) — HDL Pathway
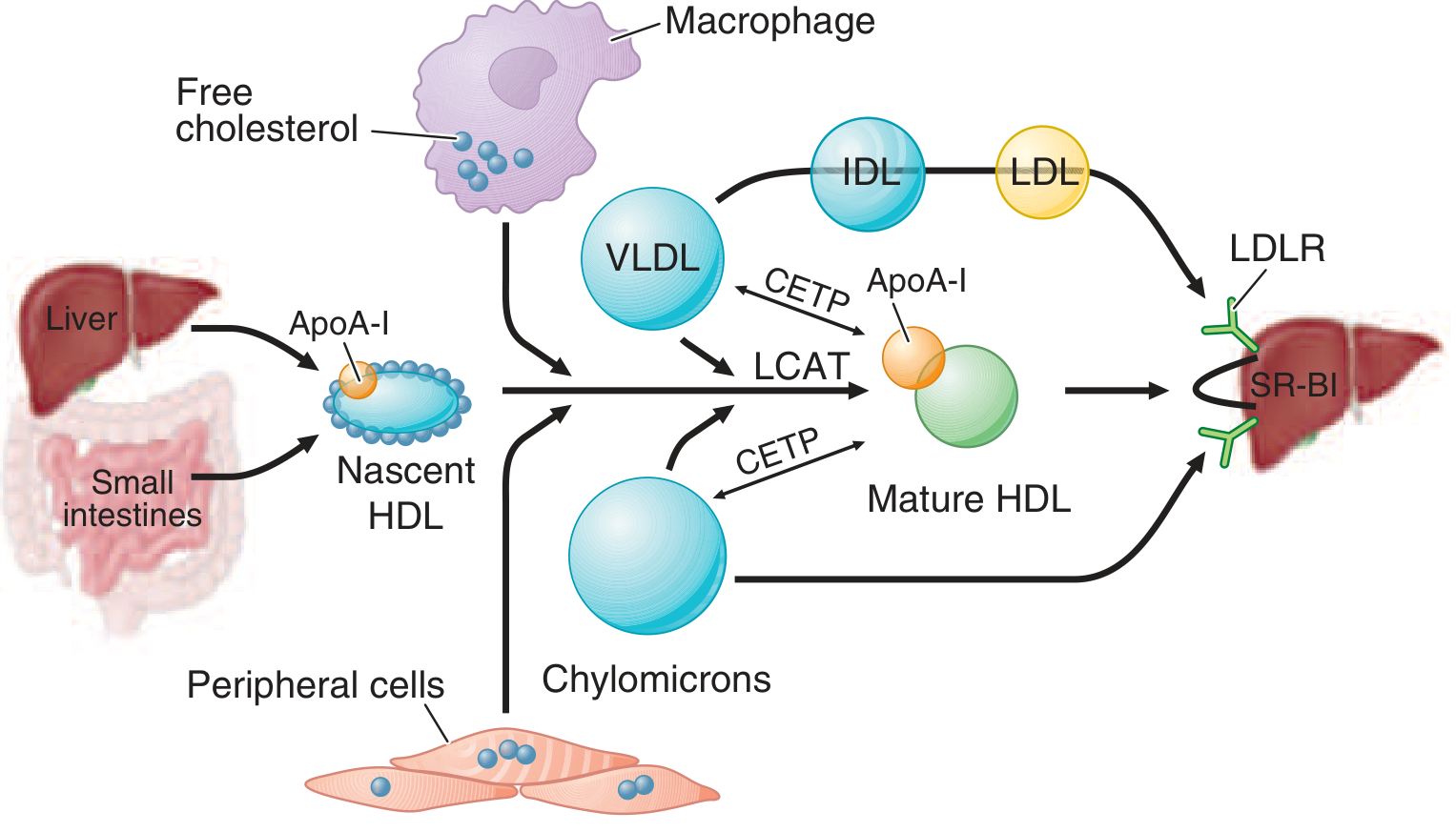
- Nascent HDL (discoidal) is secreted by the liver and intestine, primarily as ApoA-I
- ApoA-I promotes cholesterol efflux from peripheral cells (including macrophages in arterial walls) via ABCA1 and ABCG1 transporters
- LCAT (lecithin:cholesterol acyltransferase), activated by ApoA-I, esterifies free cholesterol → cholesteryl ester → sequestered in HDL core → particle becomes spherical (mature HDL / HDL3 → HDL2)
- HDL delivers cholesterol to the liver via two routes:
- Direct: SR-BI (scavenger receptor class B1) on hepatocytes selectively uptakes CE; HDL particle is recycled
- Indirect via CETP: CETP transfers CE from HDL → apoB-containing lipoproteins (VLDL, LDL) in exchange for TG → LDL receptor clears these particles in liver
- Cholesterol is excreted in bile (directly or as bile acids) — the only route for net cholesterol excretion from the body
This "RCT" is the mechanistic basis for the inverse relationship between plasma HDL and atherosclerotic risk — HDL removes cholesterol from atherosclerotic plaques.
Key Enzymes Summary
| Enzyme | Location | Substrate | Activated by | Role |
|---|---|---|---|---|
| LPL (lipoprotein lipase) | Capillary endothelium (adipose, muscle) | TG in chylomicrons + VLDL | ApoC-II | Releases FFAs to tissues |
| HL (hepatic lipase) | Hepatic sinusoidal endothelium | TG + phospholipids in IDL, HDL | — | IDL → LDL; HDL2 → HDL3 |
| LCAT | Plasma (HDL-bound) | Cholesterol on HDL | ApoA-I | Esterifies cholesterol in HDL |
| CETP | Plasma | CE (HDL) ↔ TG (VLDL) | — | Transfers CE from HDL to VLDL/LDL |
| MTP | ER (liver + intestine) | Lipid loading of ApoB | — | Assembles VLDL + chylomicrons |
| ACAT | Intracellular | Cholesterol | — | Stores excess cholesterol as esters |
Clinically Important Disorders
| Defect | Result | Disorder |
|---|---|---|
| LPL or ApoC-II deficiency | Massive hypertriglyceridemia (chylomicronemia) | Type I hyperlipoproteinemia (familial chylomicronemia); pancreatitis risk |
| LDL receptor defect | Elevated LDL-C | Familial hypercholesterolemia (FH); premature ASCVD |
| PCSK9 gain of function | LDL receptor degraded → high LDL | Autosomal dominant hypercholesterolemia |
| ApoE absence/defect | Impaired clearance of remnants | Type III hyperlipoproteinemia (broad beta disease) |
| MTP defect | No VLDL/chylomicron assembly | Abetalipoproteinemia; TG accumulates in liver/intestine; low fat-soluble vitamins |
| LCAT deficiency | Impaired cholesterol esterification in HDL | Low HDL, corneal opacities, hemolytic anemia |
Sources: Lippincott's Illustrated Reviews: Biochemistry, 8th ed. — Chapter 18; Harrison's Principles of Internal Medicine, 22E — Chapter 419; Goodman & Gilman's Pharmacological Basis of Therapeutics
This is a shared conversation. Sign in to Orris to start your own chat.