Hemochromatosis
Reading File
search_medical_text
hemochromatosis pathophysiology genetics classification types
search_medical_text
hemochromatosis diagnosis clinical features management treatment
search_medical_image
hemochromatosis liver iron deposits pathology
GradeResponse
GradeResponse
GradeResponse
Hemochromatosis
Overview
Hemochromatosis is a disorder of iron overload characterized by progressive accumulation of iron in parenchymal organs — primarily the liver, heart, pancreas, pituitary, and joints — leading to tissue damage and organ failure. The most common form is hereditary hemochromatosis (HH), an autosomal recessive condition caused by mutations in the HFE gene.
Classification
| Type | Gene Affected | Inheritance | Notes |
|---|---|---|---|
| Type 1 (HFE-related) | HFE (C282Y, H63D) | AR | Most common in Northern Europeans |
| Type 2A (Juvenile HH) | HJV (hemojuvelin) | AR | Severe; presents in 2nd–3rd decade |
| Type 2B (Juvenile HH) | HAMP (hepcidin) | AR | Similar to 2A |
| Type 3 | TfR2 (transferrin receptor 2) | AR | Moderate severity |
| Type 4 (Ferroportin disease) | SLC40A1 (ferroportin) | AD | Unique pattern |
| Secondary hemochromatosis | N/A | — | Transfusion-dependent anemias, chronic liver disease, dietary iron overload |
Pathophysiology
Normally, the body maintains 3–4 g of iron by matching intestinal absorption (~1 mg/day in men, ~1.5 mg/day in menstruating women) to daily losses. The key regulatory hormone is hepcidin — a liver-derived peptide that suppresses basolateral iron export from intestinal enterocytes and iron release from macrophages by binding and internalizing ferroportin.
In hemochromatosis, hepcidin is inappropriately low (or ferroportin is hepcidin-resistant in Type 4), resulting in unregulated intestinal absorption of ≥4 mg/day. Iron progressively saturates transferrin and accumulates in parenchymal cells, generating reactive oxygen species via the Fenton reaction, causing lipid peroxidation, DNA damage, and ultimately fibrosis and organ failure. (Harrison's, p. 11768)
Clinical Features
Symptoms typically appear in the 4th–5th decade in men (later in women due to menstrual iron losses). Classic triad: cirrhosis + diabetes + skin bronzing ("bronze diabetes") — though this represents end-stage disease.
Organ-specific manifestations:
- Liver: Hepatomegaly → cirrhosis → hepatocellular carcinoma (HCC; 200× increased risk)
- Pancreas: Diabetes mellitus (iron-induced β-cell dysfunction)
- Heart: Dilated cardiomyopathy, arrhythmias, heart block
- Pituitary/Gonads: Hypogonadotropic hypogonadism, loss of libido, impotence, amenorrhea
- Skin: Hyperpigmentation (bronze/slate-gray)
- Joints: Arthropathy (especially 2nd and 3rd MCP joints) — often the earliest symptom
- Thyroid/Adrenals: Hypothyroidism, adrenal insufficiency
Diagnosis
Lab Workup
| Test | Finding in HH | Threshold |
|---|---|---|
| Transferrin saturation (fasting) | Elevated | >45% (screening cutoff) |
| Serum ferritin | Elevated | >300 μg/L (men), >200 μg/L (women) |
| Liver enzymes (ALT/AST) | Mildly elevated | — |
| Blood glucose | Elevated (if diabetic) | — |
Genetic Testing
- HFE C282Y homozygosity confirms Type 1 HH in the right clinical context
- Compound heterozygosity (C282Y/H63D) causes milder iron loading
Liver Biopsy
Indicated when: ferritin >1000 μg/L, elevated transaminases, or hepatomegaly — to assess fibrosis stage and hepatic iron index (HII ≥1.9 mol/g/year supports HH).
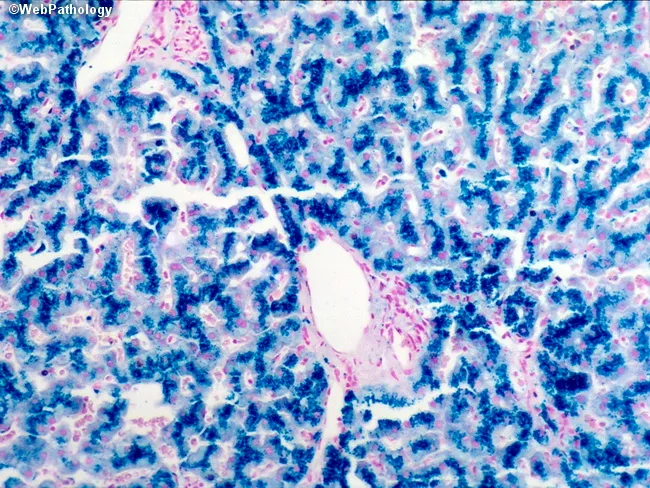
Prussian Blue stain reveals coarse blue iron granules concentrated in periportal hepatocytes (unlike secondary iron overload, which preferentially loads Kupffer cells):
MRI
T2*-weighted MRI of the liver quantifies iron without biopsy and is increasingly used for noninvasive staging.
Management
1. Phlebotomy (First-Line)
The cornerstone of treatment. (Harrison's, p. 11778)
- Induction phase: 500 mL whole blood (200–250 mg iron) removed weekly (or twice-weekly for gross overload)
- Packed red cell volume may dip to ~35 mL/dL initially, then stabilizes
- Transferrin saturation remains elevated until iron stores are depleted
- Target: serum ferritin ≤100 μg/L
- Duration: typically 1–2 years for advanced disease (≥25 g iron may need to be removed)
- Maintenance phase: Phlebotomy every 2–4 months to keep ferritin ≤100 μg/L
2. Chelation Therapy
Reserved for patients unable to tolerate phlebotomy (e.g., anemia, cardiovascular instability):
- Deferoxamine (IV/SC)
- Deferasirox (oral)
3. Management of End-Organ Damage
| Complication | Management |
|---|---|
| Cirrhosis | Surveillance for HCC (ultrasound + AFP every 6 months) |
| Diabetes | Standard insulin/oral hypoglycemics |
| Cardiomyopathy | Standard heart failure therapy |
| Hypogonadism | Testosterone/estrogen replacement |
| Arthropathy | NSAIDs; joint replacement if severe |
4. Dietary Advice
- Avoid vitamin C supplements (enhances iron absorption)
- Avoid alcohol (synergistic hepatotoxicity)
- Avoid raw shellfish (risk of Vibrio vulnificus infection — iron overload increases susceptibility)
5. Liver Transplantation
For end-stage liver disease; outcomes are inferior compared to other indications (due to cardiac and endocrine comorbidities).
Prognosis
- If treated before cirrhosis develops: normal life expectancy
- If cirrhosis is present at diagnosis: increased mortality, especially from HCC (does not fully regress with iron depletion) and hepatic failure
- Phlebotomy reverses or halts: fatigue, hepatomegaly, skin pigmentation, cardiac dysfunction, and some endocrine dysfunction
- Arthropathy and hypogonadism may not improve after treatment
Family Screening
First-degree relatives of a C282Y homozygote should be screened with:
- Transferrin saturation + ferritin
- HFE genotyping
This is a shared conversation. Sign in to Orris to start your own chat.