Nephrotic syndrome
Reading File
search_medical_text
nephrotic syndrome etiology pathophysiology clinical features diagnosis management
search_medical_image
nephrotic syndrome histology glomerular pathology
GradeResponse
GradeResponse
Nephrotic Syndrome
Definition
Nephrotic syndrome is defined by the classic triad of:
| Feature | Threshold |
|---|---|
| Proteinuria | >3.5 g/day (or >40 mg/m²/hr in children) |
| Hypoalbuminemia | Serum albumin <3.5 g/dL |
| Edema | Peripheral, periorbital, ascites, pleural effusion |
Additionally associated with hyperlipidemia (high LDL, low HDL) and lipiduria (Harrison's, p. 8392).
Pathophysiology
The central defect is glomerular barrier dysfunction, leading to loss of the size- and charge-selective filtration properties of the glomerular basement membrane (GBM) and podocytes.
Glomerular injury
↓
Loss of podocyte integrity / GBM charge barrier
↓
Massive proteinuria (>3.5 g/day)
↓
↓ Oncotic pressure (hypoalbuminemia)
↓
Fluid shift into interstitium → Edema
↓
Compensatory hepatic lipoprotein synthesis → Hyperlipidemia
↓
Lipiduria (oval fat bodies, Maltese crosses under polarized light)
Sodium and water retention occurs via both the underfill mechanism (low oncotic pressure → RAAS activation) and the overfill mechanism (primary renal Na⁺ retention).
Etiology
Primary (Idiopathic) Glomerulopathies
| Cause | Key Features | Age Group |
|---|---|---|
| Minimal Change Disease (MCD) | Normal LM, effacement of foot processes on EM | Children (most common), adults |
| Focal Segmental Glomerulosclerosis (FSGS) | Segmental scarring of glomeruli; may be primary or secondary | Adults, Black patients |
| Membranous Nephropathy (MN) | Subepithelial deposits, "spike and dome" on EM; anti-PLA2R Ab | Adults (most common in white adults) |
| Membranoproliferative GN (MPGN) | Mesangial proliferation, double-contour GBM | Any age |
| IgA Nephropathy | Mesangial IgA deposits; usually nephritic but can be nephrotic | Young adults |
Secondary Causes
| Category | Examples |
|---|---|
| Diabetes mellitus | Diabetic nephropathy (most common secondary cause worldwide) |
| Systemic diseases | SLE (Class V lupus nephritis), amyloidosis, sarcoidosis |
| Infections | HBV (membranous), HCV (MPGN), HIV (collapsing FSGS), malaria |
| Malignancy | Hodgkin lymphoma (MCD), solid tumors (membranous) |
| Drugs | NSAIDs, gold, penicillamine, heroin (FSGS) |
| Hereditary | Alport syndrome, congenital nephrotic syndrome (NPHS1/NPHS2 mutations) |
Clinical Features
- Edema: Periorbital (classically worse in the morning), dependent edema, anasarca; ascites; pleural effusions
- Foamy urine: Due to heavy proteinuria
- Weight gain: Fluid retention
- Lipiduria: Oval fat bodies with Maltese cross appearance under polarized light; fatty casts on urinalysis (Harrison's, p. 8392)
Complications
| Complication | Mechanism |
|---|---|
| Hypercoagulability / Thrombosis | Loss of antithrombin III, protein C/S; renal vein thrombosis, DVT, PE |
| Infections | Loss of immunoglobulins (IgG) and complement; especially encapsulated organisms (S. pneumoniae) |
| Hyperlipidemia / Atherosclerosis | Compensatory hepatic synthesis of VLDL/LDL |
| AKI | Reduced effective circulating volume; drug toxicity |
| Vitamin D deficiency | Loss of vitamin D-binding protein |
| Hypothyroidism | Loss of thyroid-binding globulin |
Diagnosis
Workup Algorithm
- Urinalysis — proteinuria (3–4+), oval fat bodies, fatty/waxy casts
- Urine protein:creatinine ratio (UPCR) — >3.5 mg/mg confirms nephrotic-range
- 24-hour urine protein — >3.5 g/day (gold standard quantification)
- Serum albumin — <3.5 g/dL
- Lipid panel — elevated total cholesterol, LDL; low HDL
- BMP/CMP — renal function, electrolytes
- Serology panel to identify secondary causes:
- ANA, anti-dsDNA, complement (C3/C4) → SLE
- HBsAg, HCV Ab → viral
- HIV
- Serum protein electrophoresis (SPEP) → myeloma/amyloid
- Anti-PLA2R antibodies → primary membranous nephropathy
- Fasting glucose/HbA1c → diabetes
- Renal biopsy — indicated in adults to determine underlying cause and guide treatment; generally not done in children with typical MCD presentation (empiric steroids first)
Urinalysis Findings
| Finding | Significance |
|---|---|
| Proteinuria 3–4+ | Hallmark |
| Oval fat bodies | Lipid-laden tubular cells |
| Maltese crosses (polarized light) | Cholesterol crystals |
| Fatty casts | Lipiduria |
| Absence of RBC casts | Distinguishes from nephritic syndrome |
Nephrotic vs. Nephritic Syndrome
| Feature | Nephrotic | Nephritic |
|---|---|---|
| Proteinuria | Massive (>3.5 g/day) | Mild–moderate (<3.5 g/day) |
| Hematuria | Absent or microscopic | Gross hematuria, RBC casts |
| Hypertension | Variable | Common |
| Edema | Severe | Mild |
| Serum complement | Usually normal | Often low (MPGN, SLE, post-strep) |
| GFR | May be normal | Often reduced |
Management
General Measures
- Low-sodium diet (<2 g/day) to reduce edema
- Fluid restriction if symptomatic hyponatremia
- Diuretics (loop diuretics: furosemide) — may need IV if gut edema impairs absorption
- ACE inhibitors / ARBs — reduce proteinuria and intraglomerular pressure (regardless of BP)
- Statins — manage hyperlipidemia
- Anticoagulation — consider if albumin <2.5 g/dL (high VTE risk); definitely if renal vein thrombosis confirmed
Disease-Specific Treatment
| Cause | Treatment |
|---|---|
| MCD | Prednisone (1 mg/kg/day); most respond within 8 weeks; cyclophosphamide or cyclosporine for relapsing/steroid-dependent |
| FSGS (Primary) | High-dose corticosteroids; CNI (tacrolimus/cyclosporine) for steroid-resistant; sparsentan (novel dual ET/angiotensin receptor blocker) |
| Membranous Nephropathy | Observation if low risk; for high-risk: rituximab (now first-line), cyclophosphamide + steroids (Ponticelli regimen), or CNIs |
| Diabetic Nephropathy | Glycemic control, RAAS blockade, SGLT2 inhibitors (empagliflozin, dapagliflozin), finerenone |
| Lupus Nephritis (Class V) | Hydroxychloroquine + mycophenolate mofetil ± voclosporin or belimumab |
| Amyloidosis | Treat underlying cause (anti-plasma cell therapy for AL); tafamidis for ATTR |
Histology of a Secondary Cause (Amyloidosis)
The image below demonstrates renal biopsy changes in AL amyloidosis, an important secondary cause of nephrotic syndrome:
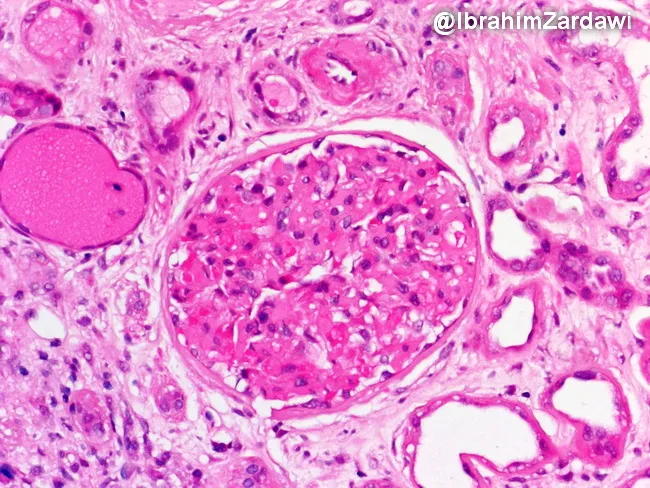
Renal biopsy (H&E): amorphous eosinophilic deposits in the interstitium and mesangial expansion with GBM thickening. Congo red staining confirms amyloid by apple-green birefringence under polarized light. This pattern underlies nephrotic-range proteinuria and progressive renal insufficiency (Webpathology).
Prognosis
Varies widely by underlying cause:
- MCD: Excellent — >90% remission with steroids; can relapse
- Primary Membranous: ~1/3 spontaneous remission, ~1/3 stable, ~1/3 progress to ESRD
- FSGS: Worst prognosis among primary causes; ~50% ESRD within 10 years if steroid-resistant
- Diabetic Nephropathy: Progressive; SGLT2 inhibitors significantly slow progression
- Amyloidosis: Depends on systemic burden and response to therapy
Key takeaway: Nephrotic syndrome is a clinical diagnosis; the underlying cause requires biopsy (in adults) and dictates treatment. Complications — especially VTE and infection — must be proactively managed regardless of etiology.
This is a shared conversation. Sign in to Orris to start your own chat.