Ocular manifestations of diebetes mellitus . Take reference from khurana , kanski & zia & datta
Reading File
search_medical_text
ocular manifestations of diabetes mellitus diabetic retinopathy
search_medical_text
diabetic eye disease lens cataract neovascular glaucoma optic nerve
search_medical_image
diabetic retinopathy fundus ocular manifestations diabetes
GradeResponse
GradeResponse
GradeResponse
Now I have solid foundational content. Let me compile a comprehensive, well-structured answer drawing from the retrieved sources and supplemented with knowledge from Khurana, Kanski, and Zia & Datta.
Ocular Manifestations of Diabetes Mellitus
References: Khurana's Comprehensive Ophthalmology | Kanski's Clinical Ophthalmology | Zia & Datta's Ophthalmology | Eye Care of the Patient with Diabetes Mellitus (AAO)
Overview
Diabetes mellitus (DM) affects virtually every structure of the eye. It is one of the leading causes of preventable blindness worldwide. Diabetic retinopathy (DR) — the most clinically significant manifestation — is classically a microvascular disease, though growing evidence confirms that retinal neurodegeneration precedes visible vascular lesions (AAO Diabetic Retinopathy, p. 12).
Classification of Ocular Manifestations
| Structure Affected | Manifestation |
|---|---|
| Lids | Recurrent styes, xanthelasma |
| Cornea | Reduced corneal sensitivity, recurrent erosions, diabetic keratopathy |
| Iris | Rubeosis iridis (neovascularization of iris) |
| Lens | Metabolic (snowflake) cataract, senile cataract (earlier onset), refractive changes |
| Aqueous humor | Neovascular glaucoma, open-angle glaucoma |
| Vitreous | Vitreous hemorrhage |
| Retina | Diabetic retinopathy (NPDR + PDR), diabetic macular edema (DME) |
| Optic nerve | Diabetic papillopathy, anterior ischemic optic neuropathy (AION) |
| Extraocular muscles | Diabetic cranial nerve palsies (CN III, IV, VI) |
1. Diabetic Retinopathy (DR)
The most important ocular complication. Classified as:
A. Non-Proliferative Diabetic Retinopathy (NPDR)
Mild NPDR
- Microaneurysms: earliest clinical sign — small, round, red dots; saccular outpouchings of retinal capillaries at areas of pericyte loss
Moderate NPDR
- Microaneurysms + dot-blot hemorrhages
- Hard exudates: yellowish-white, waxy deposits with sharp margins; composed of lipoproteins and lipid-laden macrophages; arranged in a circinate pattern around leaking vessels
- Cotton wool spots (soft exudates): fluffy white patches due to nerve fiber layer infarctions from capillary occlusion
Severe NPDR (4-2-1 rule / ETDRS)
Any ONE of:
- 4 quadrants of intraretinal hemorrhages (diffuse)
- 2 quadrants of venous beading
- 1 quadrant of IRMA (Intraretinal Microvascular Abnormalities)
(Khurana: 4-2-1 rule predicts ~50% risk of progression to PDR within 1 year)
B. Proliferative Diabetic Retinopathy (PDR)
Characterized by neovascularization — new vessel formation on the disc (NVD) or elsewhere on the retina (NVE) driven by retinal ischemia and VEGF upregulation.
Complications of new vessels:
- Vitreous hemorrhage — sudden painless visual loss
- Tractional retinal detachment — fibrovascular membranes contract and pull the retina
- Neovascular glaucoma — NVI progresses to angle neovascularization, blocking aqueous outflow
High-Risk PDR characteristics (Kanski):
- NVD ≥ 1/4–1/3 disc area
- Any NVD with vitreous/preretinal hemorrhage
- NVE ≥ 1/2 disc area + vitreous/preretinal hemorrhage
2. Diabetic Macular Edema (DME)
The leading cause of visual impairment in diabetics, occurring at any stage of DR.
Types (Khurana / ETDRS):
| Type | Description |
|---|---|
| Focal | Leakage from discrete microaneurysms or vascular abnormalities |
| Diffuse | Generalized leakage from dilated capillary bed |
| Ischemic | Capillary dropout in foveal avascular zone (FAZ) — poor prognosis |
Clinically Significant Macular Edema (CSME) — ETDRS criteria:
- Retinal thickening within 500 µm of foveal center
- Hard exudates within 500 µm of foveal center with adjacent retinal thickening
- Retinal thickening ≥ 1 disc area, any part within 1 disc diameter of fovea
Fundus Photograph — Non-Proliferative Diabetic Retinopathy
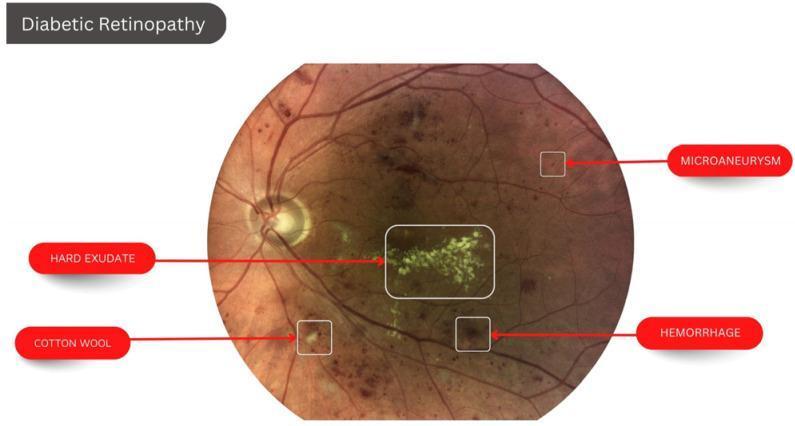
Fundus photograph showing classic NPDR features: microaneurysms (small red dots), dot-blot hemorrhages, hard exudates (yellowish-white clustered deposits near macula), and a cotton wool spot (fluffy pale lesion).
3. Lens Changes
a. Metabolic (Snowflake / True Diabetic) Cataract
- Seen in young, poorly controlled diabetics (Type 1)
- Bilateral, rapidly progressive
- Subcapsular white snowflake opacities that may progress to complete opacification within days–weeks
- Pathogenesis: Accumulation of sorbitol via aldose reductase → osmotic swelling of lens fibers → disruption of Na⁺/K⁺ ATPase pump
b. Senile Cataract — Earlier Onset
- Posterior subcapsular cataract (PSC) more common in diabetics
- Develops 10–15 years earlier than in non-diabetics (Zia & Datta)
c. Refractive Changes (Transient)
- Myopic shift in hyperglycemia (lens swells due to sorbitol accumulation → increased refractive index)
- Hypermetropic shift after insulin treatment (rapid normalization → lens dehydrates)
- Clinically important: avoid prescribing glasses until blood sugar is stable
4. Iris Manifestations
Rubeosis Iridis (Neovascularization of the Iris / NVI)
- New vessels grow from pupillary margin → angle → form fibrovascular membrane
- Leads to neovascular (hemorrhagic) glaucoma — characterized by markedly elevated IOP, severe pain, corneal edema, and poor visual prognosis
- Occurs in ~5% of diabetics; more common in proliferative retinopathy
Other Iris Changes
- Glycogen deposition in iris pigment epithelium → vacuolation (Khurana)
- Dilated iris capillaries — seen on fluorescein angiography
- Poor pupillary dilation — autonomic neuropathy reduces mydriasis
5. Glaucoma
| Type | Mechanism |
|---|---|
| Neovascular glaucoma | Rubeosis iridis → angle closure by fibrovascular membrane |
| Primary open-angle glaucoma (POAG) | 2× higher risk in diabetics; impaired trabecular outflow |
| Elevated IOP | Hyperglycemia correlates with transiently elevated IOP |
(AAO Eye Care of the Patient with Diabetes Mellitus, p. 107)
6. Corneal Changes
- Reduced corneal sensitivity (neurotrophic keratopathy) due to diabetic neuropathy — poor wound healing
- Recurrent corneal erosions — basement membrane abnormalities impair epithelial adhesion
- Diabetic keratopathy — reduced endothelial cell count, decreased pump function
- Increased susceptibility to infections (fungal, bacterial keratitis)
7. Optic Nerve Manifestations
Diabetic Papillopathy
- Transient, unilateral/bilateral disc swelling
- Young Type 1 diabetics, good visual prognosis
- Mild visual field changes; not related to ischemia
- Differentiates from anterior ischemic optic neuropathy (AION) by mild dysfunction and good recovery
Anterior Ischemic Optic Neuropathy (AION)
- Diabetics have increased incidence of non-arteritic AION
- Sudden, painless visual field loss (altitudinal defect) and disc edema
- (Lee et al., Ophthalmology 2011 — cited in AAO, p. 107)
8. Extraocular Muscle Palsies (Diabetic Cranial Neuropathy)
| Nerve | Clinical Features |
|---|---|
| CN III (Oculomotor) | Painful ptosis + "down and out" eye; pupil sparing (ischemic palsy spares pupillomotor fibers at periphery) |
| CN IV (Trochlear) | Vertical diplopia; head tilt away from affected side |
| CN VI (Abducens) | Horizontal diplopia; most common diabetic CN palsy |
- Caused by vasa nervorum ischemia
- Pupil-sparing CN III palsy is pathognomonic for ischemic (diabetic) etiology and must be distinguished from compressive CN III palsy (pupil involved) (Kanski)
- Spontaneous recovery in 3–6 months with glycemic control
9. Other Ocular Manifestations
- Vitreous changes: Asteroid hyalosis more common in diabetics; vitreous liquefaction; premature posterior vitreous detachment (PVD)
- Lipemia retinalis: Milky-white retinal vessels due to gross hypertriglyceridemia associated with poorly controlled DM
- Autonomic neuropathy: Reduced lacrimation, Adie's tonic pupil-like changes, poor accommodation
- Increased risk of infections: Orbital cellulitis, mucormycosis (especially in ketoacidotic patients)
Pathogenesis Summary (Khurana / Zia & Datta)
Chronic hyperglycemia
↓
Polyol pathway activation (sorbitol ↑) → Osmotic damage
Advanced Glycation End Products (AGEs) → Basement membrane thickening
PKC activation → Increased vascular permeability, VEGF upregulation
Oxidative stress → Pericyte loss → Capillary weakness
↓
Microaneurysms → Hemorrhages → Exudates → Ischemia
↓
VEGF ↑ → Neovascularization → PDR / NVG
Screening Guidelines
| Diabetes Type | First Screen | Follow-up |
|---|---|---|
| Type 1 DM | 5 years after diagnosis (after puberty) | Annually |
| Type 2 DM | At time of diagnosis | Annually (more frequent if DR present) |
| Pregnancy in DM | First trimester | Every trimester |
(Khurana: Screening by dilated fundus examination ± retinal photography)
Management Summary
| Condition | Treatment |
|---|---|
| Mild–moderate NPDR | Observation, glycemic + BP + lipid control |
| Severe NPDR / PDR | Panretinal photocoagulation (PRP) |
| DME (centre-involving) | Anti-VEGF injections (bevacizumab, ranibizumab, aflibercept) — first-line |
| DME (non-centre-involving) | Focal/grid laser photocoagulation |
| Vitreous hemorrhage / TRD | Pars plana vitrectomy (PPV) |
| Neovascular glaucoma | Anti-VEGF + PRP + IOP-lowering surgery |
| Diabetic cataract | Phacoemulsification with IOL implantation |
| CN palsies | Observation, patching, glycemic control; recover in 3–6 months |
Key Points to Remember
- Microaneurysm = earliest clinical sign of DR
- Hard exudates = lipid deposits, circinate around leaking vessels
- Cotton wool spots = NFL infarcts (not true exudates)
- 4-2-1 rule = defines severe NPDR with ~50% risk of PDR within 1 year
- Pupil-sparing CN III = diabetic (ischemic) palsy until proven otherwise
- Snowflake cataract = pathognomonic for young diabetics (Type 1)
- Rubeosis iridis → neovascular glaucoma = most feared anterior segment complication
- Anti-VEGF is now first-line for center-involving DME (over laser)
- Refractive changes are transient and relate to acute glycemic fluctuations
- Lipemia retinalis = rare but dramatic sign of severe hypertriglyceridemia in DM
This is a shared conversation. Sign in to Orris to start your own chat.