Explain in detail biosynthesis and catabolism of catecholamine
Reading File
Finding Sources
Finding Sources
Finding Sources
Reading File
Reading File
Reading File
Reading File
Reading File
Finding Sources
Loading Image
Finding Sources
Reading File
Reading File
Finding Sources
Reading File
Reading File
Searching Images
catecholamine catabolism MAO COMT vanillylmandelic acid pathway
Loading Image
Loading Image
I now have comprehensive information from multiple textbooks plus both key diagrams. Let me compile the complete answer.
Biosynthesis and Catabolism of Catecholamines
Catecholamines — dopamine, norepinephrine (NE), and epinephrine (EPI) — are biogenic amines defined by a catechol ring (benzene with two adjacent hydroxyl groups) and an ethylamine side chain. They function as both hormones (adrenal medulla) and neurotransmitters (CNS and peripheral sympathetic nervous system).
I. BIOSYNTHESIS
Precursor and Sites of Synthesis
The pathway begins with L-tyrosine, a non-essential amino acid available from dietary intake or from hepatic hydroxylation of phenylalanine (by phenylalanine hydroxylase). Synthesis occurs in:
- Adrenal medullary chromaffin cells (primarily EPI)
- Postganglionic sympathetic neurons (primarily NE)
- Specific CNS neurons (dopamine, NE, EPI)
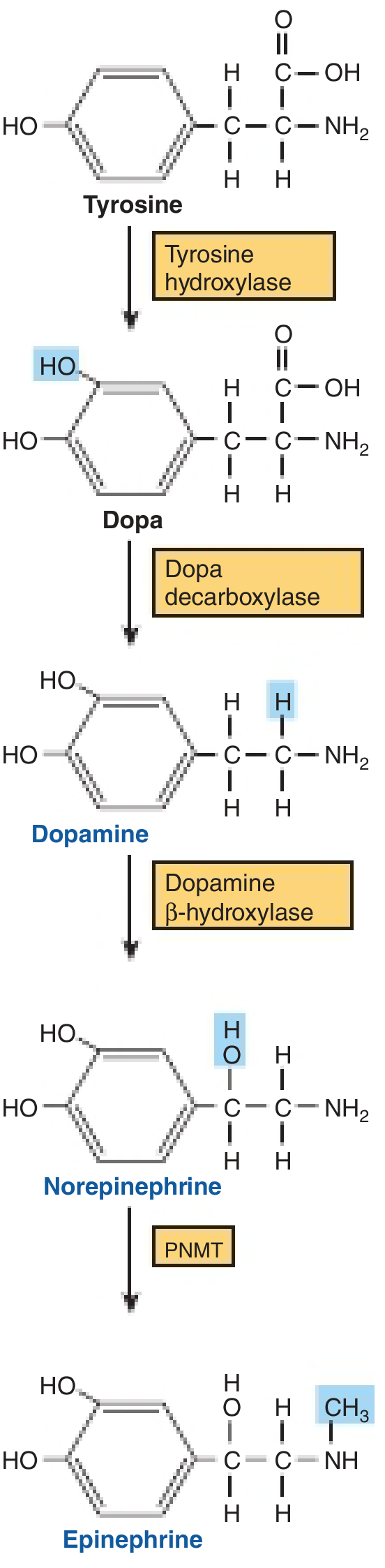
Step-by-Step Pathway
Step 1: Tyrosine → L-DOPA
Enzyme: Tyrosine hydroxylase (TH)
Location: Cytosol
Reaction: Hydroxylation of the tyrosine ring at the 3-position
Cofactor: Tetrahydrobiopterin (BH₄) — a pteridine cofactor that acts as electron donor
This is the rate-limiting step of the entire pathway. TH is found only in catecholamine-synthesizing tissues. It functions as a mixed-function oxidase (oxidoreductase).
Regulation of TH:
- End-product feedback inhibition: Free cytosolic catecholamines compete with TH for the BH₄ cofactor — this is the primary short-term regulatory mechanism
- Short-term activation: Depolarization activates protein kinases (PKA, PKC, CaM kinases) that phosphorylate TH, increasing its affinity for BH₄ and reducing sensitivity to end-product inhibition
- Long-term upregulation: Sustained sympathetic activity increases TH and dopamine-β-hydroxylase (DBH) mRNA via CREB phosphorylation and binding to CRE in the gene promoter, driving increased enzyme synthesis transported down the axon
- Competitive inhibition: α-Methyltyrosine blocks TH
Step 2: L-DOPA → Dopamine
Enzyme: DOPA decarboxylase (Aromatic amino acid decarboxylase, AADC)
Location: Cytosol
Reaction: Decarboxylation — removal of the carboxyl group
Cofactor: Pyridoxal phosphate (PLP, vitamin B₆)
This enzyme is ubiquitous (present in all tissues). Dopaminergic neurons stop here — they lack the enzymes for subsequent steps. α-Methyldopa competitively inhibits this reaction (used in some forms of hypertension).
Step 3: Dopamine → Norepinephrine
Enzyme: Dopamine β-hydroxylase (DBH)
Location: Inside secretory/storage vesicles (particulate fraction)
Reaction: β-Hydroxylation of the ethylamine side chain — a mixed-function oxygenase reaction
Cofactors: Ascorbate (vitamin C) as electron donor, Cu²⁺ as bound cofactor required for electron transfer, fumarate as modulator
Because DBH is located exclusively within storage vesicles, dopamine must first be transported from the cytosol into the vesicles (via VMAT2) before this step occurs. Norepinephrine neurons stop here.
Step 4: Norepinephrine → Epinephrine
Enzyme: Phenylethanolamine-N-methyltransferase (PNMT)
Location: Cytoplasm (soluble enzyme)
Reaction: N-methylation — transfer of a methyl group from S-adenosylmethionine (SAM) to the amine group of norepinephrine
Cofactor: SAM (indirectly dependent on folate and vitamin B₁₂ for SAM regeneration)
Key points about PNMT:
- Present in chromaffin cells of the adrenal medulla, organ of Zuckerkandl, and a subset of CNS neurons only
- Synthesis of PNMT is induced by glucocorticoids that reach the medulla via the intra-adrenal portal system, which provides a ~100-fold steroid concentration gradient over systemic blood
- Since PNMT is cytoplasmic, NE must exit its vesicles (via spontaneous leakage or post-synaptic reuptake) into the cytoplasm, be methylated to EPI, and then be transported back into vesicles for storage
Summary Table: Biosynthetic Enzymes
| Step | Substrate | Product | Enzyme | Cofactor | Location |
|---|---|---|---|---|---|
| 1 | Tyrosine | L-DOPA | Tyrosine hydroxylase (TH) | BH₄, O₂ | Cytosol |
| 2 | L-DOPA | Dopamine | AADC (Dopa decarboxylase) | PLP (B₆) | Cytosol |
| 3 | Dopamine | Norepinephrine | Dopamine β-hydroxylase (DBH) | Ascorbate, Cu²⁺ | Storage vesicle |
| 4 | Norepinephrine | Epinephrine | PNMT | SAM | Cytoplasm (adrenal medulla) |
Storage and Release
- After synthesis, dopamine is transported from the cytosol into vesicles via VMAT2 (vesicular monoamine transporter 2) — an ATP-dependent secondary active transport driven by a proton (H⁺) gradient generated by vesicular ATPase (V-ATPase)
- Intravesicular catecholamine concentration is ~0.5 M (approximately 100× cytosolic), stored complexed with ATP and chromogranins
- An action potential triggers Ca²⁺ influx → vesicle fusion with the plasma membrane → exocytosis of catecholamines, ATP, chromogranins, and DBH into the extraneuronal space
- VMAT-1 is expressed mainly in extraneuronal chromaffin cells; VMAT-2 is the neuronal form
II. CATABOLISM (Inactivation and Degradation)
Catecholamines have a plasma half-life of ~1 minute and are potent, short-acting compounds. Termination of action occurs through three mechanisms:
A. Reuptake (Primary mechanism for endogenous catecholamines)
- Uptake-1 (neuronal): Active, stereospecific, energy-requiring reuptake into the presynaptic terminal via NET (norepinephrine transporter) or DAT (dopamine transporter) — the predominant pathway for NE and dopamine at the synapse
- Blocked by tricyclic antidepressants and cocaine → elevated synaptic catecholamines
- Uptake-2 (extraneuronal): Minor pathway — NE enters effector cells where it is metabolized by MAO and COMT
- Reuptake is less relevant for exogenous catecholamines (administered pharmacologically), whose inactivation is predominantly hepatic/renal metabolism — explaining why exogenous catecholamines have longer action than endogenous ones
B. Enzymatic Degradation (Two key enzymes)
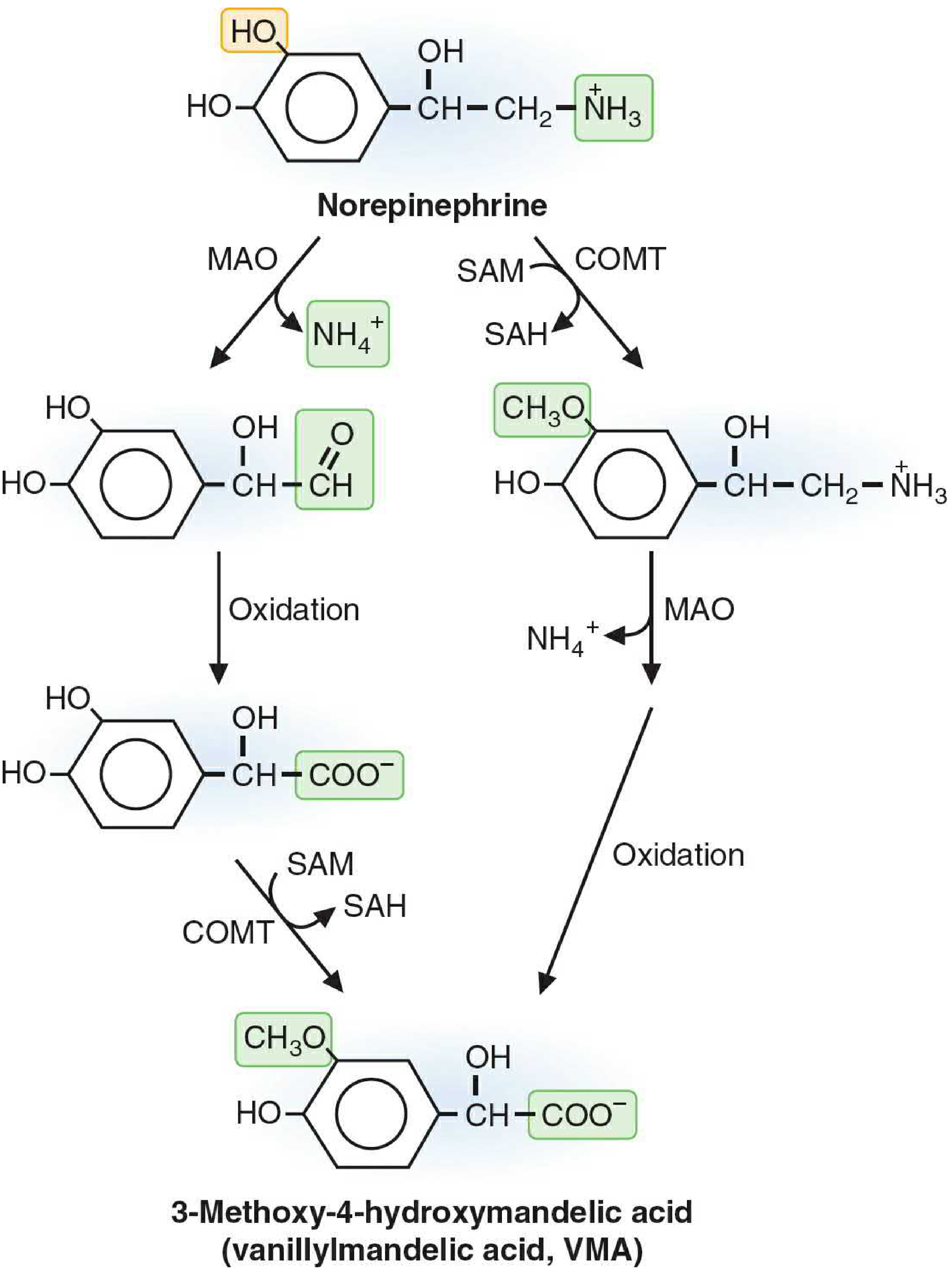
1. Monoamine Oxidase (MAO)
- Location: Outer mitochondrial membrane of many cells, including the presynaptic terminal
- Reaction: Oxidative deamination — oxidizes the carbon bearing the amino group to an aldehyde, releasing NH₄⁺
- Two isoforms:
- MAO-A: Preferentially deaminates NE and serotonin → targeted by MAO-A inhibitors (e.g., clorgyline, moclobemide)
- MAO-B: Acts on a wide spectrum of phenylethylamines (including dopamine) → inhibited by selegiline (deprenyl), used in Parkinson's disease
- In the presynaptic terminal, MAO inactivates catecholamines that are not protected within storage vesicles — thus drugs that deplete vesicles (e.g., reserpine) indirectly increase MAO-mediated degradation
2. Catechol-O-Methyltransferase (COMT)
- Location: Cytoplasm of many cells — especially liver, kidney, erythrocytes, glial cells, and extraneuronal tissues; acts on catecholamines that have diffused away from the synapse
- Reaction: O-methylation — transfers a methyl group from SAM to one of the hydroxyl groups on the catechol ring (the 3-OH group), producing methoxyderivatives
- Epinephrine → Metanephrine
- Norepinephrine → Normetanephrine
- Dopamine → 3-Methoxytyramine
- Because this reaction requires SAM, it is indirectly dependent on folate and vitamin B₁₂
C. Diffusion
- A small fraction of catecholamines escapes reuptake and metabolic degradation, diffuses into the circulation, and is metabolized by the liver and kidney (the predominant route for exogenous catecholamines)
Final Degradation Products
The sequential or combined action of MAO and COMT — in either order — generates a variety of intermediates, which ultimately converge on:
| Catecholamine | Major Urinary Metabolite |
|---|---|
| Epinephrine + Norepinephrine | Vanillylmandelic acid (VMA) = 3-methoxy-4-hydroxymandelic acid |
| Dopamine | Homovanillic acid (HVA) |
| Epinephrine | Metanephrine (after COMT alone) |
| Norepinephrine | Normetanephrine (after COMT alone) |
Metabolic routes to VMA:
- MAO first → aldehyde intermediate → oxidation → DHMA → COMT → VMA
- COMT first → normetanephrine/metanephrine → MAO → aldehyde → oxidation → VMA
- Both routes converge on VMA
Metabolites are excreted in the urine, sometimes after sulfonation or glucuronide conjugation in the liver.
Clinical note — Pheochromocytoma: Because metanephrine and normetanephrine are produced by continuous COMT activity within the tumor (even between episodic catecholamine surges), plasma fractionated metanephrines are more sensitive diagnostic markers than catecholamines themselves, which fluctuate widely. 24-hour urinary VMA and metanephrines are also used — Sabiston Textbook of Surgery.
Dopamine-Specific Catabolism
Dopamine is handled slightly differently:
- MAO → DOPAC (3,4-dihydroxyphenylacetic acid)
- COMT → 3-methoxytyramine
- Sequential MAO + COMT → Homovanillic acid (HVA)
HVA in cerebrospinal fluid serves as an indicator of dopamine turnover. Decreased CSF HVA is found in Parkinson's disease.
III. CLINICAL CORRELATES
| Pathway Component | Clinical Relevance |
|---|---|
| TH (rate-limiting step) | α-Methyltyrosine (metyrosine) blocks catecholamine synthesis — used pre-operatively in pheochromocytoma |
| AADC | L-DOPA (+ carbidopa, peripheral AADC inhibitor) for Parkinson's disease |
| DBH — requires ascorbate & Cu²⁺ | Deficiency causes severe orthostatic hypotension |
| PNMT — induced by glucocorticoids | Loss of adrenal cortex → reduced EPI synthesis |
| Reuptake (NET) | Tricyclic antidepressants, cocaine block NET → elevated NE |
| MAO | MAO inhibitors (MAOIs): antidepressants; selegiline in Parkinson's; "cheese effect" with tyramine |
| COMT | Entacapone/tolcapone (COMT inhibitors) used in Parkinson's to prolong L-DOPA effect |
| Metanephrines | Screening test for pheochromocytoma |
| VMA | Urine VMA elevated in neuroblastoma and pheochromocytoma |
Sources: Harper's Illustrated Biochemistry 32nd Ed (pp. 510–511) — Basic Medical Biochemistry: A Clinical Approach 6th Ed (Ch. 46) — Sabiston Textbook of Surgery (Ch. 75) — Barash's Clinical Anesthesia 9th Ed (Ch. 14) — Kaplan & Sadock's Comprehensive Textbook of Psychiatry (Ch. 1.7)
Explain in detail biosynthesis and catabolism of catecholamine for 10 marks
Based on the comprehensive research already completed in our previous response, here is a structured 10-mark exam answer:
Biosynthesis and Catabolism of Catecholamines
Introduction
Catecholamines — dopamine, norepinephrine (NE), and epinephrine (EPI) — are derived from the amino acid tyrosine. They act as neurotransmitters in the CNS/peripheral nervous system and as hormones from the adrenal medulla.
BIOSYNTHESIS (5 marks)
Precursor
L-Tyrosine — obtained from diet or synthesized in the liver from phenylalanine by phenylalanine hydroxylase.
Sites
- Adrenal medullary chromaffin cells (mainly EPI)
- Postganglionic sympathetic neurons (mainly NE)
- Specific CNS neurons (dopamine, NE, EPI)
Pathway (4 steps)

Step 1: Tyrosine → L-DOPA
- Enzyme: Tyrosine hydroxylase (TH)
- Cofactor: Tetrahydrobiopterin (BH₄)
- Location: Cytosol
- This is the rate-limiting step
- TH is regulated by:
- Feedback inhibition — free catecholamines compete for the BH₄ cofactor
- Phosphorylation by PKA, PKC, CaM kinases upon depolarization → increased BH₄ affinity
Step 2: L-DOPA → Dopamine
- Enzyme: DOPA decarboxylase / AADC (aromatic amino acid decarboxylase)
- Cofactor: Pyridoxal phosphate (PLP, vitamin B₆)
- Location: Cytosol
- Dopaminergic neurons stop here
Step 3: Dopamine → Norepinephrine
- Enzyme: Dopamine β-hydroxylase (DBH)
- Cofactors: Ascorbate (vitamin C), Cu²⁺
- Location: Inside storage vesicles
- Noradrenergic neurons stop here
Step 4: Norepinephrine → Epinephrine
- Enzyme: Phenylethanolamine-N-methyltransferase (PNMT)
- Cofactor: SAM (S-adenosylmethionine)
- Location: Cytoplasm of adrenal medullary cells
- PNMT is induced by glucocorticoids via the intra-adrenal portal system
- Occurs only in adrenal medulla, organ of Zuckerkandl, and select CNS neurons
Summary Table
| Step | Substrate | Product | Enzyme | Cofactor |
|---|---|---|---|---|
| 1 (Rate-limiting) | Tyrosine | L-DOPA | TH | BH₄ |
| 2 | L-DOPA | Dopamine | AADC | PLP (B₆) |
| 3 | Dopamine | Norepinephrine | DBH | Ascorbate, Cu²⁺ |
| 4 | Norepinephrine | Epinephrine | PNMT | SAM |
CATABOLISM (5 marks)
Catecholamines have a plasma half-life of ~1 minute. Inactivation occurs by three mechanisms:
1. Reuptake (Primary)
- Catecholamines are actively transported back into the presynaptic terminal (Uptake-1) via NET (norepinephrine transporter) or DAT (dopamine transporter)
- Blocked by tricyclic antidepressants and cocaine
- Minor extraneuronal uptake (Uptake-2) occurs into effector cells
2. Enzymatic Degradation — MAO and COMT

Monoamine oxidase (MAO)
- Location: Outer mitochondrial membrane
- Reaction: Oxidative deamination → converts the amine to an aldehyde + NH₄⁺
- Two isoforms:
- MAO-A — preferentially deaminates NE and serotonin
- MAO-B — acts on dopamine and phenylethylamines
- Inactivates unprotected (extra-vesicular) cytosolic catecholamines
Catechol-O-methyltransferase (COMT)
- Location: Cytoplasm of liver, kidney, erythrocytes, glia, extraneuronal cells
- Reaction: O-methylation using SAM — transfers a methyl group to the catechol ring
- NE → Normetanephrine; EPI → Metanephrine; Dopamine → 3-Methoxytyramine
3. Diffusion
- Small amounts escape into circulation and are metabolized by liver/kidney
- This is the primary route for exogenous catecholamines (explaining their longer pharmacological duration)
Final Urinary Metabolites
MAO and COMT act in either order; both pathways converge on the same final products:
| Catecholamine | Final Metabolite |
|---|---|
| Epinephrine + Norepinephrine | Vanillylmandelic acid (VMA) = 3-methoxy-4-hydroxymandelic acid |
| Dopamine | Homovanillic acid (HVA) |
| EPI (COMT alone) | Metanephrine |
| NE (COMT alone) | Normetanephrine |
Metabolites are excreted in urine, sometimes after sulfonation or glucuronide conjugation in the liver.
CLINICAL APPLICATIONS
| Enzyme/Step | Clinical Application |
|---|---|
| TH inhibition (α-methyltyrosine) | Pre-op preparation in pheochromocytoma |
| AADC inhibition (carbidopa) | Used with L-DOPA in Parkinson's disease |
| PNMT induction by glucocorticoids | Explains reduced EPI in adrenal insufficiency |
| MAO inhibitors | Antidepressants; selegiline for Parkinson's; tyramine "cheese effect" |
| COMT inhibitors (entacapone) | Prolong L-DOPA action in Parkinson's disease |
| Elevated metanephrines / VMA | Diagnosis of pheochromocytoma / neuroblastoma |
Sources: Harper's Illustrated Biochemistry 32nd Ed; Basic Medical Biochemistry: A Clinical Approach 6th Ed; Sabiston Textbook of Surgery; Barash's Clinical Anesthesia 9th Ed
In short
Biosynthesis and Catabolism of Catecholamines
BIOSYNTHESIS
Starts from L-Tyrosine in a 4-step pathway:

| Step | Reaction | Enzyme | Cofactor |
|---|---|---|---|
| 1 ⭐ | Tyrosine → L-DOPA | Tyrosine hydroxylase (TH) — rate-limiting | BH₄ |
| 2 | L-DOPA → Dopamine | DOPA decarboxylase (AADC) | PLP (B₆) |
| 3 | Dopamine → Norepinephrine | Dopamine β-hydroxylase (DBH) | Ascorbate, Cu²⁺ |
| 4 | Norepinephrine → Epinephrine | PNMT | SAM |
Key: TH is feedback-inhibited by catecholamines. PNMT is induced by glucocorticoids (adrenal medulla only).
CATABOLISM
Two key enzymes act in any order:

- MAO (mitochondria) → oxidative deamination → removes NH₄⁺
- COMT (cytoplasm) → O-methylation using SAM
Final Products
| Catecholamine | End Metabolite |
|---|---|
| NE + EPI | VMA (vanillylmandelic acid) — urine marker |
| Dopamine | HVA (homovanillic acid) |
| NE/EPI (COMT only) | Normetanephrine / Metanephrine |
3 Mechanisms of Termination
- Reuptake into presynaptic terminal (main route for endogenous)
- MAO + COMT degradation
- Diffusion → hepatic/renal metabolism (main route for exogenous)
Clinical: ↑ Urine VMA + metanephrines → Pheochromocytoma; MAO inhibitors → antidepressants; COMT inhibitors (entacapone) → Parkinson's
This is a shared conversation. Sign in to Orris to start your own chat.