Stool microscopy it doesn’t looks like parasite
stool microscopy non-parasitic findings artifacts plant cells food debris
stool microscopy artifacts plant cells pollen food particles
Stool Microscopy — Image Analysis
What You're Seeing
- Plant fiber/cellulose fragments from undigested vegetable or grain matter
- Pollen grains (some have the characteristic irregular, lobulated outline)
- Possibly starch granules partially digested
Key Distinguishing Features (Why These Are NOT Parasites)
| Feature | What's Seen | Parasite Expected |
|---|---|---|
| Shape | Irregular, angular, plant-cell-like | Rounded/oval with defined wall |
| Internal structure | No nucleus, no organelles visible | Nuclei, cysts have internal features |
| Arrangement | Random scatter | Often clustered or motile |
| Color | Bright orange-red (likely iodine-stained or autofluorescent) | Brown/gold with distinct wall |
| Size variation | Highly variable | More uniform per species |
Common Non-Parasitic Findings in Stool Microscopy
- Plant cells / vegetable fibers — very common, often mistaken for parasite fragments
- Pollen — seasonal, rounded/spiky appearance
- Muscle fibers (from meat) — cylindrical with striations
- Fat globules — round, refractile
- Starch granules — oval, stain blue-black with iodine
- Yeast cells — small, budding oval forms (Candida spp. — not always pathogenic)
- Epithelial cells — large, flat polygonal cells
Recommendation
- Repeat stool exam × 3 samples on different days
- Specific antigen tests (e.g., Giardia/Cryptosporidium EIA)
- PCR-based stool pathogen panels for higher sensitivity
How to make 0.8 % suspension ??
Preparing a 0.8% Fecal Suspension for Stool Microscopy
Preparing 0.85% Normal Saline Stock Solution
| Component | Amount |
|---|---|
| NaCl (table/lab salt) | 0.85 g |
| Distilled water | 100 mL |
Making the Stool Suspension
- Place 1–2 drops of 0.85% saline on a clean glass slide
- Pick a small amount of stool (~2 mg, approximately the size of a match head) using an applicator stick
- Emulsify thoroughly into the saline drops — mix well until evenly suspended
- The suspension should be thin enough to read newsprint through — if too thick, add more saline
- Apply a coverslip (22×22 mm)
- Examine under 10× then 40× objective
Formalin-Based Suspension (for concentration)
| Component | Ratio |
|---|---|
| Stool | 1 part (~1 g) |
| 10% formalin or SAF fixative | 9–10 parts (9–10 mL) |
Key Tips
- Always prepare two slides — one with saline (motility/trophozoites), one with Lugol's iodine (cysts/nuclei detail)
- Use fresh stool within 30 minutes for trophozoite detection
- The suspension should look faintly turbid/cloudy, not opaque
0.8% of Pooled RBC how to make ???
Preparing 0.8% Pooled RBC Suspension
What You Need
| Item | Purpose |
|---|---|
| Blood samples (2–3 donors or pooled) | Source of RBCs |
| 0.85% Normal Saline (physiological saline) | Washing & suspension medium |
| Centrifuge | Cell separation |
| Test tubes, pipettes | Labware |
Step-by-Step Protocol
Step 1 — Collect & Pool Blood
- Collect blood from 2–3 donors into EDTA tubes (anticoagulated)
- Pool equal volumes together in a clean tube
Step 2 — Wash the RBCs (×3)
- Add 5–10 mL saline to pooled blood
- Centrifuge at 1000–1500 rpm for 5 minutes
- Discard the supernatant + buffy coat (white cell layer) carefully
- Repeat washing 3 times until supernatant is clear (no hemolysis)
Step 3 — Make the 0.8% Suspension
| Component | Volume |
|---|---|
| Packed RBC pellet | 0.8 mL |
| 0.85% Normal Saline | 99.2 mL |
| Total | 100 mL |
Simply put: 0.8 mL packed RBCs + 99.2 mL saline = 0.8% RBC suspension
| Packed RBCs | Saline |
|---|---|
| 0.08 mL (80 µL) | 9.92 mL |
Quick Formula
Quality Check
- The suspension should appear uniformly turbid/pink
- No clumping or hemolysis (clear supernatant after settling)
- Use fresh preparation — ideally same day or store at 2–8°C for up to 1 week
Common Uses of 0.8% RBC Suspension
| Test | Purpose |
|---|---|
| Widal test | Salmonella antibody detection |
| TPHA | Treponema pallidum hemagglutination |
| IHA (Indirect Hemagglutination) | Parasitic/bacterial serology |
| ABO/Rh blood grouping | Crossmatching |
| Coombs test | Hemolytic anemia workup |
0.8 mL in microlitre and 99.2 mL too
Unit Conversions
Simple Rule
0.8 mL → Microlitres
99.2 mL → Microlitres
Summary Table
| Component | mL | µL |
|---|---|---|
| Packed RBCs | 0.8 mL | 800 µL |
| 0.85% Saline | 99.2 mL | 99,200 µL |
| Total | 100 mL | 100,000 µL |
For Smaller Volumes (Practical Bench Use)
| Component | mL | µL |
|---|---|---|
| Packed RBCs | 0.08 mL | 80 µL |
| 0.85% Saline | 9.92 mL | 9,920 µL |
| Total | 10 mL | 10,000 µL |
For bench work, use a micropipette for the RBC volume (80 µL) and a measuring cylinder for the saline.
It is much ??
More Practical Small-Scale Volumes
| Total Volume | Packed RBCs | Saline |
|---|---|---|
| 100 mL | 800 µL | 99.2 mL |
| 10 mL | 80 µL | 9.92 mL |
| 5 mL | 40 µL | 4.96 mL |
| 1 mL | 8 µL | 992 µL |
Most Common Bench Practice
5–10 mL total — enough for a full day's batch of tests
| Component | Amount |
|---|---|
| Packed RBCs | 80 µL |
| 0.85% Saline | 9.92 mL (~10 mL) |
| Total | 10 mL |
- Use a micropipette for the 80 µL RBCs
- Use a measuring cylinder or graduated tube for saline
- Gives enough suspension for ~100–200 test drops depending on the assay
Tip: Only prepare what you need for the day — fresh suspension gives better, more reliable results.
Can a gravida three with two prior live children with RH positive blood group . Mother is Rh negative. Can her indirect Coombs test can come negative ??
indirect Coombs test negative Rh negative mother Rh positive fetus alloimmunization
Rh isoimmunization anti-D antibody formation prevention RhoGAM
Can ICT Be Negative in This Scenario?
Short Answer
Understanding the Scenario
| Factor | Detail |
|---|---|
| Gravida | 3 |
| Para | 2 (both live, both Rh positive) |
| Mother | Rh negative |
| Current fetus | Rh positive |
| ICT result | Negative |
Reasons ICT Can Be Negative
1. Anti-D Prophylaxis (RhoGAM) Given Properly
- If the mother received anti-D immunoglobulin (RhoGAM):
- At 28 weeks in previous pregnancies
- After each delivery of Rh+ baby
- After any sensitizing events (miscarriage, amniocentesis, trauma)
- The administered anti-D is passively acquired and clears within 12 weeks
- No active alloimmunization occurs → ICT stays negative
- This is the most common reason for a negative ICT
2. Insufficient Fetomaternal Hemorrhage (FMH)
- In previous pregnancies, the volume of fetal blood entering maternal circulation may have been too small to trigger an immune response
- Sensitization requires a threshold volume of Rh+ RBCs
- Small FMH → no antibody formation → ICT negative
3. Immune Tolerance
- Some Rh-negative women simply fail to mount an antibody response despite exposure
- Approximately 30% of Rh-negative mothers never become sensitized even without prophylaxis
- Genetic factors influence immune responsiveness
4. Early Gestational Age at Testing
- Anti-D antibodies may be below detectable threshold early in pregnancy
- ICT may turn positive later — repeat testing is essential
5. Previous Children Were Actually Rh Negative
- If prior children were Rh negative (depends on father's genotype), no sensitizing exposure occurred
- Father could be heterozygous (Dd) — 50% chance each child is Rh negative
Clinical Implications — What To Do
| Action | Timing |
|---|---|
| Repeat ICT | Every 4 weeks if negative (28–36 weeks) |
| Anti-D prophylaxis | Give at 28 weeks regardless of ICT result |
| Anti-D after delivery | If baby confirmed Rh positive, give within 72 hours |
| Check father's Rh genotype | Helps predict fetal Rh status |
| Kleihauer-Betke test | If large FMH suspected — quantifies fetal cells in maternal blood |
Key Takeaway
A negative ICT does not mean the mother is safe to ignore — it means she has not yet formed antibodies. She remains at risk with every Rh+ pregnancy. Prophylaxis must continue.
If it came weak positive then ??
weak positive indirect Coombs test Rh negative mother management titration
anti-D antibody titer critical level fetal monitoring hemolytic disease newborn
Weak Positive ICT — What It Means & What To Do
First — Distinguish the Cause
| Cause | Interpretation |
|---|---|
| Recent RhoGAM injection (within 12 weeks) | Passive anti-D — not dangerous, expected finding |
| True alloimmunization (active immune response) | Dangerous — requires close monitoring |
This distinction is the most critical first step
If Passive (Post-RhoGAM)
- Weak positive is expected and reassuring
- Titer usually ≤1:4
- Will disappear within 12 weeks of injection
- No fetal risk
- Continue routine antenatal care
If True Alloimmunization (Active Anti-D)
Step 1 — Perform Antibody Titration Immediately
| Titer Level | Interpretation | Action |
|---|---|---|
| ≤ 1:4 | Weak, low risk | Repeat every 4 weeks |
| 1:8 – 1:16 | Borderline | Close monitoring, repeat every 2 weeks |
| ≥ 1:16 – 1:32 | Critical level | Refer to fetal medicine specialist urgently |
| > 1:32 | High risk | Fetal anemia likely — invasive monitoring |
Critical titer = 1:16 (most centers) — threshold above which fetal hydrops risk rises significantly
Step 2 — Fetal Surveillance
| Investigation | Purpose |
|---|---|
| Middle Cerebral Artery Doppler (MCA-PSV) | Non-invasive assessment of fetal anemia — first line |
| MCA-PSV > 1.5 MoM | Indicates moderate-severe fetal anemia |
| Fetal cordocentesis | Confirms fetal Hb directly — done if MCA abnormal |
| Intrauterine transfusion (IUT) | Treatment if fetal Hb critically low |
| Obstetric ultrasound | Look for hydrops (ascites, pleural effusion, skin edema) |
Step 3 — Serial Antibody Monitoring Schedule
- Every 4 weeks up to 28 weeks gestation
- Every 2 weeks from 28 weeks until delivery
- Refer to fetal medicine if titer reaches critical level or is rising significantly
Signs of Fetal Hemolytic Disease to Watch For on USS
- Fetal hydrops (skin edema, ascites, pleural/pericardial effusion)
- Placentomegaly
- Polyhydramnios
- Enlarged fetal liver/spleen (hepatosplenomegaly)
Delivery Planning
| Situation | Plan |
|---|---|
| Titer low, stable, no fetal compromise | Deliver at 37–38 weeks |
| Rising titer, MCA-PSV borderline | Deliver at 34–36 weeks with steroid cover |
| Fetal hydrops / severe anemia | Immediate IUT + deliver as soon as viable |
After Delivery
- Do NOT give RhoGAM if mother is truly alloimmunized — it has no benefit once sensitization has occurred
- Newborn needs:
- Direct Coombs Test (DCT)
- Cord blood Hb + bilirubin
- Monitor for neonatal jaundice / hemolytic disease of the newborn (HDN)
- Possible phototherapy or exchange transfusion
Key Takeaway
A weak positive ICT in a G3P2 Rh-negative mother with Rh-positive children means early alloimmunization is likely underway. The priority is to titrate immediately, distinguish passive from active antibody, and begin serial fetal surveillance before the titer climbs to dangerous levels.
In term of gel card grading
Gel Card (Column Agglutination) Grading for ICT
Standard Gel Card Grading Scale
| Grade | Appearance | Interpretation |
|---|---|---|
| 4+ | Single solid red band at top of gel column | Strong positive |
| 3+ | Large agglutinates in upper half of column | Moderate-strong positive |
| 2+ | Agglutinates throughout column | Moderate positive |
| 1+ | Small agglutinates in lower half, some cells at bottom | Weak positive |
| W+ (weak+) | Faint agglutinates just above cell button, small pellet | Very weak positive |
| Negative | Complete pellet at bottom, gel clear | Negative |
Visual Representation
| ==== | ← 4+ (solid band at top)
| ---- |
| |
| ---- | ← 3+ (large clumps upper half)
| ---- |
| |
| ---- | ← 2+ (agglutinates throughout)
| ---- |
| ---- |
| | ← 1+ (small clumps lower half)
| ---- |
| == | ← small pellet
| | ← W+ (faint, just above pellet)
| .|
| === | ← near-complete pellet
| | ← Negative
| |
| ==== | ← complete pellet at bottom
In Context of Your ICT Scenario
| Gel Grade | Titer Equivalent (approx.) | Clinical Significance |
|---|---|---|
| W+ | ≤ 1:2 – 1:4 | Could be passive RhoGAM — repeat & confirm |
| 1+ | ~ 1:4 – 1:8 | Early alloimmunization — monitor closely |
| 2+ | ~ 1:16 – 1:32 | Approaching critical level — refer fetal medicine |
| 3+ | ~ 1:32 – 1:64 | Above critical — urgent fetal surveillance |
| 4+ | ≥ 1:64 | Severe sensitization — high fetal risk |
Important Notes for Gel Card Reading
- Always read in good lighting against a white background
- Read within 10 minutes after centrifugation
- W+ must never be ignored in an Rh-negative pregnant mother
- A result of 1+ and above = report as positive and refer for titration
- Hemolysis in the gel = also a positive result (complement activation)
What To Do Based on Gel Grade in Your Patient
| Gel Result | Immediate Action |
|---|---|
| W+ | Check RhoGAM history — if no recent injection, treat as true positive, do titration |
| 1+ | Titrate immediately — serial monitoring every 4 weeks |
| 2+ or above | Urgent referral to fetal medicine + MCA Doppler |
Bottom line: Even a W+ on gel card in a G3 Rh-negative mother with Rh+ children should never be dismissed — it warrants full investigation and titration.
Emperipolesis in peripheral smear
emperipolesis peripheral blood smear lymphocytes neutrophils
emperipolesis peripheral smear microscopy
Emperipolesis in Peripheral Smear
Definition
Key distinction from phagocytosis (erythrophagocytosis):
| Feature | Emperipolesis | Phagocytosis |
|---|---|---|
| Engulfed cell | Intact, viable | Degraded, destroyed |
| Engulfed cell membrane | Preserved | Disrupted |
| Process | Passive — cell moves in voluntarily | Active — cell is ingested & killed |
| Surrounding vacuole | Present (cells sit in vacuole) | Phagolysosome forms |
What It Looks Like on Peripheral Smear
- Large histiocyte/monocyte with abundant pale cytoplasm
- Intact lymphocytes, neutrophils or RBCs visible inside the cytoplasm
- Engulfed cells appear normal morphologically — not fragmented
- Engulfed cells sit in a clear halo/vacuole within the host cell
Image — Emperipolesis (Rosai-Dorfman Disease)
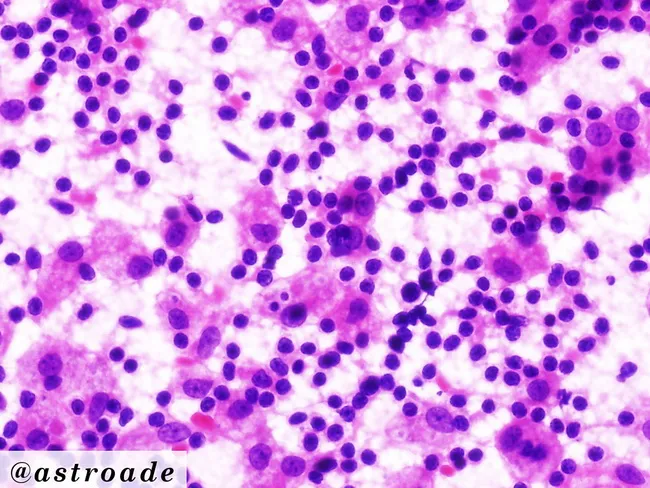
Causes / Associated Conditions
| Condition | Notes |
|---|---|
| Rosai-Dorfman Disease (RDD) | Classic cause — sinus histiocytosis with massive lymphadenopathy |
| Hemophagocytic Lymphohistiocytosis (HLH) | Emperipolesis + erythrophagocytosis both seen |
| Myeloperoxidase deficiency | Neutrophils inside monocytes |
| Chronic myeloid leukemia (CML) | Occasionally reported |
| Autoimmune conditions | SLE, rheumatoid arthritis |
| Viral infections | EBV, CMV triggered reactive histiocytosis |
| Post bone marrow transplant | Reactive phenomenon |
Rosai-Dorfman Disease — Key Features
| Feature | Detail |
|---|---|
| Presentation | Massive painless cervical lymphadenopathy |
| Systemic symptoms | Fever, night sweats, elevated ESR |
| Blood picture | Leukocytosis, elevated ESR, hypergammaglobulinemia |
| Immunophenotype | S-100 +, CD68 +, CD1a − |
| Prognosis | Usually self-limiting |
Emperipolesis vs HLH on Smear
| Feature | Emperipolesis (RDD) | HLH |
|---|---|---|
| Cell engulfed | Lymphocytes mainly | RBCs, WBCs, platelets |
| Host cell | Histiocyte | Activated macrophage |
| Engulfed cell state | Intact | Degraded |
| Clinical | Lymphadenopathy | Fever, cytopenias, hyperferritinemia |
| Ferritin | Normal/mildly raised | Markedly elevated (>500, often >10,000) |
Reporting on Peripheral Smear
"Large histiocytes noted with intact lymphocytes/neutrophils within their cytoplasm, consistent with emperipolesis. Findings may suggest Rosai-Dorfman disease or reactive histiocytosis. Correlation with clinical findings, lymph node biopsy and immunohistochemistry (S-100, CD68, CD1a) recommended."
Emperipolesis in peripheral smear
emperipolesis peripheral blood smear neutrophil inside monocyte
emperipolesis peripheral blood smear clinical significance causes
Emperipolesis on Peripheral Blood Smear — Specific Context
What You Actually See on PBF
- A monocyte or large mononuclear cell with
- Intact neutrophil(s), lymphocyte(s), or RBCs sitting inside its cytoplasm
- The engulfed cell has a clear halo around it
- The engulfed cell looks morphologically normal — not digested
Most Common Cells Involved on PBF
| Host Cell | Engulfed Cell | Associated Condition |
|---|---|---|
| Monocyte | Neutrophil | Systemic inflammation, sepsis |
| Monocyte | Lymphocyte | Viral infections, RDD |
| Large histiocyte | RBCs + WBCs + platelets | HLH |
| Monocyte | Platelets | ITP, consumptive coagulopathy |
Grading on PBF
| Finding | Significance |
|---|---|
| Occasional (1–2 per 100 WBCs) | May be incidental / artifact |
| Frequent (>5 per 100 WBCs) | Pathological — investigate further |
| Prominent + pancytopenia | Strongly suggests HLH |
Key Conditions to Consider by PBF Pattern
1. Emperipolesis + Normal CBC
- Likely reactive — viral infection, post-transfusion
- May be early Rosai-Dorfman
2. Emperipolesis + Pancytopenia + High Ferritin
- HLH until proven otherwise
- Check: Ferritin, triglycerides, fibrinogen, NK cell activity, bone marrow biopsy
3. Emperipolesis + Lymphocytosis + Lymphadenopathy
- Rosai-Dorfman Disease
- Biopsy + S100/CD68/CD1a staining needed
4. Emperipolesis + Blast Cells
- Seen in AML M4/M5 (monocytic leukemia)
- Monocytic blasts can engulf other cells
Differentials — What Can Mimic It on Smear
| Mimic | How to Distinguish |
|---|---|
| Phagocytosis | Engulfed cell is degraded/fragmented |
| Overlapping cells | Change focal plane — overlapping cells separate at different focus levels |
| Pelger-Huët anomaly | Nuclear abnormality only, no engulfment |
| Auer rods in blasts | Rod-shaped inclusions, not whole cells |
Practical Tip at the Bench
When you see a cell inside another cell on PBF:
- Focus up and down — overlapping cells will appear at different focal planes
- True emperipolesis — engulfed cell stays within the cytoplasm at all focal planes
- Check if engulfed cell is intact (emperipolesis) or broken down (phagocytosis/HLH)
- Scan multiple fields — count frequency before reporting
Reporting Template for PBF
"Emperipolesis noted — intact [neutrophils/lymphocytes] observed within the cytoplasm of monocytes/histiocytes. Frequency: [occasional/frequent]. Correlation with clinical history, ferritin, LFTs and bone marrow examination recommended to exclude HLH or Rosai-Dorfman disease."
Interpretation
Gel Card Interpretation — AHG Column
Reading the Image
| Column | Label | Finding | Grade |
|---|---|---|---|
| Left | AbId (Antibody Identification) | Single compact pellet at bottom, gel clear | Negative |
| Right | ICT | Agglutinates in middle of column + pellet at bottom | 1+ (Weak Positive) |
Interpretation
Left Column — AbId = NEGATIVE
- Complete cell button at bottom
- No agglutinates in gel
- No specific antibody identified in the antibody identification panel
- Suggests the antibody is not one of the common alloantibodies on the panel cells
Right Column — ICT = 1+ WEAK POSITIVE
- Agglutinates scattered in lower-mid portion of column
- Small residual pellet at bottom
- Confirms antibody is present in maternal serum
- But too weak to be identified on AbId panel
Clinical Correlation (Linking Back to Your Patient — G3 Rh Negative)
| Finding | Implication |
|---|---|
| ICT 1+ positive | Antibody present in maternal circulation |
| AbId negative | Antibody is below detection threshold of panel OR is a low-frequency antigen antibody not on standard panel |
| Combined result | Likely very early / low titer alloimmunization OR residual passive anti-D from RhoGAM |
Possible Explanations for This Pattern
-
Passive Anti-D from recent RhoGAM injection
- Titer too low to react with all panel cells
- ICT weakly positive, AbId negative = classic RhoGAM pattern
-
True early alloimmunization
- Anti-D forming but titer very low
- Not yet strong enough to identify on panel
-
Antibody to low-frequency antigen
- Not represented on standard panel cells
- Rare but possible
Next Steps
| Action | Reason |
|---|---|
| Check RhoGAM injection history | Rule out passive anti-D |
| Repeat ICT in 2–4 weeks | See if titer rising (true alloimmunization) |
| Perform antibody titration | Quantify antibody level |
| Enzyme technique (papain/ficin) | Enhance weak antibody reactivity for better identification |
| Extended panel if enzyme positive | Identify specific antibody |
Bottom Line
ICT 1+ with negative AbId = antibody is present but weak. In a G3 Rh-negative mother, this must be taken seriously — do not dismiss. Differentiate passive RhoGAM from true alloimmunization by clinical history and serial testing.