Thalassemia pathology for neet pg 2026 visual aod
Reading File
search_medical_text
thalassemia pathology types alpha beta molecular defects hemolysis ineffective erythropoiesis
search_medical_image
thalassemia pathology diagram blood smear target cells ineffective erythropoiesis
GradeResponse
GradeResponse
Here is a comprehensive NEET PG 2026-focused breakdown of Thalassemia Pathology — visual and high-yield.
🩸 Thalassemia — Pathology (NEET PG 2026)
Pathophysiology Diagram — Beta-Thalassemia
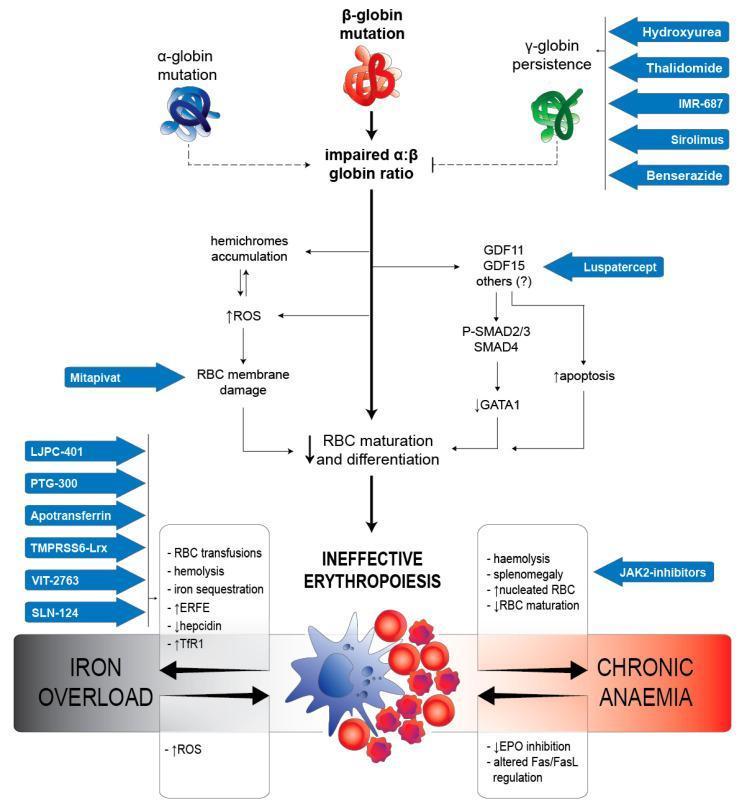
Molecular cascade from α:β imbalance → ineffective erythropoiesis → iron overload + chronic anemia
Core Concept: Globin Chain Imbalance
| Feature | β-Thalassemia | α-Thalassemia |
|---|---|---|
| Gene affected | β-globin gene (chr 11) | α-globin gene (chr 16) |
| Mutation type | Predominantly point mutations | Predominantly deletions |
| Excess chain | α-chains accumulate | β, γ, δ-chains accumulate |
| Tetramers formed | α₄ (Heinz bodies) | β₄ (HbH), γ₄ (Hb Barts) |
| O₂ affinity of tetramers | N/A (precipitate) | Very HIGH — do not deliver O₂ |
β-Thalassemia: Step-by-Step Pathology
Step 1 — Molecular Defect
- Mutations in β-globin gene → reduced (β⁺) or absent (β⁰) β-chain synthesis
- Result: Excess unpaired α-chains → cannot form stable tetramers → precipitate inside erythroblasts
Step 2 — Hemichrome Formation & ROS
- Precipitated α-chains → hemichromes → generate reactive oxygen species (ROS)
- ROS → membrane lipid peroxidation → erythroblast membrane damage
Step 3 — Ineffective Erythropoiesis ⭐ (Most Important Mechanism)
- Damaged erythroblasts are destroyed within the bone marrow before entering circulation
- This is called intramedullary hemolysis / ineffective erythropoiesis
- Predominant cause of anemia in β-thalassemia (Harrison's, p. 2927)
- GDF11/GDF15 signaling → ↑ p-SMAD2/3 → ↓ GATA1 → ↑ apoptosis of erythroid precursors
Step 4 — Peripheral Hemolysis (Minor Component)
- Cells that escape into circulation: ↓ deformability + phosphatidylserine exposure → extravascular + intravascular hemolysis
Consequences of Ineffective Erythropoiesis
Severe Anemia
↓
Marrow Expansion (erythroid hyperplasia)
↓
┌──────────────────────────┬──────────────────────────┐
│ Skeletal Changes │ Extramedullary │
│ • "Hair on end" │ Hematopoiesis │
│ skull X-ray │ • Hepatosplenomegaly │
│ • Crew-cut appearance │ • Paraspinal masses │
│ • Frontal bossing │ │
│ • Rodent facies │ │
└──────────────────────────┴──────────────────────────┘
↓
Iron Overload (↑ GI absorption + transfusions)
↓
Hemosiderosis → Liver cirrhosis, Dilated CMP,
Diabetes, Hypogonadism
Iron Overload Mechanism (High-Yield)
| Mediator | Role |
|---|---|
| ERFE (Erythroferrone) | ↑ in ineffective erythropoiesis → ↓ Hepcidin |
| Hepcidin | ↓ → ↑ Ferroportin activity → ↑ GI iron absorption |
| TfR1 (Transferrin Receptor 1) | ↑ on erythroblasts → increased iron uptake |
NEET PG Key Point: Ineffective erythropoiesis → ↑ ERFE → ↓ Hepcidin → Iron overload. This is independent of transfusions.
α-Thalassemia: Clinical Spectrum
| Genotype | Alleles deleted | Clinical Picture |
|---|---|---|
| Silent carrier | -α/αα (1 deleted) | Normal, asymptomatic |
| α-Thal trait | --/αα or -α/-α (2 deleted) | Mild microcytic anemia |
| HbH disease | --/-α (3 deleted) | Moderately severe hemolytic anemia, HbH (β₄ tetramers) |
| Hb Barts (Hydrops fetalis) | --/-- (4 deleted) | Incompatible with life, γ₄ tetramers, severe tissue hypoxia |
HbH inclusions stain with brilliant cresyl blue — "golf ball" cells
β-Thalassemia: Clinical Classification
| Type | Genotype | Hb level | Transfusion |
|---|---|---|---|
| Thalassemia Minor (Trait) | β/β⁺ or β/β⁰ | 9–11 g/dL | Not required |
| Thalassemia Intermedia | β⁺/β⁺ | 6–9 g/dL | Occasional |
| Thalassemia Major (Cooley's anemia) | β⁰/β⁰ | <6 g/dL | Regular transfusions |
Hemoglobin Electrophoresis Findings
| Condition | HbA | HbA2 | HbF |
|---|---|---|---|
| Normal | 97% | 2.5% | <1% |
| β-Thal Minor | ↓ | ↑ (>3.5%) ⭐ | ↑ slightly |
| β-Thal Major | Absent/↓↓ | ↑ | ↑↑ (up to 90%) |
| α-Thal trait | ↓ | Normal/↓ | Normal |
| HbH disease | ↓ | ↓ | Normal + HbH |
NEET PG Key: HbA2 >3.5% = hallmark of β-thalassemia trait. Used for carrier screening.
Peripheral Blood Smear Findings
- Microcytic hypochromic RBCs
- Target cells (codocytes) ⭐
- Nucleated RBCs (nRBCs) — due to marrow stress
- Basophilic stippling
- Poikilocytosis (tear drop cells, elliptocytes)
- Elevated reticulocyte count (though paradoxically low given the degree of anemia)
Complications Summary (Thalassemia Major)
| System | Complication |
|---|---|
| Cardiac | Dilated cardiomyopathy (iron) — #1 cause of death |
| Liver | Cirrhosis, hepatocellular carcinoma |
| Endocrine | Diabetes, hypogonadism, hypothyroidism, short stature |
| Bone | Osteoporosis, pathological fractures |
| Immune | Asplenia (post-splenectomy) → encapsulated organisms |
Treatment (NEET PG Essentials)
| Treatment | Details |
|---|---|
| Regular blood transfusion | Target Hb >10 g/dL; monthly |
| Iron chelation | Desferrioxamine (IV/SC), Deferasirox (oral), Deferiprone (oral) |
| Splenectomy | Reduces transfusion requirement; risk of sepsis |
| Hydroxyurea | ↑ HbF production (β-thal intermedia) |
| Luspatercept | Anti-GDF11 → ↓ ineffective erythropoiesis (newer drug) |
| HSCT (Bone Marrow Transplant) | Only curative option |
| Gene therapy | Betibeglogene (Zynteglo) — approved for β-thalassemia |
High-Yield One-Liners for NEET PG 2026
- Predominant mechanism of anemia in β-thalassemia = Ineffective erythropoiesis (not hemolysis)
- α-thalassemia = gene deletions; β-thalassemia = gene point mutations
- HbA2 >3.5% = β-thalassemia trait (carrier screening test)
- Hb Barts (γ₄) = 4-gene deletion α-thal = hydrops fetalis
- HbH (β₄) = 3-gene deletion = golf ball cells with BCB stain
- Ferritin ↑↑ in thalassemia major despite iron deficiency picture on smear (due to overload)
- Cardiac hemosiderosis = #1 cause of death in thalassemia major
- Curative treatment = Allogeneic HSCT
- Rodent facies + hair-on-end skull X-ray = thalassemia major (marrow expansion)
- Target cells on smear + microcytic anemia with normal/high serum iron = think thalassemia (not IDA)
Sources: Harrison's Principles of Internal Medicine, 21st Edition (p. 2927); PMC Clinical VQA — β-thalassemia pathophysiology diagram
This is a shared conversation. Sign in to Orris to start your own chat.