Acute lung injury full details for md exams 15marks
Reading File
search_medical_text
acute lung injury definition pathophysiology etiology ALI ARDS
search_medical_text
ARDS diagnostic criteria Berlin definition clinical features management
search_medical_image
acute lung injury ARDS chest X-ray pathology diagram
GradeResponse
GradeResponse
GradeResponse
search_medical_text
ARDS mechanical ventilation lung protective strategy PEEP treatment prognosis
I have sufficient material from the searches plus my own knowledge to deliver a comprehensive MD exam answer.
Acute Lung Injury (ALI) / ARDS — Comprehensive MD Exam Answer (15 Marks)
1. Definition & Historical Context
Acute Lung Injury (ALI) is an inflammatory syndrome characterised by non-cardiogenic pulmonary oedema, hypoxaemia, and bilateral pulmonary infiltrates resulting from increased alveolo-capillary permeability.
The American-European Consensus Conference (AECC, 1994) originally distinguished:
- ALI: PaO₂/FiO₂ ≤ 300 mmHg
- ARDS: PaO₂/FiO₂ ≤ 200 mmHg
The Berlin Definition (2012) replaced this classification and removed the separate ALI category. Today ARDS encompasses the full spectrum, stratified by oxygenation severity.
2. Berlin Definition of ARDS (2012)
(Harrison's, p. 8195; ARDS: Definition, Phenotyping and Respiratory Support Strategies, p. 2)
| Criterion | Requirement |
|---|---|
| Timing | Within 1 week of a known clinical insult or new/worsening respiratory symptoms |
| Chest imaging | Bilateral opacities not fully explained by effusions, collapse, or nodules |
| Origin of oedema | Respiratory failure not fully explained by cardiac failure or fluid overload |
| Oxygenation (on ≥5 cmH₂O PEEP) | See below |
Severity Stratification:
| Severity | PaO₂/FiO₂ | Mortality |
|---|---|---|
| Mild | 200–300 mmHg | ~27% |
| Moderate | 100–200 mmHg | ~32% |
| Severe | < 100 mmHg | ~45% |
3. Etiology / Causes
(Harrison's, p. 8195)
Causes are classified as direct (pulmonary) or indirect (extrapulmonary):
Direct (Pulmonary) Causes:
- Pneumonia (bacterial, viral, fungal) — most common
- Aspiration of gastric contents
- Pulmonary contusion
- Inhalation injury / toxic gas exposure
- Near-drowning
- Fat embolism
Indirect (Extrapulmonary) Causes:
- Sepsis — most common overall cause
- Severe trauma / polytrauma
- Burns
- Pancreatitis (acute severe)
- Massive transfusion / transfusion-related ALI (TRALI)
- Cardiopulmonary bypass
- Drug overdose (heroin, aspirin, barbiturates)
- DIC
Mnemonic for causes: "ARDS PAIN" Aspiration, Radiation, Drug/DIC, Sepsis, Pancreatitis, ALI-TRALI, Infection/Inhalation, Near-drowning
4. Pathophysiology
ARDS proceeds through three overlapping phases:
Phase 1 — Exudative Phase (Day 1–7)
- Injury to type I pneumocytes and pulmonary capillary endothelium
- Increased alveolo-capillary permeability → protein-rich exudate floods alveoli
- Neutrophil activation → release of proteases, reactive oxygen species (ROS), inflammatory cytokines (IL-1β, IL-6, TNF-α)
- Hyaline membrane formation (fibrin + plasma proteins)
- Diffuse alveolar damage (DAD) — pathological hallmark
- Surfactant dysfunction → alveolar collapse, atelectasis
- Results in: Hypoxaemia, reduced compliance, V/Q mismatch + intrapulmonary shunt
Phase 2 — Proliferative Phase (Day 7–21)
- Type II pneumocyte proliferation attempts repair
- Organisation of exudate; fibroblast invasion
- Most patients improve; some progress to fibrosis
Phase 3 — Fibrotic Phase (>21 days, minority)
- Progressive collagen deposition → pulmonary fibrosis
- Bullae formation → increased risk of pneumothorax
- Pulmonary hypertension
Key Pathophysiological Consequences:
| Mechanism | Effect |
|---|---|
| Increased permeability | Non-cardiogenic pulmonary oedema |
| Surfactant loss | Alveolar collapse, atelectasis |
| Intrapulmonary shunt | Refractory hypoxaemia |
| Reduced compliance | Increased work of breathing |
| Dead-space ventilation | Hypercapnia |
| Pulmonary vasoconstriction | Pulmonary hypertension, right heart strain |
5. Clinical Features
Symptoms:
- Severe dyspnoea of rapid onset (within hours to days of insult)
- Dry cough
- Restlessness, anxiety, confusion (hypoxaemia)
Signs:
- Tachypnoea (RR >30/min)
- Tachycardia
- Cyanosis (central)
- Use of accessory muscles
- Bilateral crepitations on auscultation
- Features of the underlying cause (fever in sepsis, abdominal tenderness in pancreatitis)
- Progression to respiratory failure requiring mechanical ventilation
6. Investigations
1. Arterial Blood Gas (ABG):
- Hypoxaemia: PaO₂/FiO₂ ratio ≤ 300 (refractory to supplemental O₂)
- Early: Respiratory alkalosis (hyperventilation)
- Late: Respiratory acidosis (fatigue, failure)
2. Chest X-Ray (CXR):
- Bilateral diffuse airspace opacities ("white-out")
- No cardiomegaly, no pleural effusions (distinguishes from cardiogenic oedema)
- Typically spares costophrenic angles early
3. CT Chest (Gold Standard Imaging):
- Dependent consolidation + non-dependent ground-glass opacities
- "Baby lung" concept — only ~30% aerated lung in severe ARDS
- Helps identify pneumothorax, fibrosis, underlying cause
4. Echocardiography:
- Excludes left heart failure (PCWP < 18 mmHg in ARDS)
- Assesses right ventricular strain (acute cor pulmonale)
5. Labs:
- CBC: Leucocytosis / leucopenia (sepsis)
- Procalcitonin, CRP (infection)
- Cultures (blood, sputum, BAL)
- LFT, RFT (multi-organ dysfunction)
- Coagulation profile (DIC)
- Serum amylase/lipase (pancreatitis)
7. ARDS CXR — Imaging Example
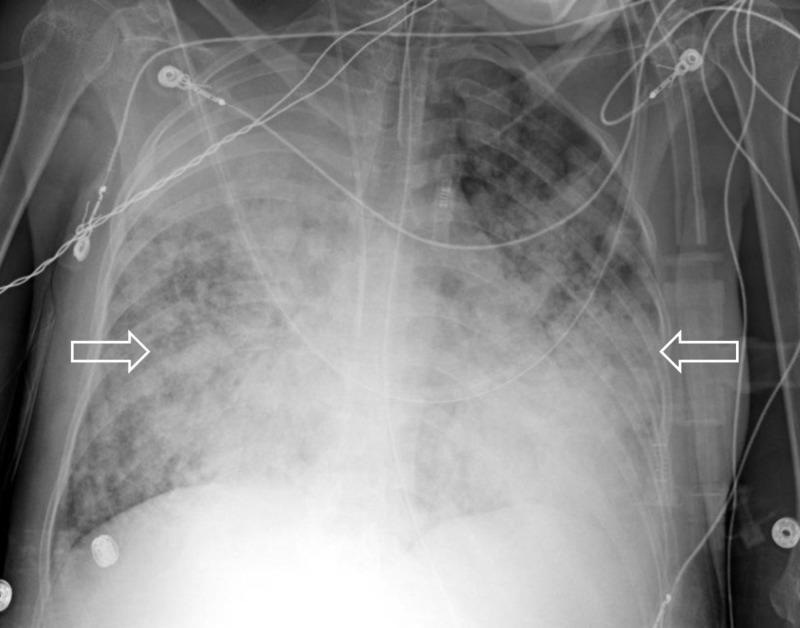
AP chest X-ray showing diffuse bilateral airspace opacities with ground-glass appearance and consolidation, typical of severe ARDS. Note endotracheal tube and central venous catheter in situ — consistent with ICU management.
8. Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| Cardiogenic pulmonary oedema | Raised JVP, S3 gallop, cardiomegaly on CXR, PCWP >18, responds to diuretics |
| Bilateral pneumonia | Fever + productive cough, unilateral or lobar pattern possible |
| Pulmonary haemorrhage | Haemoptysis, anaemia, renal involvement (Goodpasture's) |
| Cryptogenic organising pneumonia | Subacute onset, responds to steroids |
| Acute eosinophilic pneumonia | Peripheral eosinophilia, BAL >25% eosinophils |
9. Management
Management is supportive — treat the underlying cause + prevent ventilator-induced lung injury (VILI).
A. Treat Underlying Cause:
- Antibiotics for sepsis/pneumonia
- Source control (drainage, surgery)
- Discontinue offending drug
B. Lung-Protective Mechanical Ventilation (Cornerstone)
Based on ARDSNet ARMA Trial (2000):
| Parameter | Target |
|---|---|
| Tidal Volume (Vt) | 6 mL/kg predicted body weight (low Vt strategy) |
| Plateau Pressure | ≤ 30 cmH₂O |
| PEEP | Titrated (typically 5–15 cmH₂O) to optimise oxygenation |
| FiO₂ | Minimum to achieve SpO₂ 88–95% |
| RR | Up to 35/min; permissive hypercapnia acceptable |
| pH target | ≥ 7.25 (permissive hypercapnia) |
Low Vt ventilation reduced mortality by 22% (ARDSNet trial)
C. Prone Positioning
- For severe ARDS (PaO₂/FiO₂ < 150) — ≥ 16 hours/day
- PROSEVA Trial (2013): 28-day mortality reduced from 32.8% → 16%
- Mechanism: Recruits dependent atelectatic lung, improves V/Q matching, reduces VILI
D. PEEP Optimisation
- Higher PEEP strategy in moderate-severe ARDS (ALVEOLI, LOVS, ExPress trials)
- Prevents alveolar de-recruitment at end-expiration
- Must balance: alveolar recruitment vs. overdistension
E. Neuromuscular Blockade (NMB)
- Cisatracurium infusion for 48 hours in moderate-severe ARDS
- ACURASYS Trial: Improved 90-day survival, reduced barotrauma
- Note: ROSE trial (2019) did not confirm benefit — still debated
F. Corticosteroids
- Methylprednisolone in select cases:
- Early moderate-severe ARDS (within 14 days): May reduce duration of MV
- COVID-19 ARDS: Dexamethasone 6 mg/day × 10 days (RECOVERY trial) — mortality benefit
- Late ARDS (>14 days): Avoid — worsens outcomes
G. Conservative Fluid Management
- FACTT Trial (ARDSNet): Conservative fluid strategy → fewer ventilator days, no increase in renal failure
- Target: Euvolaemia / slight negative fluid balance
- Avoid fluid overload (worsens pulmonary oedema)
H. Rescue Therapies (Refractory ARDS):
| Therapy | Evidence |
|---|---|
| ECMO (VV-ECMO) | CESAR trial, EOLIA trial — benefit in severe refractory ARDS at expert centres |
| Inhaled Nitric Oxide (iNO) | Improves oxygenation transiently; no mortality benefit |
| Inhaled Prostacyclin | Pulmonary vasodilator; limited evidence |
| High-frequency oscillatory ventilation (HFOV) | OSCAR + OSCILLATE trials — no benefit; OSCILLATE showed harm |
| Surfactant therapy | Benefit in neonates; not proven in adults |
| Recruitment manoeuvres | Transient oxygenation improvement; ART trial showed harm with aggressive RM |
I. General ICU Supportive Care:
- Nutritional support (enteral preferred, early)
- VTE prophylaxis
- Stress ulcer prophylaxis
- Glycaemic control
- Minimise sedation (SAT/SBT protocols)
- Head-of-bed elevation (30–45°) to prevent VAP
10. Complications
| Complication | Notes |
|---|---|
| Ventilator-Associated Pneumonia (VAP) | Most common infectious complication |
| Barotrauma | Pneumothorax, pneumomediastinum — from high airway pressures |
| Oxygen toxicity | Avoid FiO₂ > 0.6 for prolonged periods |
| Multi-Organ Dysfunction Syndrome (MODS) | Kidneys, liver, coagulation |
| Pulmonary hypertension / Cor pulmonale | Chronic hypoxic vasoconstriction |
| ICU-acquired weakness | Prolonged immobility, NMB use |
| Pulmonary fibrosis | Late-phase complication |
| PTSD / Cognitive impairment | Long-term sequelae in survivors |
11. Prognosis
(Harrison's, p. 8195)
- Overall mortality: ~40% (ranges 27–45% by severity)
- Most deaths are from multi-organ failure, not hypoxaemia per se
- Survivors: ~50–70% regain near-normal lung function by 6–12 months
- Predictors of poor outcome: Age > 60, severe hypoxaemia, non-pulmonary organ failure, sepsis as cause, fibroproliferative phase
12. Summary Table for Quick Revision
| Feature | Detail |
|---|---|
| Definition | Bilateral infiltrates + PaO₂/FiO₂ ≤ 300 on PEEP ≥5, within 1 week, not fully cardiac |
| Most common cause | Sepsis (indirect), Pneumonia (direct) |
| Pathology | Diffuse alveolar damage (DAD) |
| Key phases | Exudative → Proliferative → Fibrotic |
| Investigation | ABG (PaO₂/FiO₂), CXR, CT, Echo |
| Cornerstone Rx | Low Vt (6 mL/kg) ventilation |
| Mortality benefit (proven) | Prone positioning (PROSEVA), Low Vt (ARDSNet), Dexamethasone in COVID-ARDS |
| Prognosis | ~40% mortality overall |
References:
- Harrison's Principles of Internal Medicine, 21st Edition, p. 8195
- Acute Respiratory Distress Syndrome: Definition, Phenotyping and Respiratory Support Strategies, p. 2 (ESICM/ERS Guidelines)
- ARDSNet ARMA Trial (2000), PROSEVA Trial (2013), FACTT Trial, RECOVERY Trial (2020)
This is a shared conversation. Sign in to Orris to start your own chat.