Ards and ventilation statergy full details for md exams 15marks
Reading File
search_medical_text
ARDS acute respiratory distress syndrome definition pathophysiology Berlin criteria diagnosis
search_medical_text
ARDS lung protective ventilation strategy low tidal volume PEEP plateau pressure ARDSNet protocol
search_medical_image
ARDS chest X-ray bilateral infiltrates ventilator settings
GradeResponse
GradeResponse
GradeResponse
Now I have strong core content. Let me compile the full comprehensive answer for MD exam standards.
ARDS and Ventilation Strategy — Complete Answer (15 Marks)
1. DEFINITION
Acute Respiratory Distress Syndrome (ARDS) is an acute, diffuse, inflammatory lung injury characterized by:
- Increased alveolo-capillary permeability → non-cardiogenic pulmonary edema
- Decreased lung compliance (increased lung elastance)
- Severe hypoxemia with increased venous admixture and dead space
(ARDS: Definition, Phenotyping and Respiratory Support Strategies, p. 2)
2. BERLIN DEFINITION (2012) — Diagnostic Criteria
| Criterion | Specification |
|---|---|
| Timing | Within 1 week of known clinical insult OR new/worsening respiratory symptoms |
| Chest Imaging | Bilateral opacities on CXR or CT — not fully explained by effusions, lobar/lung collapse, or nodules |
| Origin of Edema | Respiratory failure NOT fully explained by cardiac failure or fluid overload (Echo if no risk factor) |
| Oxygenation (on PEEP ≥ 5 cmH₂O) | Mild / Moderate / Severe (see below) |
Severity Classification (Berlin)
| Category | PaO₂/FiO₂ Ratio | PEEP Required | Mortality |
|---|---|---|---|
| Mild | 201–300 mmHg | ≥ 5 cmH₂O | ~27% |
| Moderate | 101–200 mmHg | ≥ 5 cmH₂O | ~32% |
| Severe | ≤ 100 mmHg | ≥ 5 cmH₂O | ~45% |
Key point for exams: PEEP ≥ 5 cmH₂O is MANDATORY at time of diagnosis; patients NOT on positive pressure ventilation technically cannot be classified as ARDS by Berlin criteria.
3. ETIOLOGY
Direct (Pulmonary) Causes
- Pneumonia (bacterial, viral — including COVID-19)
- Aspiration of gastric contents
- Pulmonary contusion
- Inhalation injury
- Near-drowning
Indirect (Extrapulmonary) Causes
- Sepsis (most common overall cause — ~40%)
- Severe trauma with shock
- Pancreatitis
- Burns
- Massive blood transfusion / TRALI
- Drug overdose (heroin, aspirin)
4. PATHOPHYSIOLOGY
ARDS progresses through three overlapping phases:
Phase 1: Exudative (Day 1–7)
- Activation of alveolar macrophages and neutrophils
- Release of cytokines (IL-1, IL-6, IL-8, TNF-α)
- Damage to Type I pneumocytes → loss of alveolar epithelial barrier
- Damage to capillary endothelium → protein-rich fluid floods alveoli
- Hyaline membrane formation — pathognomonic
- Surfactant depletion → alveolar collapse
Phase 2: Proliferative (Day 7–21)
- Type II pneumocyte proliferation (attempt at repair)
- Fibroblast activation → early fibrosis
- Resolution of edema begins
- Clinical improvement or progression to fibrosis
Phase 3: Fibrotic (>21 days)
- Irreversible fibrosis in some patients
- Obliteration of alveolar architecture
- Increased dead space → CO₂ retention
- Predisposition to barotrauma
5. CLINICAL FEATURES
- Onset: Within 12–48 hours of precipitating event
- Dyspnea at rest, rapidly progressive
- Tachypnea, accessory muscle use
- Hypoxemia — refractory to supplemental O₂ (hallmark)
- Diffuse crackles on auscultation
- CXR: Bilateral alveolar infiltrates ("white-out" lungs)
- No clinical evidence of left heart failure (PCWP < 18 mmHg)
Chest X-Ray / CT Findings in ARDS
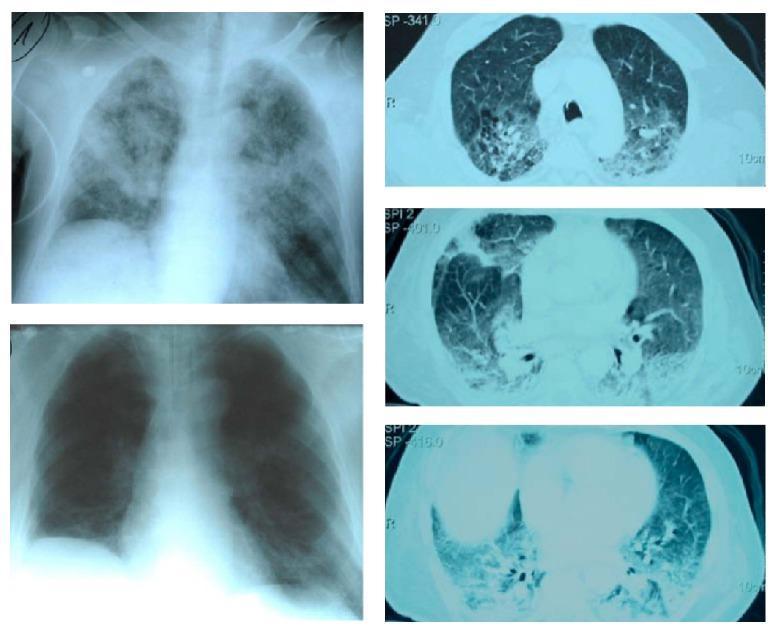
6. VENTILATION STRATEGY IN ARDS ⭐ (Most Important for MD Exam)
The cornerstone is Lung-Protective Ventilation (LPV) — designed to prevent ventilator-induced lung injury (VILI).
A. LUNG-PROTECTIVE VENTILATION (ARDSNet Protocol)
Tidal Volume (Vt)
- Target: 6 mL/kg Predicted Body Weight (PBW)
- Can reduce to 4 mL/kg PBW if plateau pressure remains > 30 cmH₂O
- AVOID: Vt > 6 mL/kg + Plateau > 30 cmH₂O
- Very low Vt may cause patient-ventilator dyssynchrony
Predicted Body Weight (PBW) Calculation
- Males: 50 + 2.3 × [height(in) − 60]
- Females: 45.5 + 2.3 × [height(in) − 60]
Plateau Pressure
- Target: ≤ 30 cmH₂O (measured during inspiratory hold, patient must be passive)
- In patients with high chest wall compliance (obesity, ascites), higher plateau pressures may be tolerated as transpulmonary pressure remains low
Driving Pressure (DP)
- DP = Plateau Pressure − PEEP
- Target: < 12–15 cmH₂O (strong predictor of mortality)
- Recent evidence suggests DP may be a better titration target than Vt alone
- (Surviving Sepsis Campaign 2021, p. 34)
PEEP (Positive End-Expiratory Pressure)
- Minimum 5 cmH₂O (required for Berlin diagnosis)
- Higher PEEP strategy for moderate-severe ARDS (PaO₂/FiO₂ < 200)
- Titrated using ARDSNet PEEP/FiO₂ tables or by esophageal manometry
ARDSNet PEEP/FiO₂ Table (Lower PEEP Strategy):
| FiO₂ | 0.3 | 0.4 | 0.5 | 0.6 | 0.7 | 0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|
| PEEP | 5 | 5–8 | 8–10 | 10 | 10–12 | 12–14 | 14–18 | 18–24 |
Respiratory Rate (RR)
- Increase RR up to 35 breaths/min to compensate for low Vt
- Maintain acceptable minute ventilation
FiO₂
- Target PaO₂: 55–80 mmHg (SpO₂: 88–95%)
- Avoid hyperoxia (FiO₂ > 0.6 for prolonged periods is toxic)
pH / Permissive Hypercapnia
- Accept pH ≥ 7.20 (permissive hypercapnia)
- If pH < 7.20: increase RR → consider sodium bicarbonate → increase Vt as last resort
- Contraindicated in raised ICP, severe pulmonary hypertension, right heart failure
B. MODE OF VENTILATION
- Volume-controlled AC (Assist Control) — most commonly used; ensures consistent Vt
- Pressure-controlled ventilation — used when precise Vt delivery is secondary to pressure limitation
- PRVC (Pressure-Regulated Volume Control) — combines benefits of both
C. PRONE POSITIONING ⭐
- Recommended for severe ARDS (PaO₂/FiO₂ < 150) unresponsive to conventional ventilation
- Duration: ≥ 16 hours/day (PROSEVA trial — 28% relative mortality reduction)
- Mechanism:
- Redistributes lung edema
- Recruits dorsal (dependent) atelectatic alveoli
- Improves V/Q matching
- Reduces lung stress/strain
- Contraindications: spinal injury, open chest/abdomen, hemodynamic instability, raised ICP
D. NEUROMUSCULAR BLOCKADE (NMB)
- Short-term NMB (cisatracurium for 48 hours) reduces mortality in moderate-severe ARDS
- Improves patient-ventilator synchrony
- Reduces O₂ consumption and cytokine release
- ACURASYS trial: benefit shown; ROSE trial: no benefit with lighter sedation baseline
- Current practice: consider NMB when dyssynchrony persists despite deep sedation
E. RECRUITMENT MANEUVERS (RM)
- Transient increase in airway pressure to open collapsed alveoli
- Methods: sustained inflation (40 cmH₂O × 40 sec), incremental PEEP, sighs
- ART trial (2017): high-pressure RM + PEEP titration strategy increased mortality → NOT routinely recommended
- Use with caution in hemodynamically unstable patients
F. HIGH-FREQUENCY OSCILLATORY VENTILATION (HFOV)
- Previously used for severe ARDS refractory to conventional LPV
- OSCILLATE and OSCAR trials (2013): HFOV showed increased mortality or no benefit
- Currently NOT recommended as routine strategy (may be used as rescue in select centers)
G. EXTRACORPOREAL MEMBRANE OXYGENATION (ECMO)
- VV-ECMO (veno-venous) for severe ARDS refractory to all conventional strategies
- CESAR trial and EOLIA trial support its use in severe ARDS (PaO₂/FiO₂ < 80 on optimal settings)
- Allows "ultra-lung-protective ventilation" while ECMO handles gas exchange
- Requires specialized center, high resources
H. OTHER ADJUNCT THERAPIES
| Therapy | Evidence |
|---|---|
| Inhaled nitric oxide (iNO) | Improves oxygenation transiently; no mortality benefit; bridge to ECMO |
| Inhaled prostacyclins | Vasodilator; improves V/Q; no survival benefit |
| Corticosteroids | Methylprednisolone in early/mid-phase ARDS may reduce duration of MV; avoid in late fibrotic phase |
| Conservative fluid strategy | Target negative/neutral fluid balance; improves oxygenation and ventilator-free days (FACTT trial) |
| Exogenous surfactant | No benefit in adults |
7. SUMMARY TABLE — VENTILATION TARGETS IN ARDS
| Parameter | Target |
|---|---|
| Tidal Volume | 6 mL/kg PBW (reduce to 4 if needed) |
| Plateau Pressure | ≤ 30 cmH₂O |
| Driving Pressure | < 12–15 cmH₂O |
| PEEP | ≥ 5 cmH₂O; titrate per FiO₂/PEEP table |
| SpO₂ | 88–95% |
| PaO₂ | 55–80 mmHg |
| pH | ≥ 7.20 (permissive hypercapnia allowed) |
| Respiratory Rate | Up to 35/min |
| Prone Position | ≥ 16 hrs/day if PaO₂/FiO₂ < 150 |
8. COMPLICATIONS OF ARDS
- Ventilator-induced lung injury (VILI): volutrauma, barotrauma, atelectrauma, biotrauma
- Pneumothorax / pneumomediastinum
- Ventilator-associated pneumonia (VAP)
- Multi-organ dysfunction syndrome (MODS)
- Post-ARDS fibrosis
- Neuromuscular weakness (ICU-acquired weakness)
- Cognitive impairment / PTSD (long-term)
9. PROGNOSIS
- Overall mortality: 35–45% (varies with severity and etiology)
- Sepsis-related ARDS has the worst prognosis
- Survivors may have long-term pulmonary, neurocognitive, and psychological sequelae (Post-Intensive Care Syndrome)
- Lung function typically recovers within 6–12 months in survivors
EXAM HIGH-YIELD POINTS 📌
- Berlin 2012 replaced the American-European Consensus Conference (AECC) 1994 definition — no longer uses "ALI" terminology
- PEEP ≥ 5 cmH₂O is mandatory for Berlin classification
- ARDSNet trial (2000): 6 mL/kg Vt vs. 12 mL/kg → 22% relative mortality reduction
- Driving pressure < 15 cmH₂O is the most predictive parameter of outcome
- PROSEVA trial: prone ≥ 16 hrs → gold standard for severe ARDS
- HFOV is harmful (OSCILLATE/OSCAR); ECMO is last resort
- Conservative fluid strategy preferred (negative fluid balance when hemodynamically stable)
- Permissive hypercapnia is accepted as long as pH ≥ 7.20
- Avoid hyperoxia — target SpO₂ 88–95%, not 100%
- Corticosteroids benefit in early/mid ARDS; NOT in late fibrotic phase
Sources: ARDS Definition, Phenotyping and Respiratory Support Strategies (p. 2); Surviving Sepsis Campaign 2021 (p. 34); ARDSNet Protocol; PROSEVA, EOLIA, ACURASYS, FACTT, ART trials.
This is a shared conversation. Sign in to Orris to start your own chat.